Key Takeaways
- Core idea: The Third Law states that the entropy of a perfect crystal approaches zero as temperature approaches absolute zero.
- Engineering use: It gives engineers a reference point for absolute entropy and explains why cryogenic cooling becomes harder near 0 K.
- What controls it: Temperature, molecular order, crystal defects, accessible microstates, and heat capacity control how real materials behave.
- Practical check: Do not assume every material has zero entropy at 0 K; real crystals can have defects, disorder, or residual entropy.
Table of Contents
Introduction
The Third Law of Thermodynamics states that the entropy of a perfect crystal approaches zero as its temperature approaches absolute zero, or 0 K. For mechanical engineers, this law explains why low-temperature systems become difficult to cool further and why entropy can be treated as an absolute thermodynamic property rather than only a relative change.
Third Law of Thermodynamics Diagram
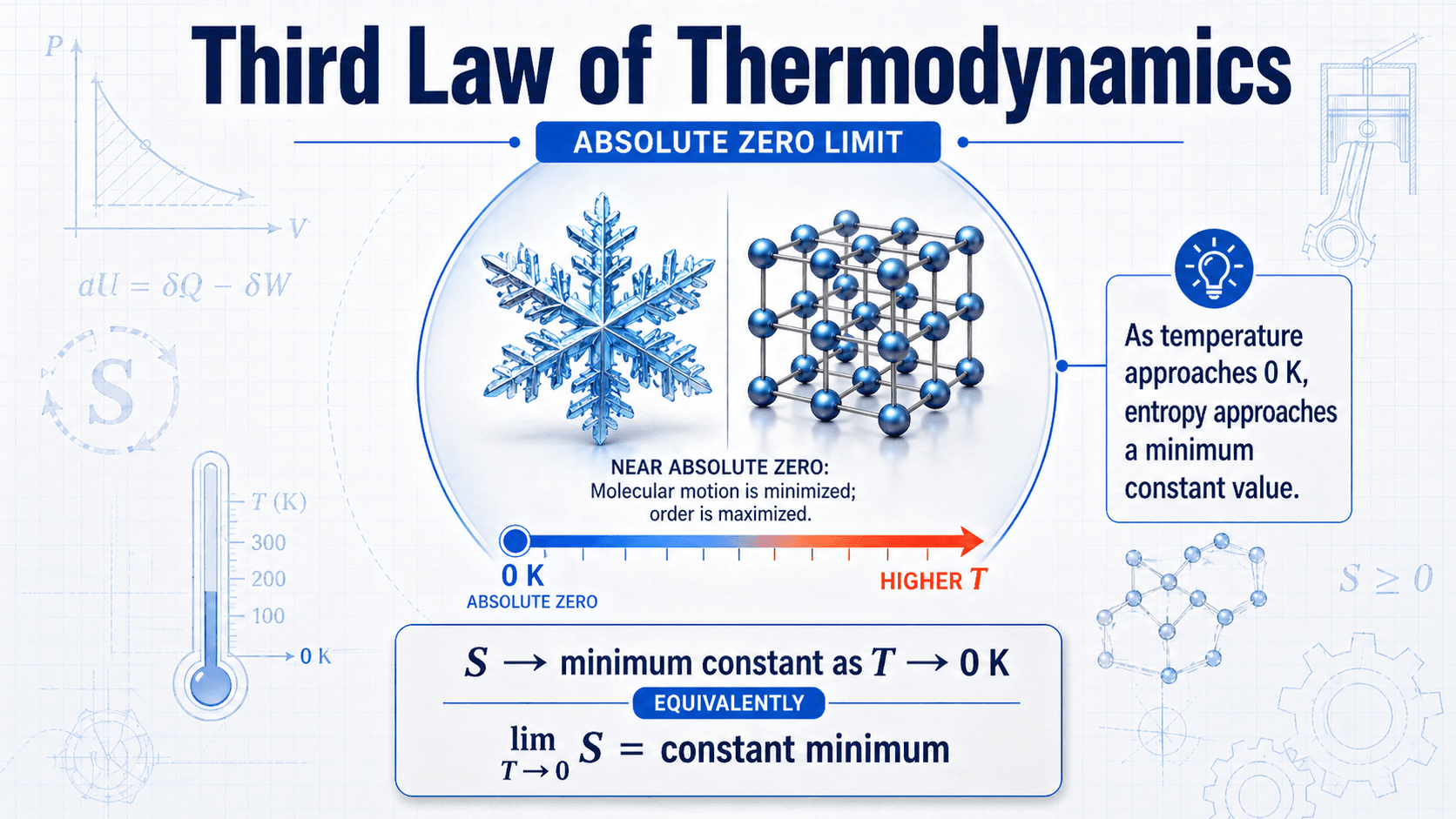
Notice that the law is about a limiting condition, not an ordinary room-temperature process. The closer a material gets to 0 K, the more important molecular order, quantum behavior, heat capacity, and crystal defects become.
What is the Third Law of Thermodynamics?
The Third Law of Thermodynamics defines the limiting behavior of entropy as temperature approaches absolute zero. In its common engineering and chemistry form, it says that the entropy of a pure, perfectly ordered crystalline substance approaches zero as the temperature approaches 0 K.
This matters because entropy is not just a classroom idea about “disorder.” In thermodynamics, entropy is a property used to analyze process direction, unavailable energy, heat transfer limits, material behavior, and equilibrium. The Third Law gives entropy a meaningful zero reference, which helps engineers and scientists compare absolute entropy values rather than only entropy changes.
The Third Law does not say that all real materials have exactly zero entropy at 0 K. It applies cleanly to an ideal perfect crystal. Real materials may contain defects, impurities, frozen-in disorder, or multiple low-energy arrangements.
How Entropy Changes as Temperature Approaches Absolute Zero
As a material cools, its atoms and molecules have less thermal energy. Vibrations decrease, fewer microscopic arrangements are accessible, and the system moves toward a more ordered state. For an ideal perfect crystal, the lowest-temperature state has only one perfectly ordered arrangement, so entropy approaches zero.
Entropy and microstates
A useful way to understand entropy is through microstates. A microstate is one possible microscopic arrangement of the particles and energy in a system. Hotter systems usually have many accessible microstates. A perfectly ordered crystal at absolute zero has one ideal arrangement, which is why the entropy reference becomes zero.
Absolute zero as a limit
Absolute zero is 0 K, or −273.15°C. It represents the lower limit of thermodynamic temperature, but it is not a normal operating point that engineers can simply reach with a larger refrigerator. As temperature falls, each additional reduction becomes more difficult because the system has less removable thermal energy and fewer accessible energy states.
Perfect crystal versus real material
The phrase “perfect crystal” is doing important work. It means every particle is arranged in a completely ordered structure with no defects, substitutions, vacancies, or orientation disorder. Real materials are rarely perfect, especially after manufacturing, thermal cycling, phase changes, or exposure to impurities.
Why the Third Law Matters in Mechanical Engineering
Most mechanical engineering projects do not operate near absolute zero, but the Third Law still shapes how engineers think about low-temperature systems, thermodynamic property tables, refrigeration limits, cryogenic equipment, and materials that behave differently when thermal motion becomes very small.
- Cryogenic systems: Liquid helium systems, low-temperature storage, and research equipment depend on careful control of heat leaks and thermal gradients.
- Superconducting equipment: Magnets, sensors, and quantum devices often rely on material behavior that appears only at very low temperatures.
- Material selection: Thermal contraction, heat capacity, brittleness, and phase stability can change significantly as temperature drops.
- Thermodynamic property work: The law supports absolute entropy values used in chemistry, energy analysis, and equilibrium calculations.
If a design depends on low-temperature behavior, check more than the final temperature. Review heat leak paths, cooldown rate, thermal contraction, material compatibility, insulation performance, and whether the property data remain valid near the operating range.
Key Factors That Control Low-Temperature Entropy Behavior
The Third Law is often stated in one sentence, but real engineering interpretation depends on material condition, temperature range, and whether the system behaves close to the ideal crystal assumption.
| Factor | Why it matters | Engineering implication |
|---|---|---|
| Temperature | Entropy generally decreases as a material is cooled toward 0 K. | Low-temperature systems require increasingly precise heat removal and insulation as temperature decreases. |
| Crystal order | A perfect crystal has one ideal arrangement at absolute zero. | Defects, vacancies, impurities, and orientation disorder can prevent real materials from matching the ideal Third Law model. |
| Heat capacity | Heat capacity typically decreases at very low temperatures. | Small heat inputs can matter, and property data must be appropriate for the low-temperature range. |
| Residual entropy | Some materials retain disorder even near 0 K. | Engineers should avoid assuming zero entropy for materials with frozen-in disorder or multiple equivalent low-energy configurations. |
| Thermal isolation | Heat leaks become more significant when the target temperature is extremely low. | Supports, wiring, radiation, seals, and instrumentation can dominate cryogenic performance. |
Third Law Formula and Entropy Reference
The Third Law is usually expressed as a limiting statement. For a pure, perfect crystalline substance, entropy approaches zero as temperature approaches absolute zero.
This equation is not a design equation in the same way as an energy balance or heat-transfer correlation. It is a reference condition for entropy. It tells the engineer or scientist where absolute entropy is anchored for the ideal case.
The Boltzmann entropy relationship helps explain the idea conceptually. If a perfect crystal at 0 K has one accessible microstate, then \(\Omega = 1\), and \(\ln(1) = 0\). That gives the ideal entropy value of zero.
- S Entropy, commonly expressed in J/K for a system or J/(mol·K) for molar entropy.
- T Absolute temperature in kelvin, where 0 K is absolute zero.
- k_B Boltzmann constant, relating microscopic states to entropy.
- Ω Number of accessible microstates for the system.
Low-Temperature Entropy Sanity Check
Use this quick review path when applying the Third Law to a real material, property table, cryogenic design, or low-temperature thermodynamics problem.
Start with the ideal statement: perfect crystal, pure substance, 0 K, zero entropy. Then ask whether the real system matches that ideal. If the material has defects, impurities, disorder, phase transitions, magnetic effects, or uncertain low-temperature data, treat the ideal Third Law statement as a reference concept rather than a complete design model.
| Check or decision | What to look for | Why it matters |
|---|---|---|
| Is the substance idealized as a perfect crystal? | Pure crystalline structure with no meaningful disorder in the model. | The zero-entropy statement depends on this ideal condition. |
| Is the temperature near the range where low-temperature effects dominate? | Kelvin-scale or cryogenic operation rather than ordinary ambient conditions. | Heat capacity, thermal contraction, and quantum effects may become important. |
| Are property values from a valid source and range? | Entropy, heat capacity, and phase data appropriate for the actual temperature range. | Extrapolating room-temperature data toward 0 K can produce misleading results. |
| Could residual entropy be present? | Orientation disorder, defects, glassy behavior, mixed phases, or frozen-in configurations. | Real materials may not reach the ideal zero-entropy condition even near absolute zero. |
| Are heat leaks controlled? | Conduction through supports, radiation, wiring, penetrations, and instrumentation. | At very low temperatures, tiny heat inputs can dominate system performance. |
Example: Why Cooling Gets Harder Near 0 K
Consider a cryogenic test setup that must cool a small material sample from room temperature down to a few kelvin. Early in the cooldown, the sample contains a large amount of removable thermal energy, and the temperature may drop quickly. Near the final temperature, each additional reduction is harder because the thermal energy scale is much smaller and heat leaks become more influential.
Engineering scenario
A sample holder, wiring, fasteners, insulation, sensors, and support structure all create possible heat paths into the cold region. Even if the cooling equipment is powerful, uncontrolled conduction or radiation can prevent the system from reaching the desired low-temperature operating point.
Interpretation
The Third Law explains the limiting thermodynamic direction: absolute zero is approached, not reached. Engineering design then adds the practical layer: heat leaks, material contraction, instrumentation, cooldown time, and data validity often control whether the system performs as expected.
Engineering Judgment and Field Reality
In real mechanical systems, the Third Law is rarely used alone. It works with entropy calculations, heat-transfer analysis, material data, cryogenic insulation design, and equipment constraints. The clean textbook statement is valuable because it gives a limiting reference, but practical engineering depends on how closely the system resembles the ideal assumptions.
The hardest part of low-temperature design is often not understanding that entropy approaches zero. It is controlling the details that keep heat out: supports, wiring, seals, radiation shields, test fixtures, measurement leads, and thermal contact resistance.
Experienced engineers also watch for material behavior that changes as temperature falls. Metals may contract, polymers may become brittle, lubricants may fail, seals may stiffen, and sensors may require recalibration. A thermodynamic limit can become a mechanical reliability problem if the whole system is not reviewed.
When This Breaks Down
The simplified version of the Third Law breaks down when readers treat the ideal perfect-crystal statement as if it automatically describes every real material, every phase, or every engineering system.
- Imperfect crystals: Defects, vacancies, impurities, and dislocations can keep the microscopic arrangement from being perfectly ordered.
- Glasses and amorphous materials: Non-crystalline solids may retain frozen-in disorder that does not match the perfect-crystal assumption.
- Residual entropy: Some materials have multiple equivalent low-energy arrangements, so entropy may not reduce to the ideal value.
- Unverified property extrapolation: Extending data far outside its measured range can create false confidence in entropy or heat-capacity calculations.
- Practical cooling limits: Equipment, insulation, and heat leaks often control achievable temperature long before theory is the only issue.
Common Mistakes and Practical Checks
Most confusion around the Third Law comes from mixing a precise thermodynamic limit with everyday language about cold objects, molecular motion, and disorder.
- Assuming absolute zero is reachable: The law describes an approach to 0 K, not a practical path to exactly reach it.
- Ignoring the word “perfect”: The zero-entropy statement applies to an ideal perfect crystal, not automatically to real materials.
- Confusing low entropy with no engineering risk: Low-temperature systems can create severe material, sealing, measurement, and safety challenges.
- Using Celsius in thermodynamic limits: Third Law temperature reasoning should use kelvin, not Celsius or Fahrenheit.
- Forgetting heat leaks: At cryogenic temperatures, small heat inputs through wires, mounts, or radiation can dominate performance.
Do not describe the Third Law as “all molecular motion stops at absolute zero.” Quantum effects and the idealized nature of absolute zero make that wording misleading. It is better to say entropy approaches its minimum for a perfect crystal as temperature approaches 0 K.
Useful References and Engineering Context
The Third Law is a foundational thermodynamics concept, so the most useful references are thermodynamics textbooks, property data sources, and low-temperature engineering references rather than building codes or design standards.
- Thermodynamics textbooks: Used to define entropy, absolute temperature, heat capacity, statistical interpretation, and the formal statement of the Third Law.
- Thermodynamic property tables: Used when engineers need entropy, enthalpy, internal energy, and heat-capacity values over a defined temperature range.
- Cryogenic engineering references: Used for practical design issues such as insulation, radiation shielding, cooldown procedures, heat leaks, and material compatibility at low temperature.
- Material data sheets: Used to confirm thermal contraction, brittleness, conductivity, and heat capacity before applying low-temperature assumptions to real components.
Frequently Asked Questions
The Third Law of Thermodynamics says that the entropy of a perfect crystal approaches zero as its temperature approaches absolute zero. In simple terms, as a perfectly ordered material gets colder, its microscopic disorder approaches the lowest possible value.
Entropy approaches zero for an ideal perfect crystal because there is only one possible perfectly ordered arrangement at absolute zero. With only one accessible microstate, the ideal entropy reference becomes zero.
Engineers and scientists can get extremely close to absolute zero, but they cannot reach exactly 0 K with a finite cooling process. Cooling becomes progressively harder as temperature decreases because there is less removable thermal energy and fewer available energy states.
The Third Law helps engineers understand low-temperature behavior, entropy references, cryogenic limits, superconducting systems, material properties, and refrigeration constraints. It is especially useful when analyzing systems where heat capacity, entropy, and molecular order change strongly with temperature.
Summary and Next Steps
The Third Law of Thermodynamics explains the limiting behavior of entropy as a perfect crystal approaches absolute zero. It gives entropy an absolute reference point and helps connect microscopic order, temperature, and low-temperature material behavior.
In engineering practice, the law is most useful when paired with real constraints: heat leaks, material imperfections, residual entropy, thermal contraction, property data limits, and cryogenic equipment performance. The key is to understand the ideal law first, then check whether the real system actually behaves like the ideal model.
Where to go next
Continue your learning path with related Turn2Engineering resources.
-
Laws of Thermodynamics
Review how the zeroth, first, second, and third laws work together as the foundation of thermodynamic analysis.
-
Entropy
Build a deeper understanding of the property that the Third Law uses to describe microscopic order and energy availability.
-
Refrigeration Cycles
Connect thermodynamic limits to real cooling systems, heat removal, and practical temperature control.