Key Takeaways
- Core idea: Reverse osmosis uses pressure to force water through a semi-permeable membrane while rejecting many dissolved salts and contaminants.
- Engineering use: RO is used for desalination, brackish groundwater treatment, high-purity process water, and advanced wastewater reuse.
- What controls it: Feedwater quality, osmotic pressure, membrane type, flux, recovery, pretreatment, cleaning strategy, and concentrate management control RO performance.
- Practical check: RO is not just a membrane choice; the full treatment train must manage fouling, scaling, pressure, finished-water stability, and brine disposal.
Table of Contents
Introduction
Reverse osmosis is a pressure-driven water treatment process that forces water through a semi-permeable membrane, producing clean permeate while leaving dissolved salts and contaminants in a concentrate or brine stream. In water resources engineering, RO is used when conventional clarification and filtration cannot meet finished-water goals for desalination, brackish groundwater, industrial water, or advanced reuse.
Reverse Osmosis System Process Flow
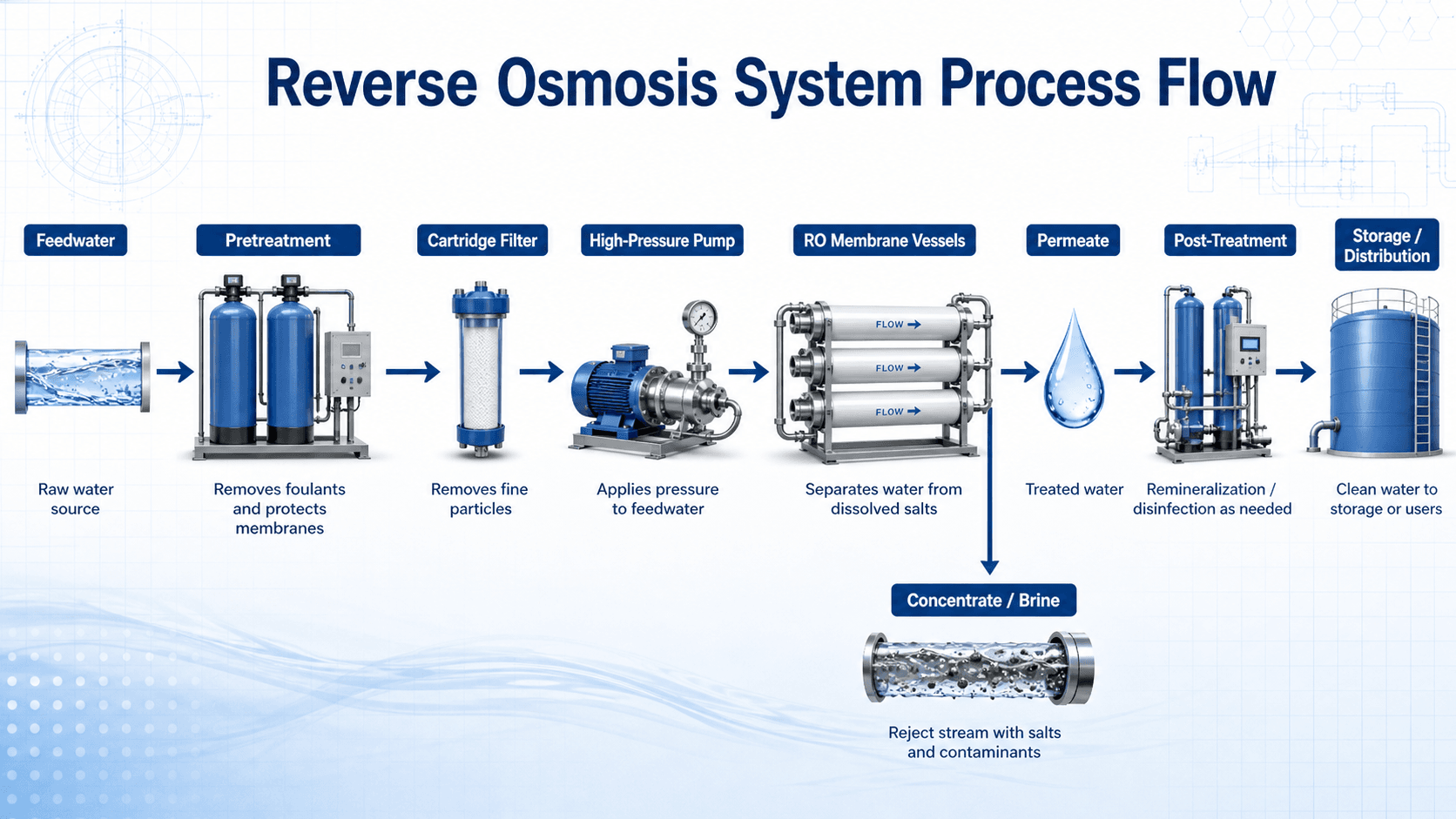
Notice that RO is not a single standalone filter. The membrane is protected by pretreatment, driven by pressure, and followed by both permeate handling and concentrate management.
What is Reverse Osmosis?
Reverse osmosis, often abbreviated as RO, is a membrane separation process that uses hydraulic pressure to move water through a semi-permeable membrane. The membrane allows water molecules to pass more readily than dissolved ions, larger molecules, microorganisms, and many trace contaminants.
Although RO is often casually called a filter, it is more accurately a membrane separation process. The membrane does not simply strain visible particles; it separates water from dissolved ions and small contaminants based on membrane chemistry, applied pressure, diffusion, and rejection behavior.
The important engineering distinction is that RO targets dissolved contaminants, not just visible particles. A conventional sand filter can remove suspended solids, but it will not remove sodium chloride from brackish groundwater. RO can reduce dissolved salts because the membrane separation mechanism is much tighter than ordinary filtration.
In a treatment plant, reverse osmosis is usually part of a larger treatment train. Pretreatment protects the membrane, the high-pressure pump provides the driving force, the membrane array separates the flow into permeate and concentrate, and post-treatment adjusts the finished water before storage, distribution, reuse, or industrial service.
How Reverse Osmosis Works
RO works by applying pressure greater than the natural osmotic pressure of the feedwater. That pressure forces water across the membrane from the concentrated feed side toward the lower-solute permeate side. Salts and other rejected constituents remain in the concentrate stream.
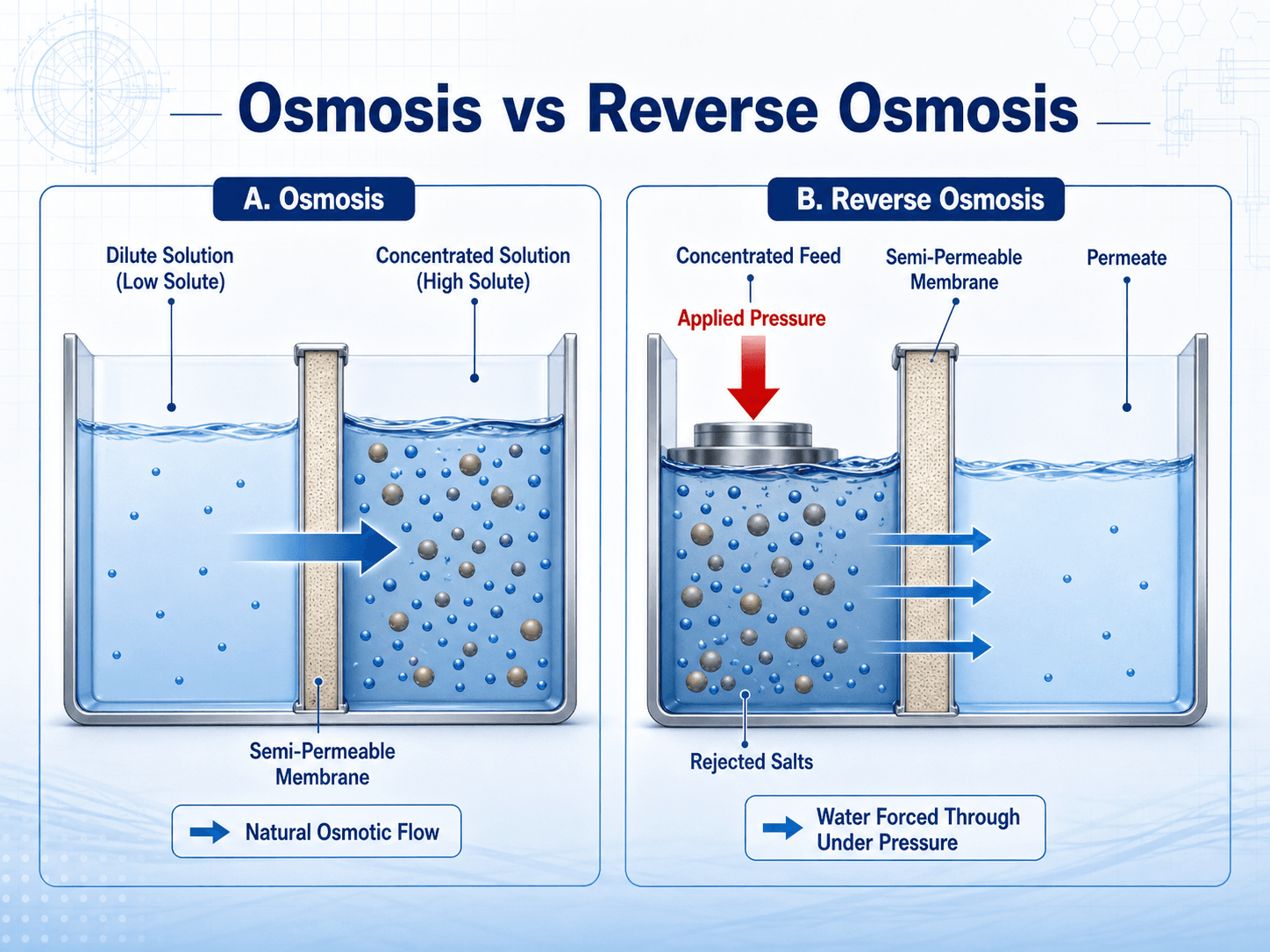
Step-by-step RO process
- Feedwater is pretreated to reduce particles, oxidants, hardness, organics, metals, and biological fouling potential.
- A high-pressure pump raises feed pressure above the osmotic pressure of the water.
- Water passes through the semi-permeable membrane and becomes permeate.
- Rejected salts and contaminants remain in the concentrate stream.
- Permeate is stabilized, blended, or disinfected before storage, distribution, reuse, or industrial service.
Feed, permeate, and concentrate
Feedwater is the raw or pretreated water entering the RO system. Permeate is the treated water that passes through the membrane. Concentrate, also called reject or brine, is the stream that carries salts and rejected contaminants away from the membrane array.
Pressure and osmotic resistance
Higher salinity increases osmotic pressure, which means the system needs more pressure and energy to produce permeate. This is why seawater RO operates at much higher pressure than brackish groundwater RO. The membrane does not magically remove salt; it requires a pressure gradient strong enough to overcome the natural tendency of water to move toward the more concentrated solution.
Membrane rejection
Rejection describes how much of a contaminant is kept out of the permeate. Actual rejection depends on membrane type, contaminant chemistry, membrane condition, temperature, pH, pressure, recovery, and how well pretreatment controls fouling and scaling.
Reverse Osmosis in Water Treatment Plants
In water treatment plants, reverse osmosis is usually an advanced treatment step rather than the first process in the plant. Raw water may need screening, clarification, filtration, chemical conditioning, or membrane pretreatment before it reaches the RO array.
A conventional-to-advanced treatment train may include upstream processes such as flocculation and coagulation, sedimentation, water filtration, cartridge filtration, reverse osmosis, remineralization, pH adjustment, disinfection, and storage. The exact order depends on the source water and finished-water target.
| Treatment train location | Purpose | RO design implication |
|---|---|---|
| Before RO | Remove particles, organics, oxidants, scalants, and biological growth potential. | Pretreatment controls membrane fouling, scaling, pressure drop, and cleaning frequency. |
| At the RO membrane | Separate feedwater into low-solute permeate and higher-solute concentrate. | Pressure, recovery, flux, membrane selection, and array configuration control performance. |
| After RO | Stabilize permeate for storage, distribution, reuse, or industrial process needs. | Low-mineral permeate may require pH adjustment, alkalinity addition, remineralization, blending, or disinfection. |
| Concentrate handling | Manage the brine stream containing rejected salts and contaminants. | Concentrate disposal, treatment, blending, or reuse can control project feasibility. |
RO should be evaluated as part of the full water treatment process, not as an isolated unit. The upstream processes protect the membrane, and downstream processes make the permeate suitable for its final use.
Main Components of a Reverse Osmosis System
A practical RO system is more than a membrane cartridge. It is a controlled process that prepares the feedwater, applies pressure, manages crossflow, monitors water quality, and handles both permeate and concentrate.
| Component | Purpose | Design implication |
|---|---|---|
| Intake or feed source | Supplies raw water to the treatment train. | Source variability controls pretreatment needs, monitoring, and operating flexibility. |
| Pretreatment | Reduces foulants, scalants, oxidants, and particles. | Poor pretreatment usually shows up as rising pressure drop, lower permeate flow, or frequent cleaning. |
| Cartridge filters | Protect membrane elements from fine particles. | Filter loading can indicate upstream solids-control problems. |
| High-pressure pump | Provides the driving pressure for permeate production. | Energy use, pressure range, and pump control strongly affect operating cost. |
| Membrane vessels | Hold RO membrane elements in pressure-rated housings. | Array configuration affects recovery, flux balance, pressure drop, and cleanability. |
| Instrumentation | Tracks flow, pressure, conductivity, pH, temperature, and differential pressure. | Monitoring is needed to detect fouling, scaling, membrane damage, and performance drift. |
| Post-treatment | Stabilizes permeate for final use. | RO permeate may need remineralization, pH adjustment, corrosion control, disinfection, or blending. |
| Concentrate handling | Manages the rejected brine stream. | Disposal or reuse of concentrate can control project feasibility. |
What Reverse Osmosis Removes
Reverse osmosis can remove or strongly reduce many dissolved and microbial contaminants, but removal is not identical for every constituent. Performance depends on the membrane, feedwater chemistry, operating pressure, recovery, temperature, pretreatment, and system age.

| Contaminant group | RO relevance | Engineering note |
|---|---|---|
| Total dissolved solids | Common RO target | High TDS increases osmotic pressure, required pumping pressure, and concentrate management difficulty. |
| Hardness minerals | Often reduced | Calcium and magnesium can also cause scaling if recovery is too high or antiscalant control is poor. |
| Nitrate, fluoride, arsenic, and metals | Often important drinking water targets | Removal should be verified for the specific contaminant, membrane, pH, and regulatory or project goal. |
| Bacteria and viruses | Can be physically rejected by intact membranes | Public water systems still need validated barriers, integrity monitoring, disinfection strategy, and distribution protection. |
| PFAS compounds | Many can be reduced | Performance depends on compound chemistry, membrane condition, recovery, and system configuration. |
| Volatile compounds and dissolved gases | Not always well controlled | Some constituents may pass through or require separate treatment such as aeration, adsorption, oxidation, or pH adjustment. |
What RO does not solve by itself
RO does not destroy contaminants; it separates them into permeate and concentrate streams. It also does not eliminate the need for pretreatment, post-treatment, monitoring, cleaning, or concentrate disposal.
- Volatile organic compounds and dissolved gases may require separate treatment.
- RO permeate may need pH adjustment, remineralization, blending, corrosion control, or disinfection.
- Membrane fouling and scaling are process-control problems, not problems solved by the membrane alone.
- Distribution system contamination can occur after RO if storage and distribution are not protected.
- PFAS and trace-contaminant treatment should be validated against project-specific water quality data and regulatory targets.
Reverse Osmosis vs Other Membrane Processes
Reverse osmosis is one of several membrane processes used in water treatment. The main difference is membrane tightness and the type of material being removed. RO generally provides the strongest dissolved salt removal, but it also requires higher pressure and more careful concentrate management.
| Process | Typical removal focus | Main use | Key limitation |
|---|---|---|---|
| Microfiltration | Suspended solids, protozoa, and some bacteria | Particle removal and pretreatment | Does not remove dissolved salts. |
| Ultrafiltration | Fine particles, bacteria, and some viruses | Surface water treatment, reuse pretreatment, and RO pretreatment | Limited dissolved ion removal. |
| Nanofiltration | Hardness, color, larger organics, and some dissolved ions | Softening, color removal, and selective ion reduction | Less complete salt removal than RO. |
| Reverse osmosis | Dissolved salts, TDS, many small contaminants, and very fine constituents | Desalination, high-purity water, brackish groundwater, and advanced reuse | Higher pressure, brine production, fouling, and scaling risk. |
Do not select RO when a looser membrane or conventional treatment process can meet the water quality goal. The tighter the membrane, the more important pressure, fouling control, recovery, and concentrate management become.
Key Factors That Control RO Performance
Reverse osmosis performance is controlled by the interaction between feedwater chemistry, membrane properties, hydraulic conditions, and operating targets. A system that looks good on paper can fail quickly if recovery is pushed too high, pretreatment is weak, or concentrate disposal is underestimated.
| Factor | Why it matters | Engineering implication |
|---|---|---|
| Feedwater TDS | Higher salinity increases osmotic pressure and energy demand. | Seawater RO needs much higher pressure than brackish water RO. |
| Recovery | Higher recovery produces more permeate from the same feed flow but concentrates salts in the brine. | Excessive recovery increases scaling, fouling, and concentrate quality challenges. |
| Flux | Flux is the permeate flow rate per membrane area. | High flux can reduce membrane area but may accelerate fouling and cleaning frequency. |
| Pretreatment quality | Particles, organics, biological growth, chlorine, hardness, and metals can damage or foul membranes. | Pretreatment often determines whether the RO system is stable or maintenance-heavy. |
| Temperature | Water viscosity and membrane permeability vary with temperature. | Seasonal temperature shifts can change permeate flow and normalized performance trends. |
| Concentrate disposal | RO creates a waste stream with elevated salts and contaminants. | Brine disposal can be the limiting cost, permitting issue, or environmental constraint. |
Reverse Osmosis Equations and Operating Metrics
Engineers use a few simple metrics to describe RO operation. These do not replace full membrane design software, but they help readers understand system performance, troubleshooting trends, and whether a process is being pushed too aggressively.
Recovery is the percentage of feedwater converted into permeate. Higher recovery reduces concentrate volume, but it also increases salt concentration near the membrane and can raise scaling risk.
Salt rejection compares feed concentration to permeate concentration. A drop in rejection may indicate membrane damage, seal leakage, changes in feed chemistry, or operation outside the membrane’s intended range.
Concentrate flow is the portion of feedwater that does not become permeate. This stream must be managed through discharge, deep well injection, evaporation, blending, further treatment, or another project-specific strategy.
Simple RO recovery example
If an RO system receives 1.0 MGD of feedwater and produces 0.75 MGD of permeate, the recovery is 75%. The remaining 0.25 MGD becomes concentrate. This does not mean contaminants disappear; it means they are carried into a smaller reject stream that must be managed.
- \(Q_f\) Feed flow entering the RO system, commonly measured in gpm, m³/h, MGD, or L/s.
- \(Q_p\) Permeate flow produced by the membrane system.
- \(Q_c\) Concentrate or reject flow leaving the membrane system.
- \(C_f\) Feed concentration for a target contaminant, often measured as mg/L or conductivity/TDS equivalent.
- \(C_p\) Permeate concentration after membrane treatment.
Reverse Osmosis Design Tradeoffs
RO design is a balance between water quality, reliability, energy, membrane life, waste production, and capital cost. A design that maximizes one goal can create problems elsewhere in the system.
| Design decision | Potential benefit | Tradeoff |
|---|---|---|
| Higher recovery | More permeate from the same feed flow and less concentrate volume. | Higher scaling risk, higher brine concentration, and more difficult concentrate management. |
| Higher flux | Less membrane area and potentially lower capital cost. | Greater fouling tendency and faster performance decline if pretreatment is weak. |
| More pretreatment | Better membrane protection and more stable operation. | Higher capital cost, chemical use, maintenance, and operational complexity. |
| Lower permeate TDS target | Higher quality finished water. | May require more stages, lower recovery, more pressure, or additional polishing. |
| More conservative design | Improved reliability, lower cleaning frequency, and longer membrane life. | Higher upfront cost and larger equipment footprint. |
Fouling, Scaling, and RO Maintenance
Fouling and scaling are among the most important real-world RO issues. They reduce permeate flow, increase pressure requirements, shorten membrane life, and increase cleaning frequency. The best maintenance strategy starts with understanding what type of performance loss is occurring.
| Problem | Common cause | Field symptom | Practical response |
|---|---|---|---|
| Particulate fouling | Silt, colloids, filter breakthrough, or poor upstream solids removal. | Rising differential pressure, especially near the front of the array. | Improve filtration, review turbidity and silt density index, and check cartridge filter performance. |
| Organic fouling | Natural organic matter, oils, polymers, or upstream chemical carryover. | Lower normalized permeate flow and more frequent cleanings. | Review pretreatment chemistry, chemical compatibility, and cleaning protocol. |
| Biofouling | Bacterial growth, nutrients, warm water, or weak biological control. | Pressure drop, slime formation, and recurring flow loss after cleaning. | Improve biological control, sanitation, upstream treatment, and monitoring. |
| Scaling | Calcium carbonate, sulfate salts, silica, barium, strontium, or high recovery. | Flux decline, pressure increase, or poor cleaning response. | Adjust recovery, pH, antiscalant, softening, blending, or staging. |
| Oxidant damage | Chlorine, ozone, or other incompatible oxidants reaching sensitive membranes. | Loss of salt rejection and higher permeate conductivity. | Verify membrane compatibility and dechlorination or oxidant control upstream. |
Cleaning should not be treated as a substitute for good pretreatment. If the same fouling pattern returns quickly after cleaning, the root cause is usually still present upstream.
Reverse Osmosis Design Review Checklist
The best RO designs begin with a clear treatment goal and then test whether the water chemistry, pretreatment, recovery target, cleaning strategy, and concentrate management plan can support that goal. The checklist below is a practical screening tool for reviewing an RO concept before detailed design.
Define the finished-water target → characterize the feedwater → identify foulants and scalants → select pretreatment → estimate pressure and recovery → check concentrate disposal → verify monitoring and cleaning strategy → confirm whether post-treatment is needed.
| Check or decision | What to look for | Why it matters |
|---|---|---|
| Finished-water goal | Target TDS, nitrate, fluoride, arsenic, hardness, conductivity, or reuse requirement. | The contaminant target controls membrane selection, staging, recovery, and post-treatment. |
| Feedwater variability | Seasonal changes in temperature, turbidity, organics, algae, iron, manganese, or salinity. | RO systems must handle real source-water variation, not just one lab sample. |
| Scaling potential | Calcium carbonate, sulfate salts, silica, barium, strontium, and high recovery operation. | Scaling can reduce flux, raise pressure, and shorten membrane life. |
| Fouling potential | Silt, colloids, natural organic matter, biofilm growth, oils, polymers, or upstream filter breakthrough. | Fouling often controls cleaning frequency and operating stability. |
| Oxidant exposure | Chlorine, chloramine, ozone, or oxidants upstream of thin-film composite membranes. | Some RO membranes can be damaged by oxidants if dechlorination or compatibility is not addressed. |
| Concentrate pathway | Discharge permit, sewer capacity, evaporation pond, deep well, blending, or further treatment. | Brine management can be more difficult than producing the permeate. |
| Post-treatment needs | Low alkalinity, low hardness, low pH stability, corrosivity, taste, and disinfectant residual. | RO permeate may need stabilization before distribution or final use. |
Example: Reading an RO Process Flow
Consider a brackish groundwater treatment plant where the source water has elevated TDS and hardness. A typical process may include raw water pumping, pretreatment for suspended solids and scaling control, cartridge filtration, RO membranes, permeate stabilization, disinfection, storage, and concentrate disposal.
Example assumptions
Suppose the feed flow is 1.0 MGD and the system operates at 75% recovery. The permeate flow is approximately 0.75 MGD and the concentrate flow is approximately 0.25 MGD. If feed TDS is 2,000 mg/L and permeate TDS is 80 mg/L, the apparent salt rejection is about 96%.
Interpretation or engineering meaning
A 96% rejection value indicates strong TDS reduction, but the engineer would still compare the permeate quality with the finished-water target and track whether rejection changes over time. The 75% recovery may look efficient from a water-production standpoint, but the concentrate stream carries a much higher salt load than the feed.
Engineering Judgment and Field Reality
Reverse osmosis diagrams often show clean separation: feedwater enters, permeate exits, and brine leaves the system. Real RO operation is more complicated. Membrane performance changes with temperature, fouling, scaling, cleaning history, feedwater variability, and how evenly flow is distributed through pressure vessels.
Experienced engineers look at normalized permeate flow, differential pressure, permeate conductivity, feed pressure, recovery, pH, temperature, and cleaning frequency together. A single number rarely tells the full story. For example, lower permeate flow may be caused by colder water rather than fouling, while rising differential pressure across the first stage may point toward particulate or biological fouling.
RO problems are often upstream problems. Poor clarification, overloaded filters, oxidant carryover, inadequate cartridge filtration, unstable source water, or incompatible chemicals can appear later as membrane fouling, scaling, pressure rise, and reduced permeate quality.
When This Breaks Down
Reverse osmosis is powerful, but it is not a universal solution. The simplified idea of “water passes, contaminants stay behind” breaks down when feedwater quality, operating targets, or system maintenance are not compatible with membrane operation.
- High fouling load: Algae, organics, colloids, iron, manganese, or biofilm can reduce membrane productivity if pretreatment is inadequate.
- High scaling risk: Hardness, silica, sulfate salts, and high recovery can cause mineral precipitation on the membrane surface.
- Poor brine management: A technically effective RO system may still be impractical if concentrate disposal is too expensive or difficult to permit.
- Unstable post-treatment: Low-mineral permeate may be corrosive or poorly buffered unless pH, alkalinity, hardness, and disinfection are adjusted.
- Wrong treatment target: RO may be excessive for problems better solved by conventional filtration, activated carbon, ion exchange, oxidation, or disinfection.
Common Mistakes and Practical Checks
Many RO misunderstandings come from treating reverse osmosis as a single filter rather than a pressure-driven treatment system. In engineering applications, the membrane is only one part of a larger process that must be designed, monitored, cleaned, and operated.
- Ignoring the concentrate stream: RO does not destroy contaminants; it separates them into a smaller, more concentrated waste stream.
- Assuming all contaminants are removed equally: Removal varies by contaminant chemistry, membrane type, condition, and operating environment.
- Skipping pretreatment: Suspended solids, organics, hardness, oxidants, and biological growth can shorten membrane life.
- Pushing recovery too high: Higher recovery can reduce waste flow but increase scaling and fouling risk.
- Forgetting post-treatment: RO permeate may need stabilization, remineralization, blending, or disinfection before use.
Do not evaluate RO only by permeate quality. A good design also checks feedwater variability, membrane protection, energy use, cleaning frequency, recovery, concentrate disposal, and finished-water stability.
Engineering References and Design Guidance
Reverse osmosis design should be supported by project-specific water quality data, membrane manufacturer criteria, pilot testing when needed, regulatory requirements, and recognized membrane treatment guidance. Public guidance is especially useful for terminology, treatment-process comparison, and understanding where RO fits within the broader membrane process family.
- AWWA membrane process guidance: AWWA membrane processes overview explains major membrane treatment categories, including reverse osmosis, and helps place RO in context with microfiltration, ultrafiltration, nanofiltration, and other membrane approaches.
- Project-specific criteria: Source water quality, finished-water standards, concentrate disposal options, owner requirements, and local permitting conditions often control the final RO configuration.
- Engineering use: Engineers combine published guidance with bench testing, pilot testing, membrane projections, antiscalant review, cleaning protocols, and lifecycle cost analysis before finalizing an RO system.
Frequently Asked Questions
Reverse osmosis is a pressure-driven membrane treatment process that forces water through a semi-permeable membrane while leaving many dissolved salts, minerals, metals, and other contaminants in a concentrate or brine stream. In engineered water systems, RO is commonly used for desalination, brackish groundwater treatment, high-purity process water, and advanced reuse.
Reverse osmosis can remove or strongly reduce total dissolved solids, sodium, chloride, hardness minerals, nitrate, fluoride, arsenic, lead, sulfates, some PFAS compounds, bacteria, and viruses, depending on membrane type, pretreatment, operating pressure, recovery, and system condition. Performance should be verified against the target contaminants and finished-water requirements.
Reverse osmosis needs pretreatment because RO membranes are vulnerable to fouling, scaling, oxidation, and plugging. Pretreatment may reduce suspended solids, organics, hardness, iron, manganese, chlorine, biological growth, or fine particles so the membrane can operate at stable flux, pressure, and recovery.
Permeate is the treated water that passes through the RO membrane. Concentrate, also called brine or reject, is the stream that carries the salts and contaminants that did not pass through the membrane. A complete RO design must account for both the desired permeate quality and the disposal or management of the concentrate stream.
Reverse osmosis is often casually called filtration, but it is more accurately a pressure-driven membrane separation process. Conventional filters mainly remove suspended particles, while RO membranes can separate water from many dissolved ions and small contaminants when the system is properly designed and operated.
Summary and Next Steps
Reverse osmosis is a pressure-driven membrane process used to separate clean permeate from dissolved salts and other contaminants. It is one of the most important advanced treatment options for desalination, brackish water treatment, high-purity industrial water, and water reuse.
The most important engineering lesson is that RO performance depends on the entire system: pretreatment, membrane selection, pressure, recovery, flux, fouling control, scaling control, monitoring, cleaning, concentrate disposal, and post-treatment. A strong RO design protects the membrane while producing stable finished water at a practical operating cost.
Where to go next
Continue your learning path with related Turn2Engineering resources.
-
Water Treatment Processes
Understand how RO fits into a broader treatment train with clarification, filtration, disinfection, and advanced treatment.
-
Water Treatment Plant Components
Review the major equipment and process units used in municipal and industrial water treatment systems.
-
Water Softening
Review hardness removal concepts that often affect RO scaling, pretreatment, and finished-water stabilization.