Key Takeaways
- Core idea: Drinking water treatment uses multiple physical, chemical, and disinfection barriers to convert source water into potable water.
- Engineering use: Engineers arrange unit processes into a treatment train so each step improves the reliability and performance of the next step.
- What controls it: Source water quality, turbidity, organic matter, pathogens, pH, alkalinity, temperature, target contaminants, and distribution residual drive treatment decisions.
- Practical check: Clear water is not automatically safe water; filtration performance, disinfectant contact time, residual maintenance, and distribution stability all matter.
Table of Contents
Introduction
Drinking water treatment is the engineered process of removing particles, microorganisms, organic matter, dissolved chemicals, taste and odor compounds, and other water quality concerns before water is delivered to the public. A reliable system does not depend on one process; it uses multiple treatment barriers from source water intake through storage and distribution.
Drinking Water Treatment Process Diagram
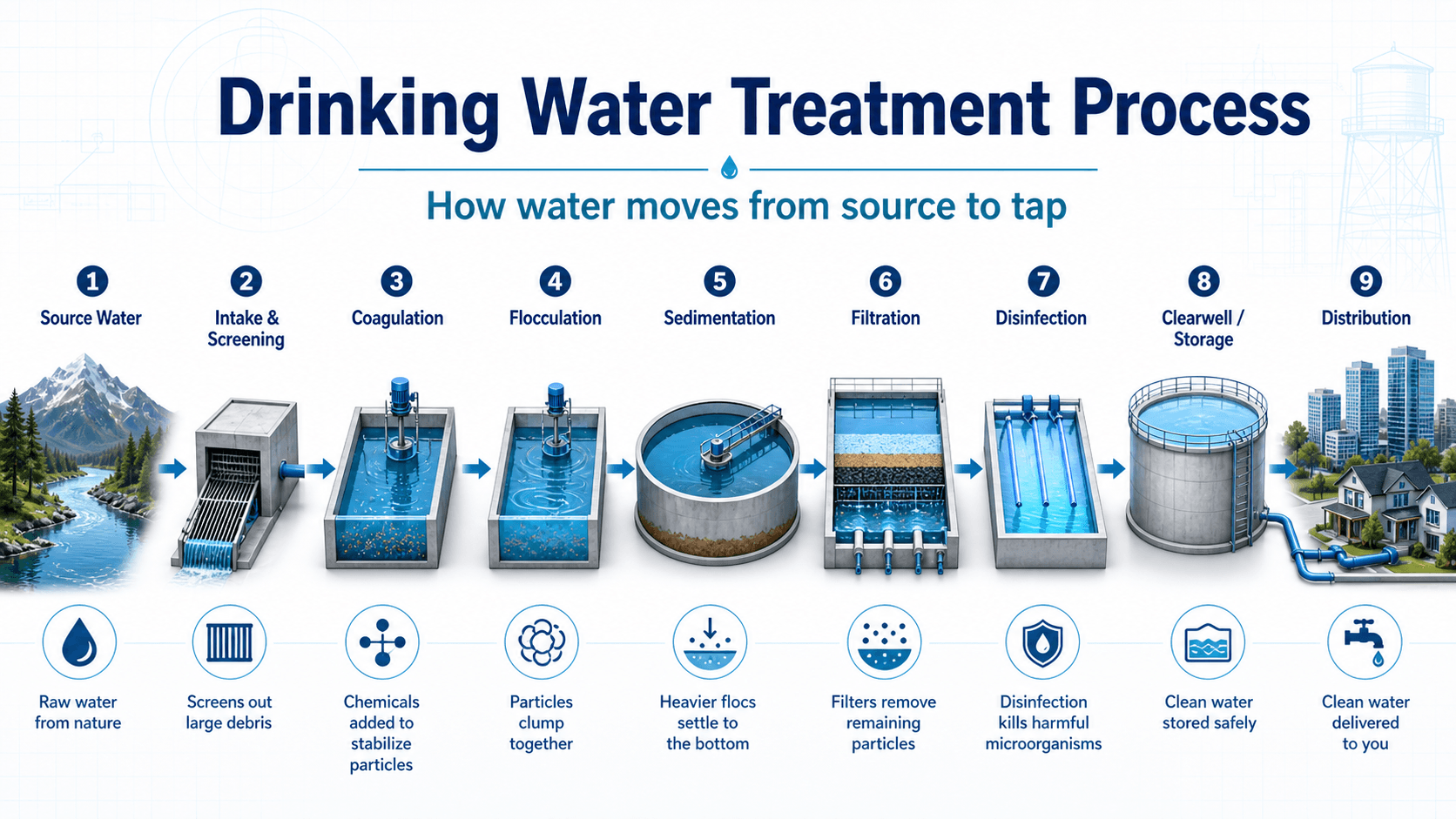
Notice the order of the steps. Screening protects equipment, coagulation and flocculation make small particles removable, sedimentation and filtration reduce particle load, and disinfection is most reliable after turbidity and organic demand have been controlled.
What is Drinking Water Treatment?
Drinking water treatment is the process of converting raw source water into potable water that can be delivered through a public or private water system. In water resources engineering, the goal is not simply to make water appear clean. The treatment system must reduce health risk, meet finished water quality targets, protect downstream storage and pipes, and remain stable as source water conditions change.
The important engineering concept is the treatment train. A treatment train is a sequence of unit processes selected so that each barrier supports the next one. Coagulation and flocculation prepare fine suspended particles for settling, sedimentation removes much of that solids load, filtration polishes the water, and disinfection inactivates microorganisms after the water has already been clarified.
A shallow explanation often treats drinking water treatment as a fixed list of steps. In practice, engineers choose and adjust those steps based on source water testing, seasonal variability, target contaminants, reliability requirements, available operators, residuals handling, finished water chemistry, and distribution system behavior.
Municipal Drinking Water Treatment vs Home Water Treatment
Most engineering discussions of drinking water treatment focus on municipal or utility-scale treatment. These systems treat water before it enters storage tanks, pressure zones, water mains, and customer service connections. They are designed around public health protection, continuous operation, monitoring, operator response, and distribution system reliability.
Home water treatment systems are different. Carbon filters, water softeners, ultraviolet units, reverse osmosis units, distillers, and point-of-entry systems are selected for specific household concerns after water has already reached a building. A home filter may improve taste, reduce selected contaminants, or provide added protection, but it does not replace source water protection, public treatment, distribution residuals, and system-level monitoring.
The right home treatment device depends on actual water testing. A carbon filter, softener, UV unit, and reverse osmosis system solve different problems, so selecting a device based only on taste or appearance can miss the real water quality issue.
How the Drinking Water Treatment Train Works
The drinking water treatment train works by matching contaminant type to a removal or control mechanism. Large debris is screened, fine particles are chemically destabilized, floc is settled, remaining particles are filtered, and pathogens are inactivated by disinfection. Advanced processes may be added when conventional treatment does not address a specific contaminant.
Source water and intake
Source water may come from a river, lake, reservoir, aquifer, or blended supply. Intake structures move raw water into the plant while screens remove large material such as leaves, sticks, algae mats, trash, and debris that could damage pumps or clog downstream equipment.
Coagulation and flocculation
Coagulation uses chemicals such as aluminum or iron salts to destabilize fine particles and colloids. Flocculation then applies gentle mixing so destabilized particles collide and form larger flocs. This step is highly sensitive to pH, alkalinity, temperature, mixing energy, chemical dose, and raw water organic matter.
Sedimentation and filtration
Sedimentation removes heavier flocs by gravity in basins or clarifiers. Filtration then removes smaller particles that remain after settling. Filtration can be an important microbial barrier, especially for particle-associated organisms and protozoa, but its effectiveness depends on filter design, media condition, loading rate, upstream coagulation, and operation.
Disinfection, storage, and distribution
Disinfection inactivates pathogens and helps maintain microbiological safety. Chlorine and chloramine are commonly used when a residual is needed in the distribution system, while ultraviolet light and ozone can provide strong treatment at the plant but do not provide the same continuing residual in pipes. Finished water is then stored and distributed under controlled pressure.
What Each Treatment Step Removes
Each treatment barrier is strongest against certain water quality problems. A plant that performs well usually does not rely on a single process to remove everything. Instead, it layers barriers so particle removal, microbial control, taste and odor control, and chemical treatment work together.
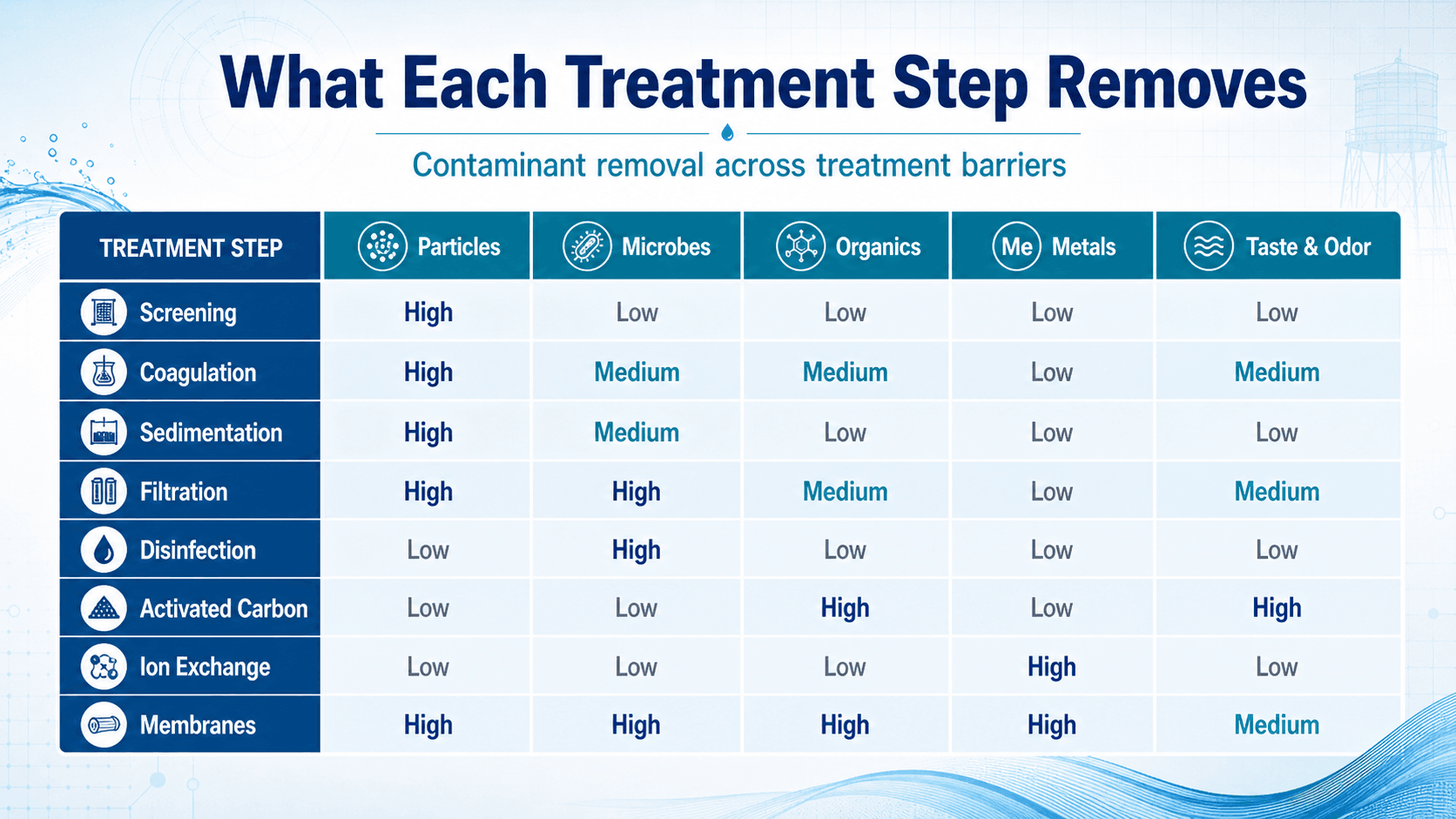
The “high, medium, low” concept is a practical way to read the treatment barriers, not a design guarantee. Actual removal depends on source water chemistry, treatment design, operating conditions, media condition, chemical dose, contact time, and monitoring results.
| Treatment barrier | Primary purpose | Engineering implication |
|---|---|---|
| Screening | Remove large debris and protect equipment. | Improves reliability but does not address fine particles, dissolved contaminants, or pathogens. |
| Coagulation and flocculation | Convert fine particles and colloids into larger flocs. | Requires dose control, proper pH, adequate mixing, and operator response to changing raw water. |
| Sedimentation | Remove heavier flocs by gravity. | Reduces solids loading on filters but can suffer from short-circuiting, density currents, and poor sludge removal. |
| Filtration | Remove remaining particles and improve clarity. | Filter performance depends on media condition, loading rate, backwash control, and upstream clarification. |
| Disinfection | Inactivate bacteria, viruses, and other microorganisms. | Performance depends on disinfectant type, concentration, contact time, pH, temperature, turbidity, and demand. |
| Advanced treatment | Target dissolved or difficult contaminants. | Activated carbon, membranes, ion exchange, softening, or oxidation may be needed for specific source water problems. |
Common Drinking Water Contaminants and Treatment Methods
Drinking water treatment is selected around the contaminants found in the source water. Some concerns are particle-related, some are microbial, and others are dissolved chemicals that require targeted treatment. This is why water testing and treatment goals come before process selection.
| Water quality concern | Why it matters | Common treatment approach |
|---|---|---|
| Turbidity and suspended solids | Can carry particles, reduce clarity, clog filters, and interfere with disinfection. | Coagulation, flocculation, sedimentation, and filtration. |
| Bacteria and viruses | Can create acute public health risk if not controlled. | Filtration, chlorine, chloramine, ozone, ultraviolet disinfection, and residual maintenance. |
| Giardia and Cryptosporidium | Protozoa can be more resistant to some disinfectants than bacteria. | Effective filtration, ultraviolet disinfection, ozone, and validated treatment barriers. |
| Natural organic matter | Can cause color, taste, odor, disinfectant demand, and disinfection byproduct precursors. | Enhanced coagulation, activated carbon, oxidation, and process optimization. |
| Iron and manganese | Can cause staining, deposits, metallic taste, and customer complaints. | Aeration, oxidation, filtration, and pH control. |
| Hardness | Can cause scale and affect plumbing, appliances, industrial uses, and customer acceptance. | Lime softening, ion exchange, or membranes. |
| Nitrate | A common groundwater concern with health-based limits. | Ion exchange, reverse osmosis, or biological treatment where appropriate. |
| Arsenic | A naturally occurring groundwater contaminant in some regions. | Oxidation, adsorption media, coagulation/filtration, or membranes. |
| PFAS | Persistent trace contaminants that require specialized treatment and careful waste handling. | Granular activated carbon, ion exchange resins, or high-pressure membranes. |
| Lead and copper | Often related to corrosion of premise plumbing or distribution materials after treatment. | Corrosion control through pH, alkalinity, orthophosphate, and distribution monitoring. |
Many contaminants are not removed equally by a single technology. Ion exchange and adsorption can target selected dissolved ions, but effectiveness depends on contaminant species, competing ions, pH, media type, empty bed contact time, regeneration or replacement schedule, and waste handling.
Surface Water vs Groundwater Treatment
Drinking water treatment is not identical for every source. Surface water is exposed to runoff, algae, sediment, organic matter, and microbial contamination, so it often requires a full conventional treatment train. Groundwater is naturally filtered by soil and geologic layers, but it may contain dissolved minerals or gases that require different treatment.

Some groundwater sources are influenced by surface water and may need treatment closer to a surface water plant. The correct classification depends on hydrogeology, water quality data, microbial risk, seasonal changes, and regulatory review.
| Source type | Common water quality concerns | Common treatment response |
|---|---|---|
| Surface water | Turbidity, algae, pathogens, color, taste and odor, natural organic matter, storm-event variability. | Screening, coagulation, flocculation, sedimentation, filtration, disinfection, and sometimes activated carbon or oxidation. |
| Groundwater | Hardness, iron, manganese, nitrate, arsenic, hydrogen sulfide, dissolved gases, radionuclides, corrosivity. | Aeration, oxidation, filtration, softening, ion exchange, adsorption, membranes, disinfection, and corrosion control as needed. |
| Blended supply | Variable chemistry, disinfectant demand, pH and alkalinity changes, seasonal source switching. | Process control must account for changing raw water chemistry and distribution system stability. |
Key Factors That Control Treatment Performance
Drinking water treatment performance depends on both raw water quality and process control. The same plant can perform well during normal conditions and struggle during storms, algae blooms, temperature changes, droughts, chemical feed problems, filter aging, or distribution system water age issues.
| Factor | Why it matters | Engineering implication |
|---|---|---|
| Turbidity | High turbidity increases particle loading and can shield microorganisms from disinfection. | Coagulation, settling, filtration, and filter backwash frequency may need adjustment during storms or runoff events. |
| Natural organic matter | Organic matter affects color, taste, odor, coagulant demand, and disinfectant byproduct formation. | Engineers may adjust coagulation, add activated carbon, change disinfectant strategy, or improve precursor removal. |
| pH and alkalinity | pH influences coagulation chemistry, corrosion control, disinfectant effectiveness, and metal solubility. | Chemical dosing must consider buffering capacity and finished water stability, not just immediate clarity. |
| Temperature | Cold water slows floc formation and can reduce reaction rates. | Plants may need longer flocculation, adjusted dose, or tighter filter monitoring during cold periods. |
| Disinfectant demand | Particles, organics, ammonia, iron, manganese, and biofilms consume disinfectant. | Operators must maintain contact time and residual without overfeeding chemicals or increasing byproduct risk. |
| Distribution water age | Water quality can change after treatment as water sits in tanks or dead-end mains. | Storage turnover, residual monitoring, flushing, pressure management, and pipe material control become part of treatment reliability. |
A treatment process should be reviewed as a full system. Good filter effluent quality is important, but finished water also needs adequate disinfectant contact, stable chemistry, controlled residuals, and distribution conditions that preserve water quality to the customer.
How Engineers Select Drinking Water Treatment Technologies
Treatment selection starts with water quality data, not with a preferred technology. Engineers identify the contaminants or performance issues, determine which treatment barriers are needed, then evaluate reliability, operator needs, residuals, waste streams, cost, redundancy, and distribution impacts.
| Problem found in source water | Typical treatment options | Design concern |
|---|---|---|
| High turbidity | Coagulation, flocculation, sedimentation, filtration. | Dose control, mixing energy, basin hydraulics, filter loading, and sludge removal. |
| Pathogens | Filtration, chlorine, chloramine, ozone, ultraviolet disinfection. | Contact time, residual, pH, temperature, turbidity, and validation of treatment barriers. |
| Taste and odor | Activated carbon, oxidation, source management, algae control. | Carbon life, oxidant dose, byproduct formation, and timing of seasonal events. |
| Hardness | Lime softening, ion exchange, reverse osmosis. | Scale control, sludge or brine handling, finished water stability, and customer expectations. |
| Iron and manganese | Aeration, oxidation, greensand or media filtration, pH adjustment. | Oxidant dose, contact time, filter fouling, backwash frequency, and aesthetic complaints. |
| Nitrate | Ion exchange, reverse osmosis, biological treatment. | Waste stream management, monitoring, competing ions, and blending feasibility. |
| PFAS | Granular activated carbon, ion exchange resins, high-pressure membranes. | Media life, breakthrough monitoring, compound mix, background chemistry, concentrate or spent media disposal. |
| Corrosion and lead/copper risk | pH adjustment, alkalinity control, orthophosphate, distribution monitoring. | Finished water stability, pipe scales, premise plumbing, residual chemistry, and customer sampling. |
The best technology is not always the most advanced one. A treatment method must be reliable under expected conditions, maintainable by the operating staff, compatible with the existing plant, and practical for residuals and waste handling.
Process Control Methods Engineers Watch Closely
Drinking water treatment plants use monitoring and operational targets to keep performance stable. The exact values depend on plant design and regulatory requirements, but the engineering logic is consistent: measure the variables that indicate whether each barrier is working before the next barrier is overloaded.
Jar testing and chemical dose control
Jar testing helps operators estimate coagulant dose under changing raw water conditions. It is especially useful when turbidity, color, alkalinity, pH, or organic matter changes. The goal is not the lowest chemical dose; the goal is a stable floc that settles and filters well without causing downstream problems.
Filter performance and backwash timing
Filters are monitored for turbidity, headloss, run time, and breakthrough behavior. A filter that runs too long may release particles, while a filter that is backwashed too aggressively or returned to service too quickly can produce poor initial effluent quality.
Disinfectant contact time
Disinfection is affected by disinfectant concentration, effective contact time, temperature, pH, and water quality. A simplified way to think about contact time is:
- C Disinfectant concentration, commonly evaluated as a residual concentration in mg/L.
- T Effective contact time, often evaluated in minutes after accounting for basin hydraulics and short-circuiting.
- CT The concentration-time exposure used to evaluate disinfection performance for a target organism and operating condition.
The simplified equation is useful conceptually, but engineers do not treat all basins as perfect plug-flow reactors. Baffles, mixing, short-circuiting, dead zones, and changing water levels affect the contact time actually achieved.
Drinking Water Treatment Review Workflow
A practical review starts with the water source, then follows the treatment train in order. This prevents the common mistake of focusing only on a single unit process while ignoring the upstream condition that caused the problem.
Start with source water quality → confirm the target contaminants → verify the required treatment barriers → check chemical dose and mixing → review settling and filter performance → confirm disinfection contact and residual → evaluate storage and distribution conditions.
| Check or decision | What to look for | Why it matters |
|---|---|---|
| Confirm source water type | Surface water, groundwater, blended supply, seasonal switching, storm influence, or algae risk. | The source determines whether the plant needs conventional clarification, groundwater-specific treatment, or advanced barriers. |
| Match contaminants to barriers | Particles, microbes, organics, metals, hardness, nitrate, taste and odor, PFAS, or corrosion concerns. | No single treatment step removes every contaminant well, so the treatment train must address the actual risk profile. |
| Review coagulation performance | Raw water turbidity, pH, alkalinity, floc quality, settled water turbidity, and dose response. | Poor coagulation often causes problems later in sedimentation, filtration, disinfection, and byproduct control. |
| Check filter stability | Filter effluent turbidity, headloss rate, run length, backwash effectiveness, and breakthrough trends. | Filters are a critical final particle barrier before disinfection and finished water storage. |
| Verify disinfection conditions | Disinfectant residual, contact time, pH, temperature, demand, and basin hydraulics. | Disinfection must be evaluated under actual operating conditions, not just theoretical chemical feed rate. |
| Evaluate distribution stability | Tank turnover, residual decay, pressure, water age, flushing needs, pipe materials, and customer complaints. | Finished water can degrade after leaving the plant if storage and distribution are not controlled. |
Conventional vs Advanced Drinking Water Treatment
Conventional treatment is built around clarification, filtration, and disinfection. It is effective for many surface water supplies, especially when the main issues are turbidity, suspended solids, color, natural organic matter, and microbial risk. Advanced treatment is added when the water contains contaminants that are not adequately controlled by conventional barriers.
| Treatment approach | Best suited for | Important limitation |
|---|---|---|
| Conventional treatment | Surface water with particles, turbidity, color, organic matter, and microbial exposure. | May not adequately remove dissolved salts, nitrate, PFAS, some metals, or specific taste and odor compounds. |
| Activated carbon | Taste and odor, some organic compounds, algal byproducts, and selected trace contaminants. | Requires media management and replacement or regeneration as adsorption capacity is exhausted. |
| Ion exchange | Hardness, nitrate, selected metals, and selected dissolved ions. | Effectiveness depends on competing ions, resin selection, regeneration needs, and waste stream handling. |
| Membranes | High particle removal, microbial barriers, desalination, and specialized contaminant control. | Higher energy, fouling risk, concentrate disposal, and pretreatment requirements can control feasibility. |
| Oxidation or advanced oxidation | Taste and odor, iron, manganese, sulfide, algal toxins, and some trace organics. | Oxidant selection must consider byproducts, dose control, and downstream filtration requirements. |
The best treatment train is rarely the most complicated one. Engineers usually prefer the simplest reliable system that meets finished water goals, can be operated consistently, and can handle the worst reasonable source water conditions.
Finished Water and Distribution System Protection
Drinking water treatment does not end at the last basin or filter. Finished water must remain stable as it moves through clearwells, storage tanks, pumps, pressure zones, transmission mains, distribution mains, service lines, and building plumbing. Distribution system protection is one reason residual disinfectants, pressure management, corrosion control, and tank turnover matter.
| Distribution issue | What can happen | Practical control |
|---|---|---|
| Low disinfectant residual | Microbial risk can increase as residual decays with time, temperature, demand, and pipe conditions. | Maintain residual targets, manage water age, monitor dead ends, and adjust storage turnover. |
| Excessive water age | Taste, odor, residual decay, nitrification risk, and customer complaints can increase. | Improve tank cycling, reduce stagnant zones, flush strategically, and review system hydraulics. |
| Corrosion instability | Pipe scales and plumbing materials may release metals such as lead or copper. | Control pH, alkalinity, corrosion inhibitors, disinfectant chemistry, and distribution monitoring. |
| Pressure loss or intrusion risk | Contaminated water can enter through leaks, cross-connections, or low-pressure events. | Maintain positive pressure, manage valves and pumps, detect leaks, and prevent cross-connections. |
Drinking Water Treatment vs Wastewater Treatment
Drinking water treatment prepares source water for human use. Wastewater treatment removes pollutants from used water before discharge, reuse, or further processing. Some process names overlap, such as filtration, disinfection, sedimentation, and chemical treatment, but the design objectives are different.
A drinking water plant is judged by finished water quality, microbial protection, contaminant limits, residual stability, and distribution system performance. A wastewater plant is judged by effluent quality, solids handling, nutrient removal, organic load reduction, receiving water protection, and permit limits.
Engineering Judgment and Field Reality
Drinking water treatment diagrams are clean, but real plants deal with changing source water, imperfect hydraulics, maintenance constraints, sensor drift, chemical delivery delays, filter aging, operator staffing, residuals handling, and distribution system feedback. The most useful engineering review asks whether the whole system remains robust when conditions shift away from the normal day.
Many treatment issues show up downstream from their root cause. A filter turbidity problem may be a coagulation problem. A disinfection issue may be caused by high organic demand. A customer taste complaint may trace back to algae, tank turnover, residual decay, or corrosion control rather than the final treatment step alone.
Engineers also consider operability. A process that looks excellent on paper can be a poor fit if it requires constant chemical adjustment, produces residuals that the plant cannot handle, depends on specialized maintenance, or creates failure modes that operators cannot detect quickly.
When This Breaks Down
A simplified drinking water treatment sequence breaks down when it assumes source water is stable, hydraulics are ideal, and each barrier performs independently. Real treatment systems are interconnected, so a weakness early in the train can reduce the safety margin of later barriers.
- Storm-driven turbidity spikes: High runoff can increase suspended solids and organic demand faster than normal chemical dose settings can handle.
- Algae and taste-and-odor events: Algae can affect pH, filterability, toxins, odor compounds, and disinfectant demand.
- Cold water conditions: Lower temperature can slow floc formation and make clarification more difficult.
- Short-circuiting in basins: Water may move through sedimentation or contact basins faster than the nominal detention time suggests.
- Distribution system changes: Water age, low pressure, storage turnover, pipe scales, and residual decay can change finished water quality after treatment.
Common Mistakes and Practical Checks
The most common mistakes come from treating drinking water treatment as a simple checklist rather than a connected system. Each step must be evaluated based on what enters it, what it removes, what it passes downstream, and how it affects the next process.
- Assuming clear water is safe water: Low visible turbidity does not prove microbial safety or chemical compliance.
- Ignoring source water variability: A treatment train that performs well in dry weather may need different operation during storms, algae blooms, or seasonal turnover.
- Overlooking pH and alkalinity: These control coagulation, corrosion, disinfectant effectiveness, and finished water stability.
- Evaluating disinfection without upstream performance: Poor particle removal and high organic demand can reduce disinfection reliability.
- Forgetting distribution: Storage tanks, dead ends, pressure zones, pipe materials, and water age can affect what customers actually receive.
Do not design or explain a drinking water treatment plant as if every source needs the same process train. The correct treatment approach depends on tested source water quality, target contaminants, seasonal variation, operator capability, and the reliability required for public distribution.
Useful References and Design Context
Drinking water treatment is shaped by public health goals, treatment technique requirements, monitoring, operator practice, source water testing, and site-specific design criteria. A good engineering reference should help connect contaminants to practical treatment technologies.
- EPA drinking water technology guidance: EPA drinking water technologies guidance provides technology-focused context for evaluating drinking water treatment methods and contaminant removal approaches.
- Project-specific criteria: Final treatment requirements depend on source water testing, applicable drinking water regulations, state primacy agency requirements, owner standards, plant permits, and distribution system conditions.
- Engineering use: Engineers use references like this to screen treatment alternatives, evaluate contaminant-specific methods, plan pilot testing, and compare reliability, residuals, waste handling, and operational complexity.
Frequently Asked Questions
The main steps in conventional drinking water treatment are intake and screening, coagulation, flocculation, sedimentation, filtration, disinfection, storage, and distribution. Some plants also use advanced treatment such as activated carbon, membranes, ion exchange, oxidation, or softening depending on source water quality and target contaminants.
Groundwater is not always treated the same way as surface water. Surface water usually has more turbidity, algae, organic matter, and microbial exposure, so it often needs clarification and filtration. Groundwater may need treatment for hardness, iron, manganese, nitrate, arsenic, dissolved gases, or corrosion control.
Municipal drinking water treatment is designed to treat water for an entire public system before storage and distribution. Home treatment systems, such as carbon filters, softeners, UV units, or reverse osmosis units, are point-of-use or point-of-entry devices selected for specific household water quality concerns.
No. Treatment trains vary based on source water quality, target contaminants, regulations, plant size, operator capacity, and distribution system needs. Surface water plants often use clarification and filtration, while groundwater systems may focus on dissolved minerals, hardness, iron, manganese, nitrate, arsenic, or corrosion control.
Drinking water treatment problems often occur when source water quality changes faster than the plant can respond, chemical dose is not adjusted, filters are overloaded, sedimentation is short-circuited, disinfectant demand is underestimated, or distribution system conditions reduce residual protection before water reaches customers.
Summary and Next Steps
Drinking water treatment is the engineered sequence of barriers that turns raw source water into finished water suitable for public use. The core treatment train often includes intake, screening, coagulation, flocculation, sedimentation, filtration, disinfection, storage, and distribution.
The key practical lesson is that drinking water treatment is a system, not a single process. Source water quality, contaminant type, chemical dose, settling, filter performance, disinfection contact time, residual maintenance, corrosion control, and distribution conditions all affect the quality of water that reaches the tap.
Where to go next
Continue your learning path with related Turn2Engineering resources.
-
Water Treatment Processes
Learn how treatment processes are arranged into broader treatment trains for drinking water, wastewater, reuse, and industrial applications.
-
Flocculation & Coagulation
Deepen your understanding of the particle destabilization and floc formation steps that support clarification and filtration.
-
Water Supply Chain
See how source water, treatment, storage, distribution, and end users connect in a complete water supply system.