Key Takeaways
- Core idea: The Second Law explains why real processes move in a preferred direction and why total entropy cannot decrease in an isolated system.
- Engineering use: Engineers use it to judge heat engine limits, refrigeration performance, waste heat, entropy generation, and lost useful work.
- What controls it: Temperature differences, heat transfer, friction, pressure drop, mixing, turbulence, and finite-rate processes control irreversibility.
- Practical check: If an analysis predicts no waste heat, no losses, or 100% conversion of heat into work, the model is violating real Second Law behavior.
Table of Contents
Introduction
The Second Law of Thermodynamics states that natural processes move in the direction of increasing total entropy. In engineering, it explains why heat flows from hot to cold, why real energy systems produce waste heat, why engines cannot be perfectly efficient, and why every practical thermal process includes irreversibility.
How the Second Law of Thermodynamics Works
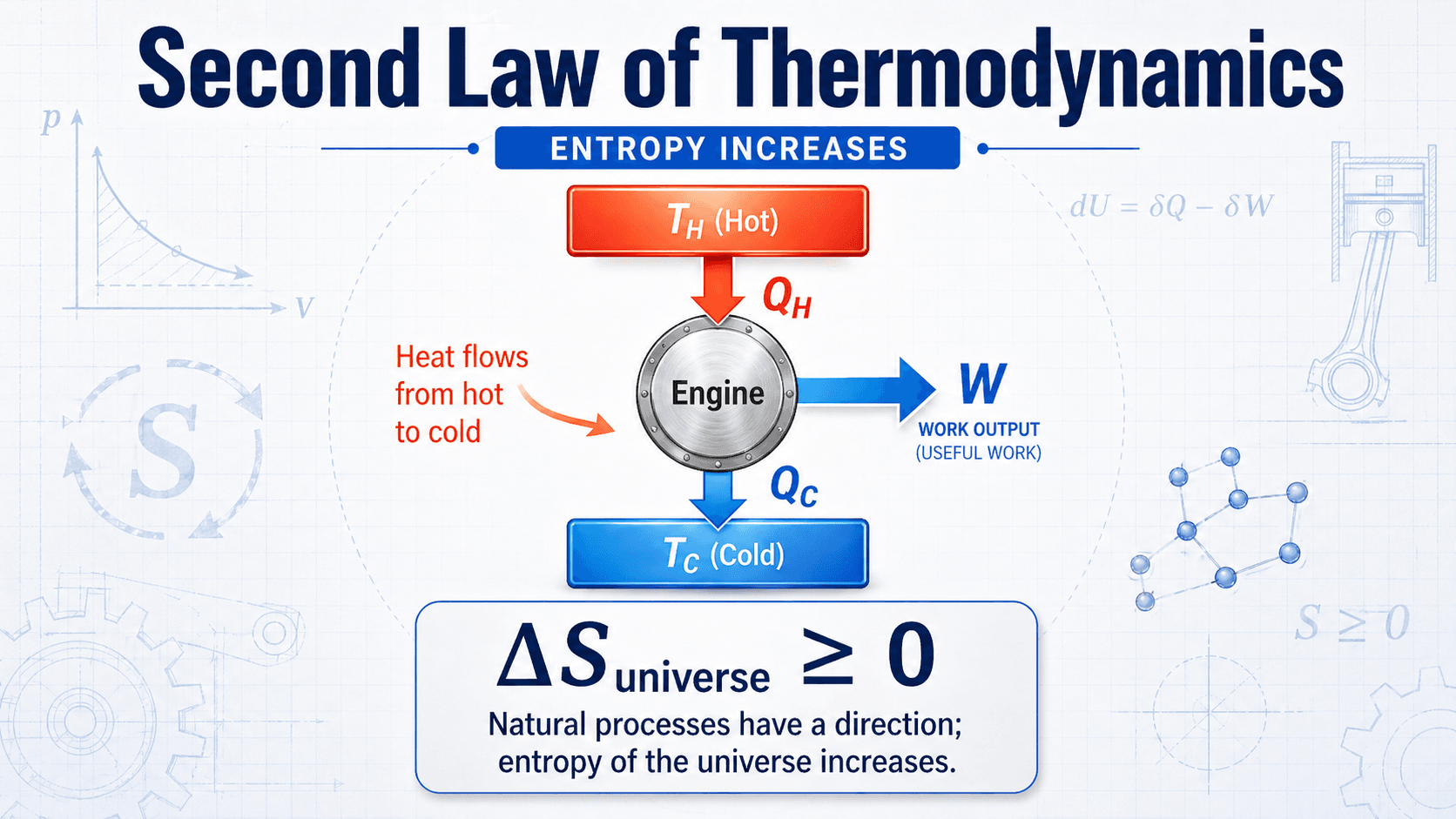
Notice the direction of heat flow and the split between useful work and rejected heat. That split is not just a design inconvenience; it is a thermodynamic limit that controls engines, turbines, refrigeration cycles, heat pumps, and power plants.
What is the Second Law of Thermodynamics?
The Second Law of Thermodynamics is the law that gives energy processes a direction. The First Law of Thermodynamics says energy is conserved, but the Second Law explains why not all energy is equally useful after a real process occurs.
A falling weight, a spinning shaft, a hot gas, and a fuel stream may all contain energy, but the quality of that energy is not the same. Once friction, heat transfer across a finite temperature difference, mixing, throttling, or turbulence occurs, some ability to produce useful work is destroyed. Engineers often describe that loss using entropy generation and exergy destruction.
The Second Law is not only about “disorder.” For engineering systems, it is mainly about direction, irreversibility, and the loss of useful work potential.
Entropy and Process Direction
Entropy is the thermodynamic property most closely tied to the Second Law. It helps describe how energy spreads out and how much of a process is irreversible. A system can experience a local entropy decrease, but the combined entropy change of the system and its surroundings cannot be negative for a real isolated process.
Entropy is not just disorder
Many introductory explanations describe entropy as disorder, but that shortcut can be misleading. For mechanical engineering, it is usually more useful to think of entropy as a measure connected to energy dispersal, unavailable energy, and the number of microscopic ways a state can exist.
Total entropy determines whether a process is possible
A real process must satisfy the entropy balance for the system and surroundings together. If the total entropy change is positive, the process is irreversible. If it is zero, the process is ideal and reversible. If it is negative, the proposed process cannot occur without some other compensating change.
Second Law Equations Engineers Use
The Second Law can be written in several useful forms. For a reversible heat transfer process at absolute temperature, entropy change is related to heat transfer by:
For a more general process, engineers often evaluate the total entropy change of the system plus surroundings:
- \( \Delta S \) Entropy change, usually expressed in J/K, kJ/K, J/(kg·K), or kJ/(kg·K).
- \( Q_{rev} \) Heat transfer for an equivalent reversible path, not simply any uncontrolled real heat transfer.
- \( T \) Absolute temperature in kelvin or degrees Rankine. Celsius and Fahrenheit are not valid for entropy ratios.
| Result | Meaning | Engineering interpretation |
|---|---|---|
| \( \Delta S_{total} = 0 \) | Reversible ideal process | Useful as a theoretical benchmark, not a real operating condition. |
| \( \Delta S_{total} > 0 \) | Irreversible real process | Expected for real turbines, compressors, valves, engines, and heat exchangers. |
| \( \Delta S_{total} < 0 \) | Impossible process | The model violates the Second Law unless another compensating process is included. |
Clausius and Kelvin-Planck Statements
The Second Law is often written in two classic forms. They may sound different, but both describe the same physical limitation: real energy conversion requires direction, temperature difference, and unavoidable loss.
Clausius statement
Heat does not spontaneously flow from a colder body to a hotter body. Refrigerators and heat pumps can move heat from cold to hot, but only by receiving work input from a compressor or another external source.
Kelvin-Planck statement
No cyclic heat engine can convert all heat taken from a single thermal reservoir into work. A real engine must reject some heat to a lower-temperature sink, which is why 100% thermal efficiency is not possible.
How Engineers Use the Second Law
Mechanical engineers use the Second Law to move beyond basic energy accounting. An energy balance can show that energy is conserved, but a Second Law check shows whether the process is realistic, efficient, and directionally possible.
- Heat engines: Used to evaluate thermal efficiency, rejected heat, and the gap between real and ideal cycle performance.
- Refrigeration and heat pumps: Used to understand coefficient of performance, compressor work, and why heat can be moved from cold to hot only with input work.
- Turbines and compressors: Used to estimate losses from non-isentropic behavior, pressure drop, leakage, and mechanical inefficiency.
- Heat exchangers: Used to understand why finite temperature differences allow heat transfer but also create irreversibility.
- Power plants: Used to identify waste heat, cooling system requirements, and opportunities for improved cycle efficiency.
A good Second Law review asks: where is entropy generated, where is useful work destroyed, and which losses are large enough to matter for design or operation?
What Controls Irreversibility?
Irreversibility is the reason real processes fall short of ideal behavior. It is not caused by one single factor. It usually comes from a combination of thermal, fluid, mechanical, and material effects that appear throughout a system.
| Factor | Why it matters | Engineering implication |
|---|---|---|
| Finite temperature difference | Heat transfer needs a temperature difference, but larger differences generate more entropy. | Heat exchangers trade compact size and high heat rate against Second Law efficiency. |
| Friction and pressure drop | Mechanical energy is dissipated into thermal energy that is less useful for work. | Piping, ducts, valves, bearings, and seals can reduce system performance. |
| Mixing | Mixing streams at different temperatures, pressures, or compositions destroys useful separation potential. | Important in combustion, HVAC mixing boxes, exhaust streams, and chemical processes. |
| Turbulence and throttling | Rapid flow changes and pressure reductions can be highly irreversible. | Valves and restrictions may be simple and reliable, but they sacrifice recoverable energy. |
| Real equipment behavior | Compressors, turbines, pumps, and engines do not follow ideal reversible paths. | Isentropic efficiency, mechanical efficiency, and heat losses must be considered. |
Second Law Design Review Checklist
Use this checklist when reviewing a thermodynamic system, cycle model, or homework solution. It helps separate a valid energy balance from a physically realistic engineering result.
Start with the energy balance, identify the system boundary, list heat and work interactions, then check whether the assumed process direction and entropy change are physically possible. If the result implies no losses, no rejected heat, or heat moving from cold to hot without work, revisit the assumptions.
| Check or decision | What to look for | Why it matters |
|---|---|---|
| Define the boundary | Closed system, open system, control volume, or complete isolated system. | Entropy may decrease locally, but the full system plus surroundings must satisfy the Second Law. |
| Track heat reservoirs | Hot source, cold sink, heat input, and rejected heat. | Heat engines need a sink; refrigeration systems need work input. |
| Check absolute temperature | Use K or °R in entropy and efficiency relations. | Using Celsius or Fahrenheit in temperature ratios creates invalid results. |
| Look for hidden irreversibilities | Friction, pressure drop, finite temperature difference, throttling, mixing, leakage, and turbulence. | These are often the real reasons equipment performs below ideal cycle predictions. |
| Compare ideal and real performance | Carnot limit, isentropic efficiency, COP, or measured operating data. | The Second Law is most useful when it shows how far the real system is from the ideal limit. |
Worked Example: Entropy Change from Heat Transfer
Suppose a system receives 600 J of heat through a reversible path at a constant absolute temperature of 300 K. The entropy change is:
Assumption
This calculation assumes a reversible heat transfer path at constant absolute temperature. That makes it useful for learning the relationship between heat, temperature, and entropy, but real equipment usually requires a broader entropy balance.
Engineering meaning
The same amount of heat produces a larger entropy change when transferred at a lower temperature. That is one reason low-temperature waste heat is harder to convert into useful work than high-temperature heat from combustion, steam, or concentrated solar sources.
Engineering Judgment and Field Reality
In real mechanical systems, the Second Law shows up as performance shortfall. A turbine produces less shaft work than an ideal expansion predicts. A compressor requires more input power than a reversible compression would require. A heat exchanger needs a finite temperature approach. A refrigeration system consumes more power as the temperature lift increases.
The biggest Second Law losses are often not in the component with the most obvious heat transfer. They may be hidden in pressure drop, throttling valves, poor controls, fouled heat exchanger surfaces, air leakage, excessive temperature lift, or off-design operation.
When This Breaks Down
Simplified Second Law explanations are valuable, but they can break down when readers treat ideal statements as direct equipment predictions. The law remains valid, but the simplified model may not include enough detail to describe the real system.
- Local system only: A local entropy decrease does not violate the Second Law if the surroundings experience a larger entropy increase.
- Ideal reversible paths: Reversible equations are reference models; real equipment has entropy generation.
- Single-temperature assumptions: Real heat transfer often occurs across varying temperatures, so an integral or property-table approach may be required.
- Neglected pressure losses: Pressure drop in pipes, ducts, filters, valves, and heat exchangers can dominate real performance.
- Steady operation assumptions: Startup, shutdown, thermal storage, transient loads, and control cycling can change the entropy balance.
Common Mistakes and Practical Checks
Most mistakes come from mixing up energy conservation with energy usefulness. The First Law can be satisfied while the design is still impossible, inefficient, or physically incomplete under the Second Law.
- Calling entropy only “disorder”: This misses the engineering link to useful work, irreversibility, and energy quality.
- Using Celsius in entropy ratios: Entropy and thermal efficiency relationships require absolute temperature.
- Ignoring the surroundings: Refrigerators, heat pumps, and living systems can reduce entropy locally only by increasing entropy elsewhere.
- Assuming all heat is equally useful: High-temperature heat has more work potential than low-temperature heat.
- Expecting 100% heat-to-work conversion: A cyclic engine must reject heat to a lower-temperature sink.
If a cycle analysis shows all heat input becoming work output, it is not just optimistic; it violates the Kelvin-Planck form of the Second Law.
Useful References and Engineering Context
The Second Law is a foundational concept rather than a single design code requirement. Engineers usually apply it through thermodynamics textbooks, property data, equipment standards, and discipline-specific design guidance.
- Engineering thermodynamics textbooks: Common references such as Moran and Shapiro or Çengel and Boles explain entropy, exergy, heat engines, and refrigeration cycles in a mechanical engineering format.
- ASHRAE guidance: HVAC and refrigeration design resources use Second Law concepts when discussing heat pumps, refrigeration cycles, coefficient of performance, and energy efficiency.
- Manufacturer performance data: Turbine, compressor, heat exchanger, pump, and chiller data help compare ideal thermodynamic limits with measured equipment behavior.
Frequently Asked Questions
The Second Law of Thermodynamics states that the total entropy of an isolated system cannot decrease. In practical terms, real processes have a preferred direction: heat flows from hot to cold, energy spreads out, and every real engine or refrigeration system has unavoidable losses.
A heat engine cannot convert all heat input into work because a cyclic device must reject some heat to a lower-temperature reservoir. The Second Law sets this limit, so real engines always have waste heat, friction, pressure losses, and other irreversibilities.
Entropy does not have to increase inside every local system, but the total entropy of an isolated system must stay the same for an ideal reversible process or increase for a real irreversible process. A refrigerator can lower entropy inside a cold space only by using work and rejecting more entropy to the surroundings.
Mechanical engineers use the Second Law to evaluate thermal efficiency, refrigeration performance, waste heat, entropy generation, and lost work. It helps explain why turbines, compressors, heat exchangers, engines, and HVAC systems perform worse than ideal models.
Summary and Next Steps
The Second Law of Thermodynamics explains why real processes have direction and why energy becomes less available to do useful work. It connects entropy, irreversibility, heat flow, waste heat, and the limits of energy conversion.
In engineering practice, the most useful Second Law habit is to look for where useful work is destroyed. Temperature differences, friction, pressure drop, throttling, mixing, and real equipment losses often explain why a system performs below its ideal model.
Where to go next
Continue your learning path with related Turn2Engineering resources.
-
Entropy
Learn the property most directly connected to irreversibility, unavailable energy, and process direction.
-
Heat Engines
See how the Second Law limits engine efficiency and requires rejected heat.
-
Reversible and Irreversible Processes
Compare ideal reversible models with the real processes that generate entropy.