Key Takeaways
- Core idea: Atomic structure describes how protons, neutrons, and electrons are arranged within an atom.
- Engineering use: Atomic structure helps explain bonding, crystal structure, conductivity, hardness, ductility, brittleness, reactivity, and other material properties.
- What controls it: The nucleus controls atomic identity and mass, while electrons—especially valence electrons—control bonding and many property trends.
- Practical check: Atomic structure is only the starting point; final material behavior also depends on bonding, defects, grain structure, phases, and processing.
Table of Contents
Introduction
Atomic structure is the arrangement of protons, neutrons, and electrons within an atom. In materials science, it matters because electrons control bonding, bonding controls atomic arrangement, and atomic arrangement helps determine mechanical, electrical, thermal, magnetic, optical, and chemical properties.
How Atomic Structure Connects to Material Properties
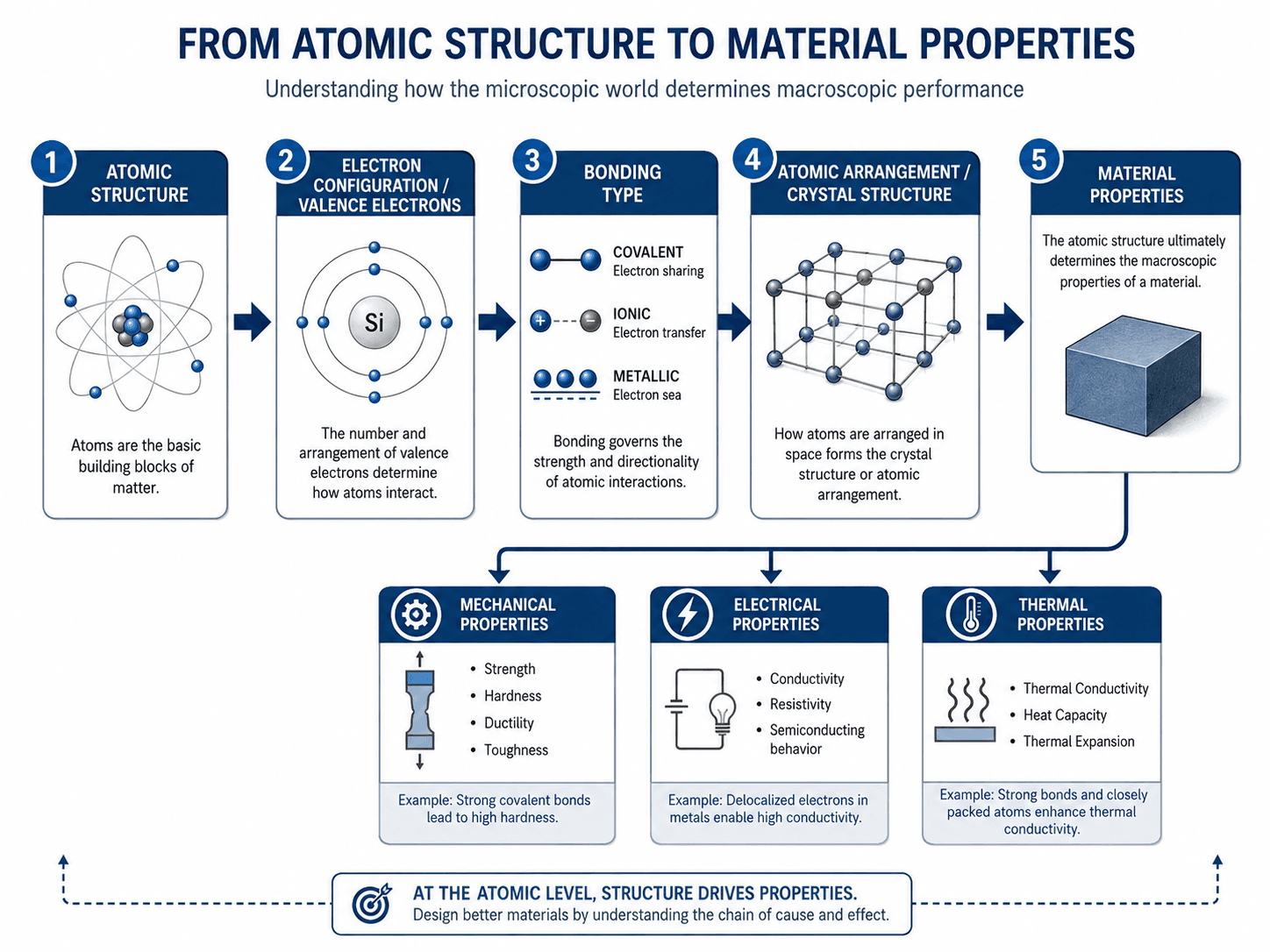
Start at the left side of the diagram and follow the pathway toward material properties. The most important idea is that atoms do not determine material behavior in isolation; bonding, structure, and scale all matter.
What Is Atomic Structure?
Atomic structure describes the internal makeup of an atom: a dense nucleus containing protons and neutrons, surrounded by electrons that occupy regions often described as shells, orbitals, or an electron cloud. The number of protons identifies the element, while the electron arrangement helps determine how that element bonds with other atoms.
For engineering materials, atomic structure is important because materials are built from atoms. The way atoms bond, pack, share electrons, and respond to energy determines many observable properties, including stiffness, conductivity, melting temperature, corrosion behavior, hardness, brittleness, and optical response.
Atomic structure explains why elements behave differently, but it does not fully predict final material performance by itself. Real materials also depend on crystal structure, defects, grain size, phases, processing history, and service environment.
Parts of an Atom: Protons, Neutrons, and Electrons
The three basic subatomic particles are protons, neutrons, and electrons. Protons and neutrons are located in the nucleus and account for most of the atom’s mass. Electrons occupy the surrounding electron cloud and strongly influence bonding, conductivity, reactivity, and many material property trends.
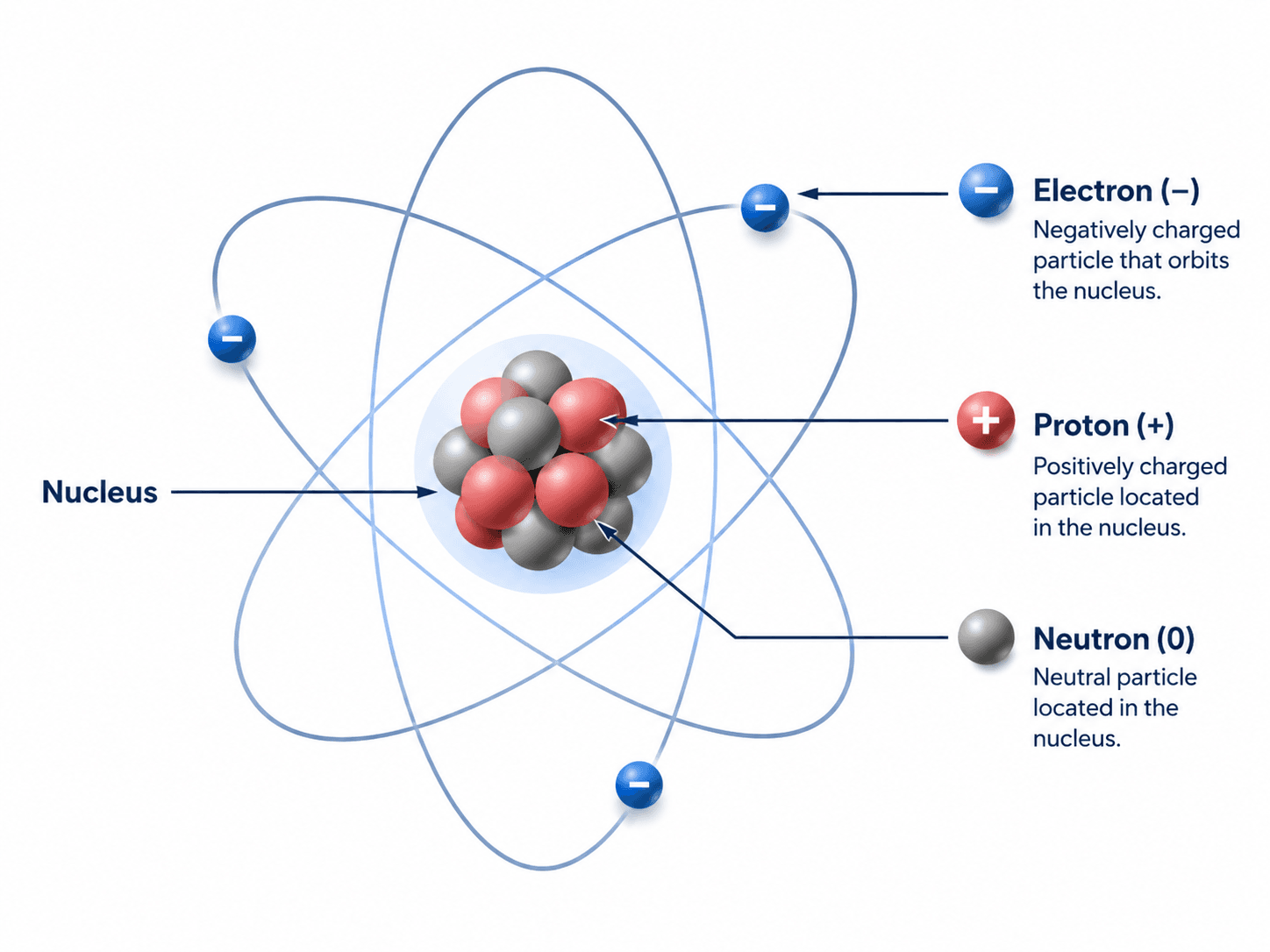
| Particle | Charge | Location | Why it matters in materials science |
|---|---|---|---|
| Proton | Positive | Nucleus | Determines the atomic number and identifies the element. |
| Neutron | Neutral | Nucleus | Affects isotope mass and nuclear stability but usually has less direct effect on ordinary bonding than electrons. |
| Electron | Negative | Electron cloud around the nucleus | Controls bonding, chemical reactivity, conductivity, optical behavior, and many property trends. |
Atomic Number, Mass Number, Atomic Mass, and Isotopes
Several basic terms are used to describe atomic structure. These terms are often confused, but they answer different questions about element identity, nuclear makeup, and average mass.
| Term | Meaning | Simple relationship | Why it matters |
|---|---|---|---|
| Atomic number | Number of protons in the nucleus. | \(Z = \text{protons}\) | Identifies the element. Carbon always has 6 protons; iron always has 26 protons. |
| Mass number | Total number of protons and neutrons in one atom. | \(A = \text{protons} + \text{neutrons}\) | Identifies a specific isotope of an element. |
| Atomic mass | Weighted average mass of naturally occurring isotopes. | Average value, not usually a whole number | Used in chemistry and materials calculations involving moles and composition. |
| Isotope | Atoms of the same element with different neutron counts. | Same \(Z\), different \(A\) | Important in nuclear materials, dating methods, radiation behavior, and some specialized engineering applications. |
For most materials property discussions, electrons and bonding matter more than isotope differences. Isotopes become more important in nuclear, radiation, and isotope-sensitive applications.
Electron Configuration and Valence Electrons
Electron configuration describes how electrons are arranged around the nucleus. For materials science, the most important electrons are usually the outermost electrons, called valence electrons, because they participate most directly in bonding.
Valence electrons help explain why sodium reacts strongly, why copper conducts electricity, why carbon can form diamond or graphite, and why silicon behaves as a semiconductor. In engineering materials, electron behavior connects atomic structure to bonding type, band behavior, conductivity, reactivity, and optical response.
| Electron concept | What it means | Material property connection |
|---|---|---|
| Valence electrons | Electrons in the outermost energy level involved in bonding. | Control bonding type, chemical reactivity, and many trends across the periodic table. |
| Electron sharing | Atoms share electrons to form covalent bonds. | Important in diamond, ceramics, polymers, and semiconductors. |
| Electron transfer | One atom gives up electrons and another accepts them. | Important in ionic solids such as many ceramics and salts. |
| Delocalized electrons | Electrons move through a larger metallic structure rather than staying between two atoms. | Explains why many metals conduct electricity and heat well. |
Atomic Structure and Bonding Types
Atomic structure controls bonding because electrons determine how atoms interact. Bonding type strongly affects stiffness, melting point, brittleness, ductility, electrical conductivity, thermal conductivity, and chemical resistance.
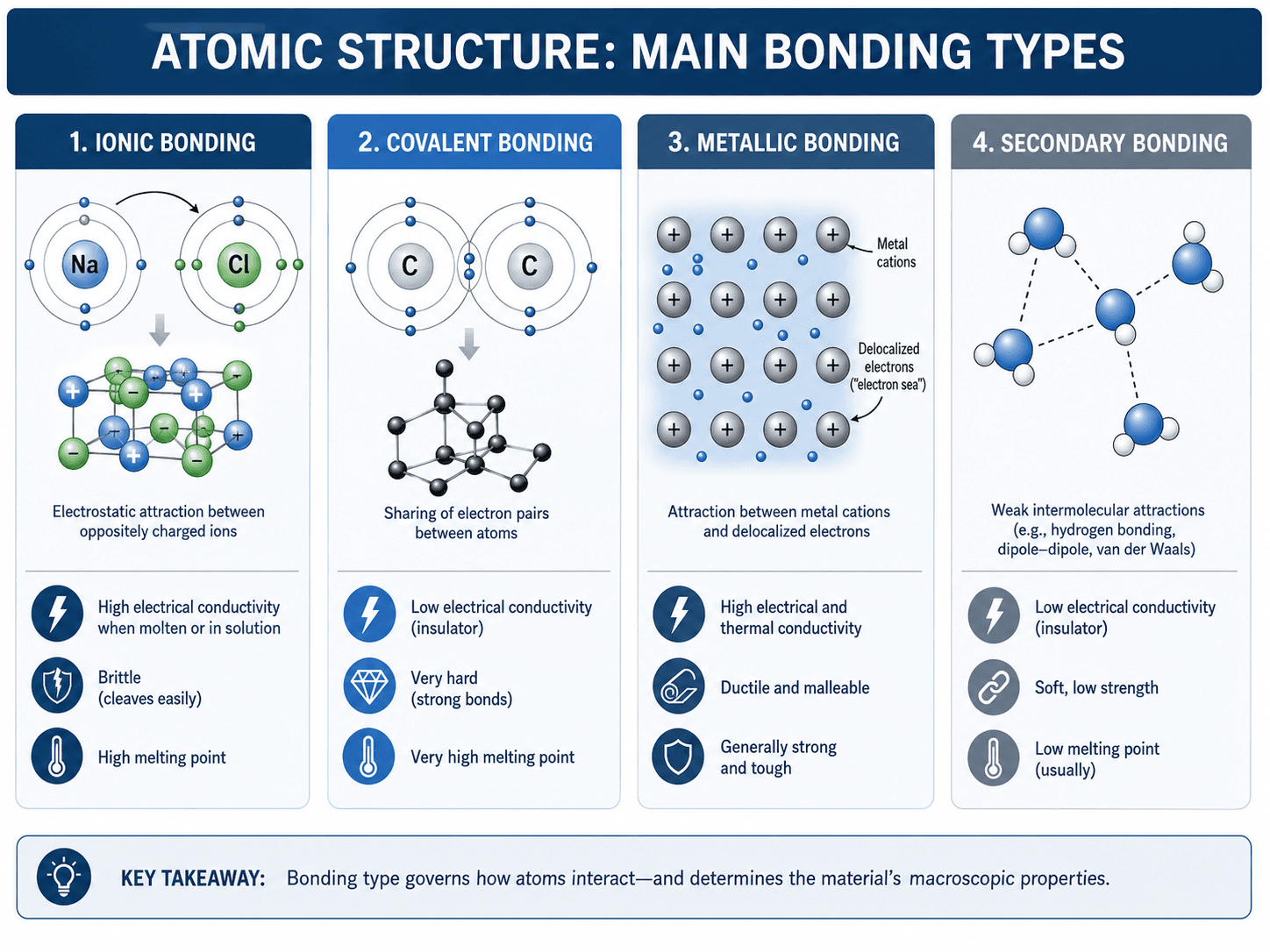
| Bonding type | Electron behavior | Common material examples | Typical property effect |
|---|---|---|---|
| Ionic bonding | Electrons transfer between atoms, creating oppositely charged ions. | Many ceramics, salts, oxides. | Often high melting point, brittle behavior, and low electrical conductivity as a solid. |
| Covalent bonding | Atoms share electron pairs directionally. | Diamond, silicon, many ceramics, polymer chains. | Can produce high hardness, high stiffness, strong directionality, and semiconducting behavior. |
| Metallic bonding | Positive metal ions are surrounded by delocalized electrons. | Steel, aluminum, copper, titanium, nickel alloys. | Often high electrical and thermal conductivity, ductility, and toughness. |
| Secondary bonding | Weak attractions occur between molecules or chain segments. | Polymers, molecular solids, layered materials. | Influences melting/softening behavior, flexibility, thermal response, and polymer properties. |
Atomic Structure vs Crystal Structure vs Microstructure
Atomic structure is often confused with crystal structure and microstructure. These ideas are related, but they describe different length scales.
| Concept | Scale | What it describes | Why it matters |
|---|---|---|---|
| Atomic structure | Inside a single atom | Protons, neutrons, electrons, electron configuration, and valence behavior. | Explains element identity and bonding tendencies. |
| Crystal structure | Arrangement of atoms in a solid | Repeating atom pattern such as body-centered cubic, face-centered cubic, hexagonal close-packed, or other lattice types. | Influences slip systems, ductility, density, anisotropy, and phase behavior. |
| Microstructure | Features visible at microscopic material scale | Grains, phases, precipitates, pores, inclusions, cracks, and defects. | Often controls real strength, toughness, fatigue behavior, corrosion resistance, and failure modes. |
A material can have the same chemical elements but very different properties because its crystal structure, microstructure, heat treatment, and defects are different.
How Atomic Structure Affects Material Properties
Atomic structure influences material properties by controlling how atoms bond and how electrons behave. The effect becomes visible through higher-level structure: crystal arrangement, defects, grain boundaries, phases, molecular chains, and processing history.
| Property category | Atomic-level connection | Engineering example |
|---|---|---|
| Mechanical properties | Bond strength, bond directionality, crystal structure, and defects influence stiffness, strength, hardness, ductility, and brittleness. | Diamond is extremely hard because of strong covalent bonding in a rigid network. |
| Electrical properties | Electron mobility and band behavior control conductivity, insulation, and semiconducting response. | Copper conducts well because metallic bonding provides mobile electrons. |
| Thermal properties | Bond strength, electron movement, atomic mass, and lattice vibrations influence heat capacity, thermal expansion, and thermal conductivity. | Metals often conduct heat well because mobile electrons help carry thermal energy. |
| Chemical properties | Valence electrons and bonding tendencies control reactivity, oxidation, corrosion, and compatibility. | Aluminum forms a protective oxide layer that changes its surface behavior. |
| Magnetic properties | Electron spin, unpaired electrons, and atomic arrangement influence magnetic response. | Iron can exhibit strong magnetic behavior due to electron structure and magnetic domain behavior. |
| Optical properties | Electron transitions and band structure affect absorption, reflection, transparency, and color. | Semiconductors absorb and emit light based on their band gap. |
Atomic Structure in Metals, Ceramics, Polymers, and Semiconductors
Different material classes behave differently because their atomic structure, electron behavior, bonding, and atom arrangement are different. This is why materials science often starts with atoms before moving to properties and design.
| Material class | Atomic and bonding pattern | Typical property result |
|---|---|---|
| Metals | Metal atoms share delocalized electrons in metallic bonding. | Good electrical and thermal conductivity, ductility, toughness, and formability. |
| Ceramics | Often ionic, covalent, or mixed bonding with strong directional or electrostatic bonds. | High hardness, high temperature resistance, brittleness, and low ductility. |
| Polymers | Covalent bonds along chains with secondary bonding between chains. | Low density, flexibility, viscoelastic behavior, and strong temperature sensitivity. |
| Semiconductors | Covalent atomic networks with electronic band behavior that can be controlled by doping. | Controlled conductivity, diode behavior, photovoltaic response, and electronic switching. |
| Composites | Combine materials with different atomic structures and bonding types. | Tailored strength, stiffness, weight, thermal behavior, and damage resistance. |
Atomic Structure Misconception Table
Atomic structure is a beginner-friendly topic, but simplified diagrams can create misunderstandings. The table below separates useful learning shortcuts from more accurate engineering interpretation.
Use atomic structure to understand element identity and bonding tendencies, then move up in scale to bonding, crystal structure, microstructure, and processing before predicting real material performance.
| Common misconception | Better engineering explanation | Why it matters |
|---|---|---|
| Electrons orbit the nucleus like planets. | Planet-like diagrams are learning models; real electrons are better described by probability regions and quantum states. | Electron behavior controls bonding and properties, so oversimplified pictures should not be treated as exact physical paths. |
| Atomic structure and crystal structure are the same thing. | Atomic structure describes particles inside atoms; crystal structure describes how atoms repeat in a solid. | Crystal structure affects ductility, slip, density, anisotropy, and phase changes. |
| Atomic structure alone determines material strength. | Strength also depends on bonds, defects, grain size, heat treatment, phases, cracks, and loading conditions. | Real engineering materials fail at the microstructure and defect scale, not just the atom scale. |
| Atomic mass and mass number are identical. | Mass number is protons plus neutrons for one isotope; atomic mass is an average over naturally occurring isotopes. | The distinction matters in chemistry calculations and isotope-sensitive applications. |
| All materials with the same element behave the same. | The same element can form different structures and phases with very different properties. | Carbon can appear as diamond, graphite, graphene, or amorphous carbon, each with different properties. |
Engineering Judgment and Field Reality
Atomic structure is essential for understanding why materials behave differently, but engineers rarely design from atomic structure alone. Real design decisions usually require property data, standards, testing, processing history, environmental exposure, and failure mode analysis.
Textbook explanations often move from atom to property in a straight line. Real materials are more complicated because defects, impurities, grain boundaries, phase transformations, manufacturing processes, and service conditions can dominate performance.
For example, two steels may both contain iron atoms and metallic bonding, but heat treatment, carbon content, alloying, grain size, inclusions, residual stress, and corrosion exposure can make their strength and toughness very different.
When This Breaks Down
Atomic structure is a powerful starting point, but it becomes incomplete when used as the only explanation for material behavior.
- Microstructure dominates: Grain size, cracks, voids, inclusions, and phase distribution can control failure more directly than atomic identity.
- Processing changes properties: Heat treatment, cold working, sintering, casting, welding, and additive manufacturing can change the final structure.
- Temperature changes behavior: Materials can soften, embrittle, expand, transform phases, or lose strength at different temperatures.
- Environment changes performance: Corrosion, oxidation, moisture, radiation, chemicals, and UV exposure can alter surface and bulk behavior.
- Scale matters: Atomic explanations are useful, but engineering design often depends on macroscale loads, geometry, tolerances, and safety factors.
Useful Reference for Atomic and Electronic Structure
Atomic structure is a foundational topic in chemistry and materials science, so the best reference for deeper study is an educational source that connects atoms, electrons, bonding, and solid-state behavior.
- MIT OpenCourseWare-based solid-state chemistry notes: atomic and electronic structure notes from LibreTexts provide a deeper foundation for atomic number, atomic mass, isotopes, electron structure, and the role of atoms in solid-state materials.
- Engineering use: Use atomic structure as the first step in understanding bonding and property trends, then verify final material behavior using material data, testing, processing history, and application-specific requirements.
Frequently Asked Questions
Atomic structure is the arrangement of protons, neutrons, and electrons within an atom. Protons and neutrons form the nucleus, while electrons occupy the surrounding electron cloud. In materials science, atomic structure matters because electrons and bonding help determine how materials conduct, deform, melt, react, and fail.
Atomic structure is important because it controls how atoms bond and arrange themselves in materials. Those bonds and arrangements influence mechanical strength, electrical conductivity, thermal behavior, chemical stability, magnetic response, optical behavior, and many other material properties.
Atomic structure describes the subatomic parts of an atom, including protons, neutrons, and electrons. Crystal structure describes how atoms are arranged in a repeating pattern within a solid. Atomic structure helps explain bonding, while crystal structure helps explain how bonded atoms form an ordered material.
Electrons, especially valence electrons, control bonding behavior and strongly influence material properties. They help determine whether a material behaves like a metal, ceramic, polymer, or semiconductor and affect conductivity, reactivity, bonding strength, and optical response.
No. Atomic structure is a foundation for material behavior, but final strength also depends on bonding, crystal structure, defects, grain size, phases, processing history, temperature, loading direction, and service environment.
Summary and Next Steps
Atomic structure explains the internal makeup of atoms and provides the foundation for understanding why materials behave the way they do. Protons identify the element, neutrons affect isotope mass, and electrons control bonding and many property trends.
The key engineering connection is the pathway from atomic structure to electron behavior, bonding, atom arrangement, crystal structure, microstructure, and final material properties. To use the concept correctly, move beyond simplified atom diagrams and connect atomic-scale behavior to real material data and processing history.
Where to go next
Continue your learning path with related Turn2Engineering resources.
-
Material Properties
Learn how atomic structure connects to broader categories such as mechanical, thermal, electrical, chemical, magnetic, optical, and acoustic properties.
-
Mechanical Properties
See how bonding, structure, defects, and processing influence strength, stiffness, ductility, hardness, and toughness.
-
Materials Science
Explore the broader materials science hub for structure-property relationships and engineering material selection.