Key Takeaways
- Core idea: A reversible process is an ideal limiting process with no entropy generation; an irreversible process is a real process that leaves a net effect on the system, surroundings, or both.
- Engineering use: Reversible processes define maximum work output, minimum work input, ideal cycle efficiency, and the benchmark used to judge real equipment.
- What controls it: Friction, turbulence, finite temperature differences, pressure losses, mixing, throttling, shocks, and electrical resistance create irreversibility.
- Practical check: Slow does not automatically mean reversible; a slow process with friction, viscosity, heat loss, or resistance still generates entropy.
Table of Contents
Introduction
Reversible and irreversible processes describe whether a thermodynamic change can be undone without leaving a net effect on the system and surroundings. A reversible process is an ideal, nearly equilibrium path with no entropy generation. An irreversible process is a real process with losses such as friction, turbulence, mixing, pressure drop, or heat transfer across a finite temperature difference.
Reversible vs Irreversible Process Diagram
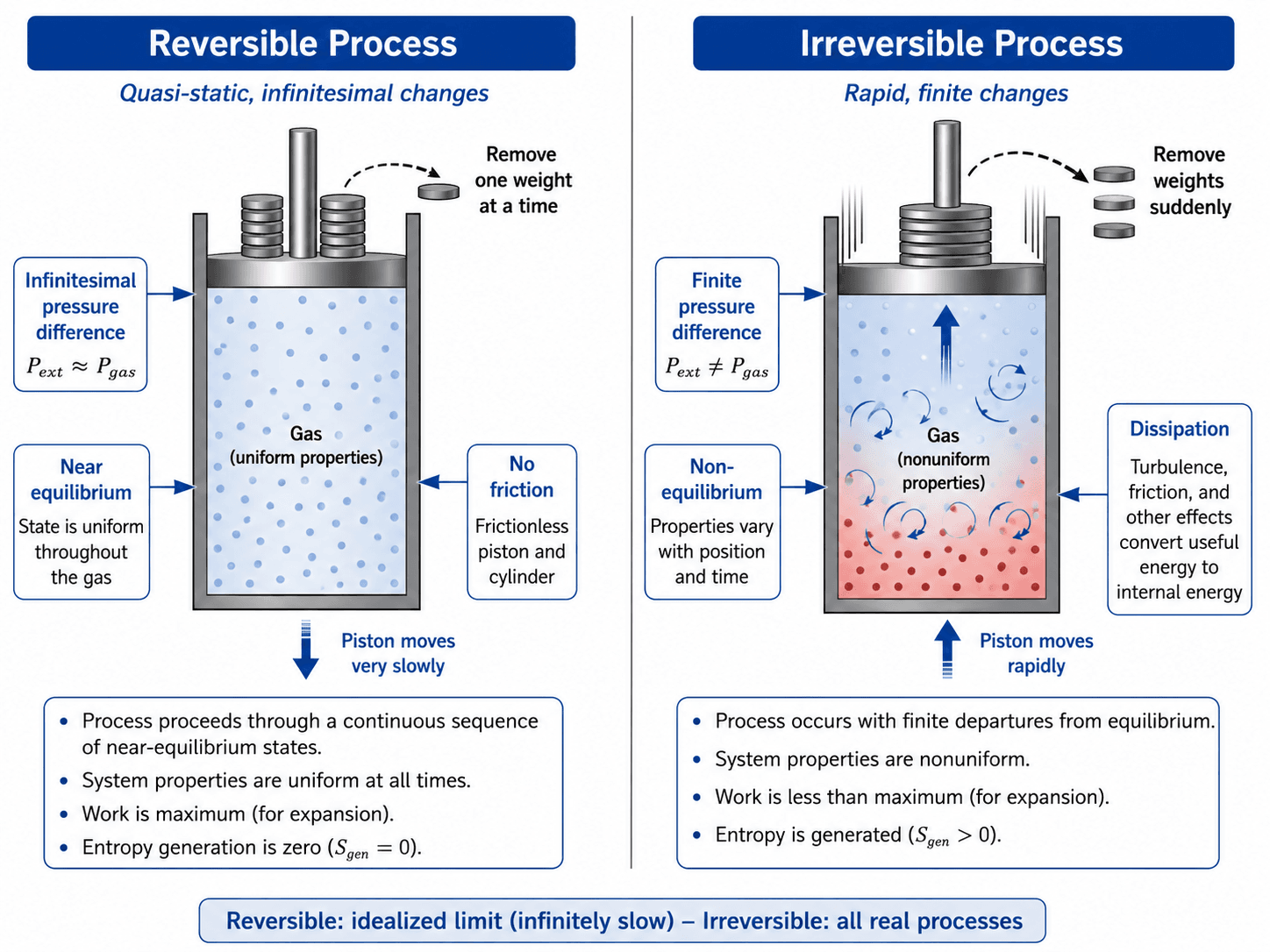
Notice the size of the driving force first. Reversibility requires infinitesimal pressure or temperature differences, while real processes need finite differences to occur at a practical rate.
What Are Reversible and Irreversible Processes?
A reversible process is a theoretical thermodynamic process that can be reversed by an infinitesimal change in the surroundings while returning both the system and surroundings to their original states. It must proceed through a continuous sequence of equilibrium states and avoid dissipative effects such as friction, viscosity, turbulence, electrical resistance, and uncontrolled heat transfer.
An irreversible process is any real process that cannot be exactly undone without leaving some residual effect. The system may be returned to its original state, but the surroundings will not also return to their original state without additional changes. That difference matters because it reveals where useful work potential was destroyed.
| Term | Simple meaning | Main thermodynamic test |
|---|---|---|
| Reversible process | Ideal process that can be undone with no net effect on the system and surroundings. | \(S_{\text{gen}} = 0\) |
| Irreversible process | Real process that cannot be undone without leaving a net effect. | \(S_{\text{gen}} > 0\) |
| Impossible process | Proposed process that would reduce total entropy of the system plus surroundings. | \(S_{\text{gen}} < 0\) |
Reversible processes are not used because engineers expect to build perfect machines. They are used because they define the best possible case. Real machines are judged by how far they fall below that ideal.
How Reversibility Works in Thermodynamics
Reversibility is controlled by how close the system stays to equilibrium while the process occurs. In a piston-cylinder example, a reversible expansion would require the external pressure to be only infinitesimally lower than the gas pressure. The piston moves extremely slowly, the gas properties remain nearly uniform, and the path can be retraced.
Reversible processes require equilibrium states
A reversible process must be quasi-static, meaning the system is close enough to equilibrium that pressure, temperature, and other properties are well defined throughout the process. That is why reversible paths can be drawn cleanly on thermodynamic property diagrams.
Irreversible processes require finite driving forces
Real processes occur because there is a finite pressure difference, temperature difference, voltage difference, concentration difference, or other driving force. That finite difference creates a practical rate of change, but it also creates irreversibility. The stronger the departure from equilibrium, the more likely the process will generate entropy.
Internal vs external reversibility
Internal reversibility means no irreversibilities occur inside the system boundary. External reversibility means no irreversibilities occur in the surroundings. A process is fully reversible only when both conditions are satisfied. For example, a process can be internally reversible but externally irreversible if heat transfer occurs across a finite temperature difference outside the chosen system boundary.
Quasi-static does not automatically mean reversible
A slow process can still be irreversible. For example, a piston may move slowly while sliding against a rough cylinder wall. The system may remain close to mechanical equilibrium, but friction converts organized mechanical work into internal energy and generates entropy.
Examples of Reversible and Irreversible Processes
Examples are often the easiest way to understand the difference. Ideal reversible examples are useful as benchmarks, while irreversible examples describe the real behavior found in engines, compressors, turbines, heat exchangers, refrigeration systems, valves, ducts, and fluid systems.
| Example | Classification | Why it is classified that way |
|---|---|---|
| Ideal frictionless isothermal expansion | Reversible idealization | The gas expands through infinitesimal pressure differences while temperature is held constant by reversible heat transfer. |
| Ideal reversible adiabatic expansion | Reversible idealization | No heat transfer occurs and no entropy is generated, so the process is also isentropic. |
| Carnot cycle step | Reversible idealization | The cycle uses reversible heat-transfer and adiabatic steps to define the ideal heat-engine efficiency limit. |
| Heat transfer across a finite temperature difference | Irreversible | Heat flows naturally from hot to cold, but the finite temperature difference generates entropy. |
| Free expansion of a gas | Irreversible | The gas expands without a controlled boundary pressure path and useful work opportunity is lost. |
| Throttling through a valve | Irreversible | Pressure drops without producing useful work, even though the process is often modeled as approximately isenthalpic. |
| Mixing of gases or fluids | Irreversible | Mixing and diffusion occur spontaneously and cannot be undone without additional work or separation processes. |
| Frictional sliding or viscous flow | Irreversible | Organized mechanical energy is dissipated into internal energy. |
Entropy Generation and the Second Law
Entropy generation is the clearest way to separate reversible, irreversible, and impossible processes. The First Law of Thermodynamics tells whether energy is conserved. The Second Law of Thermodynamics tells whether the process direction is possible and how much useful work potential is destroyed.
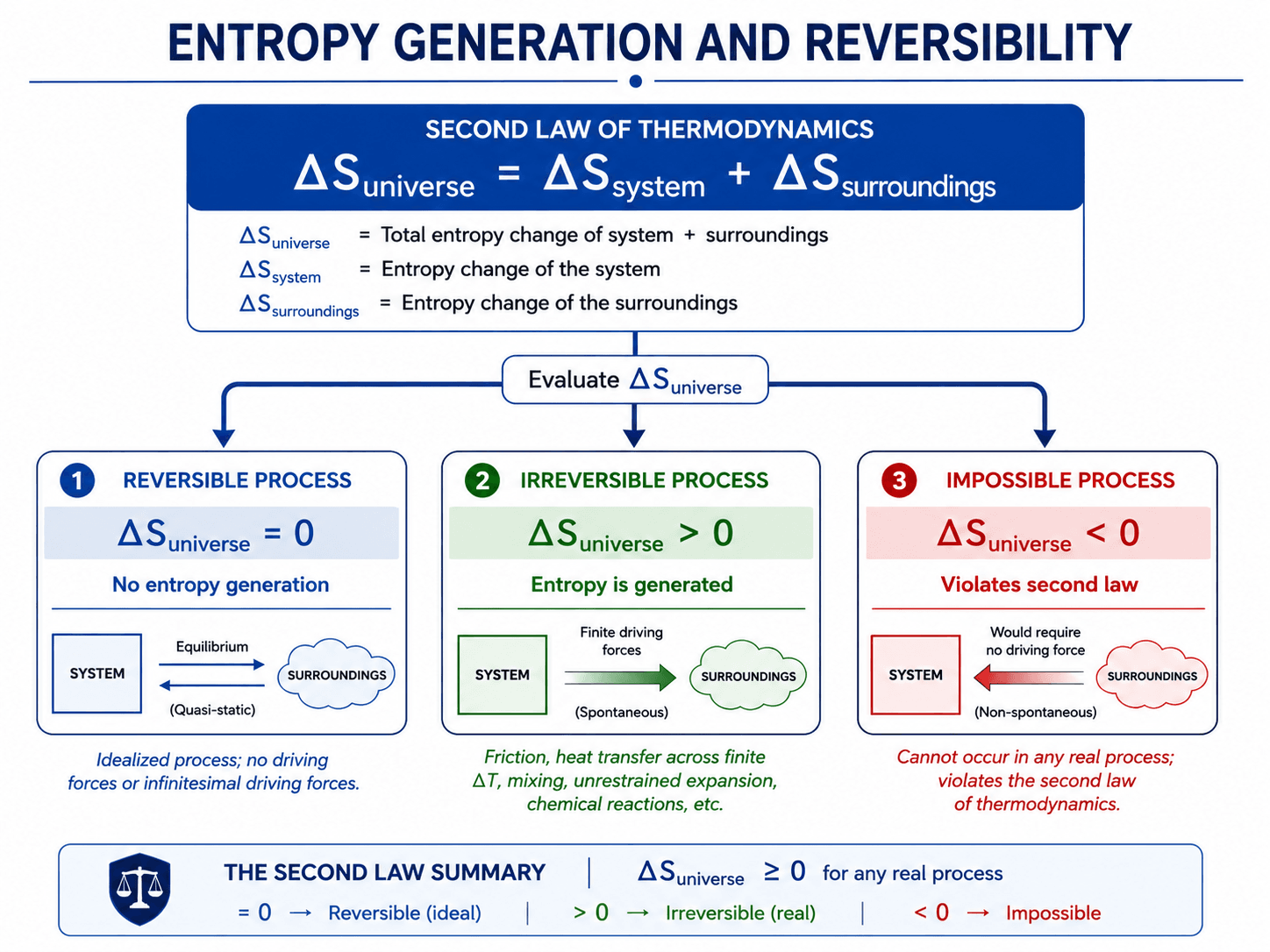
| Entropy result | Process type | Meaning |
|---|---|---|
| \(S_{\text{gen}} = 0\) | Reversible | Ideal limiting process with no entropy generation in the complete system plus surroundings. |
| \(S_{\text{gen}} > 0\) | Irreversible | Real possible process where entropy is generated and useful work potential is destroyed. |
| \(S_{\text{gen}} < 0\) | Impossible | Violates the Second Law unless the boundary definition is incomplete or another compensating effect is missing. |
The important nuance is that entropy of a local system can decrease. A refrigerator lowers entropy inside a cold compartment, for example. But the compressor work and heat rejection increase the entropy of the surroundings by more than the local decrease, so the combined process remains irreversible.
Worked Example: Entropy Generation from Heat Transfer
Heat transfer across a finite temperature difference is one of the most common irreversible processes. Consider 10 kJ of heat transferred from a hot reservoir at 600 K to a cold reservoir at 300 K.
Because the total entropy change is positive, the process is irreversible. The heat transfer is physically possible, but the finite temperature difference destroys useful work potential.
Smaller temperature differences reduce entropy generation, but they usually require larger heat exchangers, slower heat transfer, or higher equipment cost. That tradeoff is why real engineering design balances performance against practicality.
Reversible vs Irreversible Process Comparison
The difference is not just a vocabulary distinction. It changes the work prediction, heat-transfer interpretation, efficiency limit, and whether a path can be drawn on a property diagram.
| Feature | Reversible process | Irreversible process |
|---|---|---|
| Physical meaning | Ideal limiting process that can be retraced without a net effect. | Real process that leaves a net effect on the system, surroundings, or both. |
| Equilibrium behavior | Passes through near-equilibrium states. | May pass through non-equilibrium states that are not fully defined. |
| Driving force | Infinitesimal pressure, temperature, or potential difference. | Finite pressure, temperature, concentration, voltage, or velocity difference. |
| Entropy generation | Zero for the complete system plus surroundings. | Positive because real losses degrade useful work potential. |
| Expansion work | Maximum work output for the same initial and final states. | Less work output because some potential is destroyed by irreversibilities. |
| Compression work | Minimum work input for the same initial and final states. | More work input is required because friction, pressure loss, and heating add penalties. |
| Typical examples | Ideal frictionless isothermal expansion, reversible adiabatic process, Carnot cycle step. | Free expansion, throttling, mixing, combustion, friction, turbulence, finite-temperature heat transfer. |
How Reversible and Irreversible Processes Appear on a P-V Diagram
A pressure-volume diagram is useful only when the intermediate states are meaningful. A reversible or quasi-static process can be shown as a smooth path because the system passes through a sequence of equilibrium states. A strongly irreversible process may only have a defined initial and final state, not a fully defined path between them.
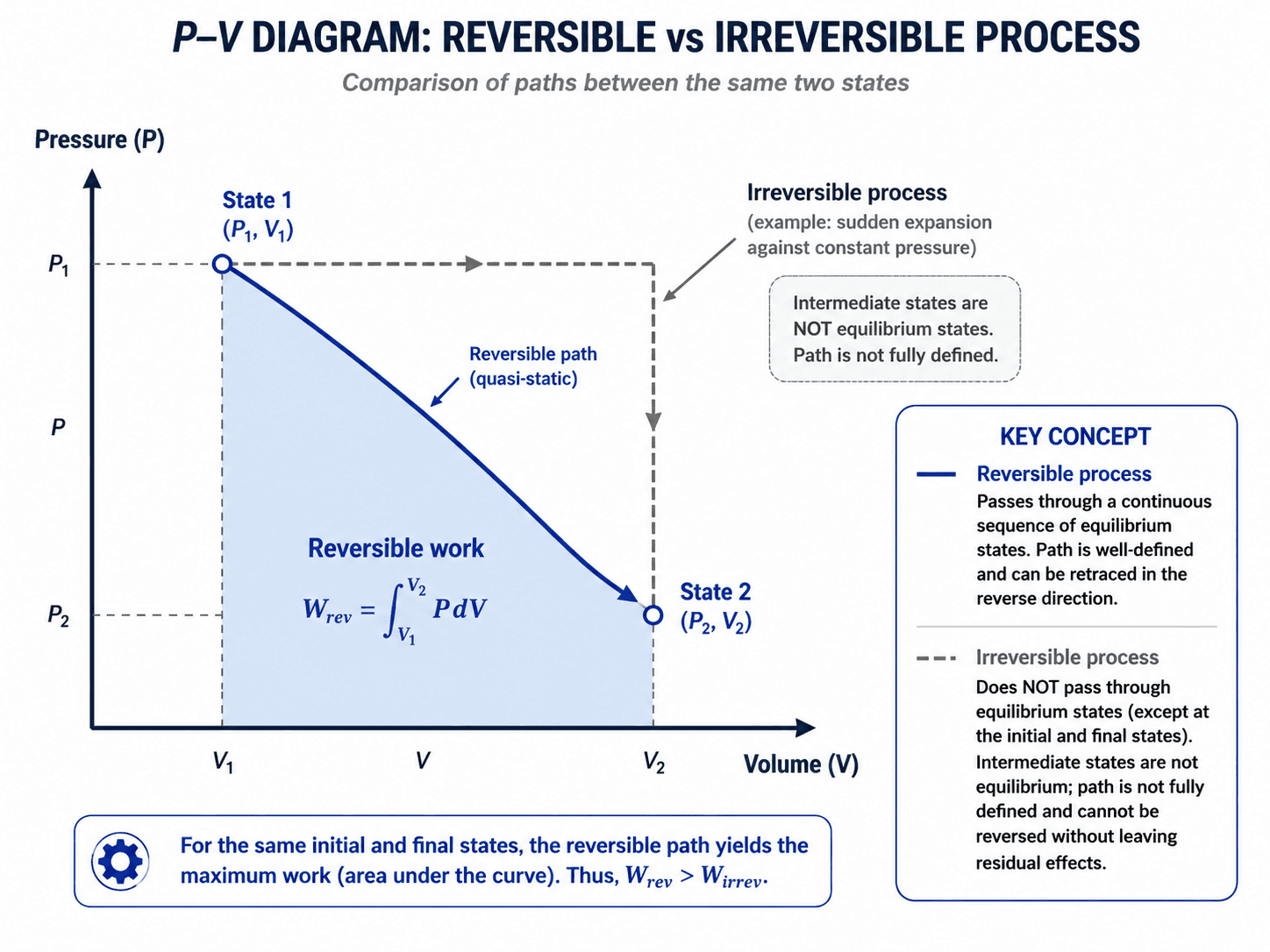
Work is the area under the boundary pressure versus volume path. For a reversible piston-cylinder process, the system pressure and boundary pressure are essentially the same at each step. For a rapid irreversible process, the internal gas pressure may not be uniform, and the boundary pressure may not describe a clean equilibrium path.
For an ideal reversible expansion between two states, this integral gives the maximum boundary work. In free expansion, volume changes but boundary work can be zero because the gas expands into a vacuum or uncontrolled space instead of pushing against a useful resisting pressure.
Why Engineers Care About Reversible and Irreversible Processes
Reversibility is one of the main tools engineers use to separate an ideal performance limit from real equipment behavior. It is central to thermal efficiency, compressor work, turbine work, refrigeration coefficient of performance, heat-exchanger losses, and second-law analysis.
- Turbines: Irreversible expansion reduces shaft work compared with the ideal reversible or isentropic case.
- Compressors and pumps: Irreversible compression increases required input work because of friction, heating, leakage, and pressure loss.
- Heat exchangers: Finite approach temperature makes heat transfer practical but generates entropy.
- Throttle valves: Pressure reduction is simple and inexpensive, but available work is destroyed instead of recovered.
- Nozzles and diffusers: Friction, shocks, and flow separation reduce useful kinetic energy conversion or pressure recovery.
- Refrigeration cycles: Compressor inefficiency, throttling, heat-exchanger approach temperature, and pressure drops reduce coefficient of performance.
When a thermodynamic calculation looks too good, ask where entropy is being generated. Real equipment almost always loses performance through finite heat-transfer temperature differences, pressure drop, mechanical friction, throttling, mixing, or nonideal compression and expansion.
Connection to Lost Work and Exergy
Entropy generation is not just an abstract property change. In engineering analysis, it is directly connected to lost work, also called exergy destruction. The more entropy a real process generates, the more useful work potential it destroys.
- \(W_{\text{lost}}\) Useful work potential destroyed by irreversibility, commonly expressed in kJ, Btu, kW, or hp depending on whether the analysis is energy or power based.
- \(T_0\) Dead-state or environment temperature in absolute units such as K or °R.
- \(S_{\text{gen}}\) Entropy generated by the real process because of friction, finite gradients, mixing, throttling, heat transfer, or other irreversibilities.
This is why reducing entropy generation improves second-law performance. A larger heat exchanger, smoother flow path, better compressor, lower pressure drop, or smaller temperature difference may reduce lost work, but those improvements must be weighed against cost, space, complexity, and operating requirements.
What Causes Irreversibility?
Irreversibility appears when a process occurs with finite gradients or dissipative effects. These are not small bookkeeping details. In real systems, they often explain why measured performance is far below a clean textbook model.
| Cause of irreversibility | What happens physically | Engineering implication |
|---|---|---|
| Friction | Organized mechanical energy is converted into internal energy. | Increases work input for compressors and reduces work output from turbines and engines. |
| Finite temperature difference | Heat flows from hot to cold at a practical rate. | Necessary in real heat exchangers, but larger temperature differences generate more entropy. |
| Turbulence and viscosity | Fluid motion dissipates mechanical energy into heat. | Creates pressure loss in ducts, pipes, nozzles, heat exchangers, and turbomachinery. |
| Free expansion | A gas expands into a vacuum or low-pressure region without controlled boundary work. | Useful work opportunity is lost even if the gas reaches the same final equilibrium state. |
| Mixing and diffusion | Species, energy, or momentum spread from nonuniform to more uniform states. | Important in combustion, HVAC air mixing, chemical processing, and thermal systems. |
| Throttling | Pressure drops through a valve or restriction without useful work output. | Common in refrigeration and steam systems; often modeled as isenthalpic but irreversible. |
| Electrical resistance | Electrical work is dissipated as heat. | Important in heaters, motors, power electronics, battery systems, and electrical losses. |
| Shock waves | Flow properties change abruptly with strong entropy generation. | Critical in high-speed nozzles, compressors, diffusers, turbines, and aerospace applications. |
Equations Engineers Use to Check Reversibility
Reversibility is usually checked through entropy, not by simply asking whether a process can be run backward. The practical question is whether the complete system and surroundings can be restored without net entropy generation.
This equation defines entropy change using a reversible heat-transfer path. It does not mean the actual process must be reversible to calculate a state-property change. Because entropy is a property, engineers often evaluate entropy change between two states using a convenient reversible path even when the real process is irreversible.
- \(S_{\text{gen}}\) Entropy generated by irreversibilities. It is zero for a reversible ideal process and positive for a real irreversible process.
- \(T\) Absolute temperature in kelvin or rankine. Entropy calculations require absolute temperature.
- \(\delta Q_{\text{rev}}\) Differential heat transfer along a reversible path used to evaluate entropy change.
How to Identify Irreversibility
Use this practical check when reviewing a thermodynamics problem, cycle model, or equipment assumption. The goal is not to prove a real process is perfectly reversible. The goal is to identify where the ideal model departs from real behavior.
Start with the initial and final states. Ask whether the path stays near equilibrium. Identify finite gradients, friction, pressure loss, heat-transfer temperature differences, mixing, throttling, or rapid motion. Then decide whether the reversible model is being used as a true process assumption, a benchmark, or only a convenient path for calculating a property change.
| Check or decision | What to look for | Why it matters |
|---|---|---|
| Can the path be drawn as equilibrium states? | Uniform pressure, temperature, and properties at each step. | If intermediate states are not equilibrium states, a normal property-path diagram may be misleading. |
| Are driving forces infinitesimal or finite? | Large pressure, temperature, voltage, concentration, or velocity differences. | Finite driving forces allow practical rates but generate irreversibility. |
| Are dissipative effects present? | Friction, viscosity, turbulence, electrical resistance, pressure drop, or plastic deformation. | Any dissipative effect prevents the process from being perfectly reversible. |
| Is there uncontrolled expansion, throttling, or mixing? | Valve drops, free expansion, fluid mixing, diffusion, or rapid filling/emptying. | These processes are common in real systems and are strongly irreversible. |
| Is the model being used for a benchmark? | Words such as ideal, isentropic, Carnot, maximum work, or minimum work. | Ideal reversible models are often valid as comparison limits, not as physical descriptions of real equipment. |
| Does the entropy balance make sense? | \(S_{\text{gen}} = 0\) for ideal reversible cases and \(S_{\text{gen}} > 0\) for real cases. | A negative total entropy generation means the model, sign convention, or boundary definition is wrong. |
Example: Free Expansion vs Controlled Reversible Expansion
Consider a gas expanding from the same initial state to the same final volume. In a controlled reversible expansion, the gas pushes against a piston while the external pressure is adjusted in infinitesimal steps. In free expansion, a valve opens and the gas expands into an evacuated space without producing useful boundary work.
Same final state does not mean same process
The gas may eventually settle into a final equilibrium state in both cases, but the path and work transfer are different. The reversible expansion can produce maximum work because the boundary force is controlled throughout the motion. Free expansion produces no useful boundary work against the vacuum and is strongly irreversible.
Engineering meaning
This is the same logic behind many real losses. A pressure drop across a valve, rapid flow through a restriction, or uncontrolled expansion may be easy to build, but it destroys useful work potential that a more idealized expansion device could theoretically recover.
Engineering Judgment and Field Reality
In real equipment, irreversibility is rarely caused by only one thing. A compressor may have mechanical friction, heat transfer with the casing, leakage, pressure losses, and nonuniform flow entering the impeller. A heat exchanger may have finite temperature differences, pressure drop, fouling, maldistribution, and heat loss to ambient surroundings.
This is why experienced engineers treat reversible models as clean baselines. The model tells you what the device could do in the best thermodynamic case. Field data, component efficiencies, pressure measurements, temperature approach values, and power measurements reveal how far the actual system falls below that ideal.
A real process can be improved but not made perfectly reversible. The practical goal is to reduce the largest sources of entropy generation enough to improve performance, reliability, or operating cost without making the system impractical or uneconomical.
When This Breaks Down
The reversible-versus-irreversible distinction is powerful, but simplified diagrams can become misleading if the assumptions are forgotten. The biggest issue is treating ideal paths as if they describe fast, real, spatially nonuniform processes.
- Rapid transients: Startup, shutdown, valve opening, shock waves, and fast expansion may not pass through well-defined equilibrium states.
- Large gradients: Large temperature or pressure differences make the process occur faster, but they also increase irreversibility.
- Real component behavior: Pumps, turbines, compressors, nozzles, ducts, and valves require efficiency factors, pressure-loss models, or measured performance data.
- Path confusion: Heat and work depend on the path, while properties such as internal energy and entropy depend on state. Mixing these ideas causes many thermodynamics errors.
- Natural processes: In practice, all natural processes are irreversible because real processes require finite gradients or dissipative effects to occur.
Common Mistakes and Practical Checks
Most confusion comes from using everyday language instead of strict thermodynamic definitions. A process is not reversible just because a machine can be forced to run in the opposite direction.
| Common mistake | Correct interpretation | Practical check |
|---|---|---|
| Reversible means simply running backward. | The system and surroundings must both return to original states with no net effect. | Ask what changed in the surroundings after the process was reversed. |
| Slow means reversible. | Slow plus friction, viscosity, resistance, or heat loss is still irreversible. | Look for dissipative effects, not just process speed. |
| Entropy of the system must always increase. | System entropy can decrease, but total entropy of the system plus surroundings cannot decrease. | Define the full system boundary before judging entropy generation. |
| Irreversible means energy is destroyed. | Energy is conserved, but useful work potential is destroyed. | Use lost work or exergy destruction to understand the performance penalty. |
| Free expansion has a normal P-V path. | Intermediate states are not equilibrium states, so the path is not fully defined. | Do not apply a smooth equilibrium curve to a strongly non-equilibrium process. |
Do not label a process reversible only because it is idealized or slow. A reversible process must be quasi-static and free of dissipative effects.
Useful References and Engineering Context
Reversible and irreversible processes are foundational thermodynamics concepts rather than project-specific code requirements. Engineers usually apply them through thermodynamics textbooks, cycle analysis, component efficiency models, and second-law reasoning.
- OpenStax University Physics: OpenStax reversible and irreversible processes reference explains the ideal reversible limit, real irreversible behavior, and the connection between process direction and thermodynamic reasoning.
- Project-specific criteria: Equipment datasheets, owner performance requirements, operating conditions, and measured field data usually control the final engineering evaluation of a real thermodynamic system.
- Engineering use: Use the reversible case as the upper-bound comparison, then account for component efficiencies, pressure losses, heat-transfer approach temperatures, throttling, leakage, and other real sources of entropy generation.
Frequently Asked Questions
A reversible process is an ideal process that can be reversed without leaving any net effect on the system or surroundings. An irreversible process is a real process with finite driving forces, friction, turbulence, mixing, pressure losses, heat transfer across finite temperature differences, or other effects that generate entropy.
Perfectly reversible processes are ideal limits, not real operating processes. Engineers still use them because they define maximum work output, minimum work input, ideal cycle efficiency, and the benchmark used to compare real turbines, compressors, engines, refrigeration systems, and heat exchangers.
No. A reversible process must be quasi-static, but a quasi-static process is not automatically reversible. A process can be very slow and still irreversible if friction, viscosity, electrical resistance, turbulence, pressure loss, or other dissipative effects are present.
Entropy generation is the thermodynamic signature of irreversibility. For an ideal reversible process, total entropy generation is zero. For a real irreversible process, entropy is generated, meaning useful work potential is destroyed even though total energy is still conserved.
A reversible expansion produces maximum work because the process occurs through infinitesimal pressure differences, so the gas can push against the highest possible external pressure at each step. In a real irreversible expansion, pressure losses, turbulence, free expansion, and friction reduce the useful boundary work that can be recovered.
Summary and Next Steps
Reversible and irreversible processes explain the difference between an ideal thermodynamic limit and real process behavior. A reversible process stays near equilibrium and generates no entropy. An irreversible process includes real effects such as friction, turbulence, mixing, throttling, pressure loss, electrical resistance, shocks, and finite-temperature heat transfer.
The most useful engineering habit is to identify where entropy is generated. Those locations usually explain why real equipment produces less work, requires more input power, transfers heat less ideally, or performs below a clean reversible model.
Where to go next
Continue your learning path with related Turn2Engineering resources.
-
Entropy
Learn the property most directly tied to irreversibility, process direction, and useful work potential.
-
Second Law of Thermodynamics
Review the law that connects entropy generation, process direction, and the limits of energy conversion.
-
Carnot Cycle
See how reversible processes define the ideal efficiency limit for heat engines.