Key Takeaways
- Core idea: An isolated system exchanges neither mass nor energy with its surroundings.
- Engineering use: Engineers use isolated-system assumptions to simplify energy balances, entropy analysis, insulated tanks, rapid processes, and ideal laboratory models.
- What controls it: The boundary condition controls the classification: mass transfer, heat transfer, and work transfer must all be blocked.
- Practical check: Real systems are only approximately isolated because perfect insulation and perfect boundary separation are not achievable in practice.
Table of Contents
Introduction
An isolated system is a thermodynamic system that exchanges neither mass nor energy with its surroundings. No matter crosses the boundary, and ideally no heat or work crosses it either. This makes isolated systems useful for studying conservation of energy, entropy generation, equilibrium, and the limiting behavior of real insulated systems.
Open vs Closed vs Isolated Systems Diagram
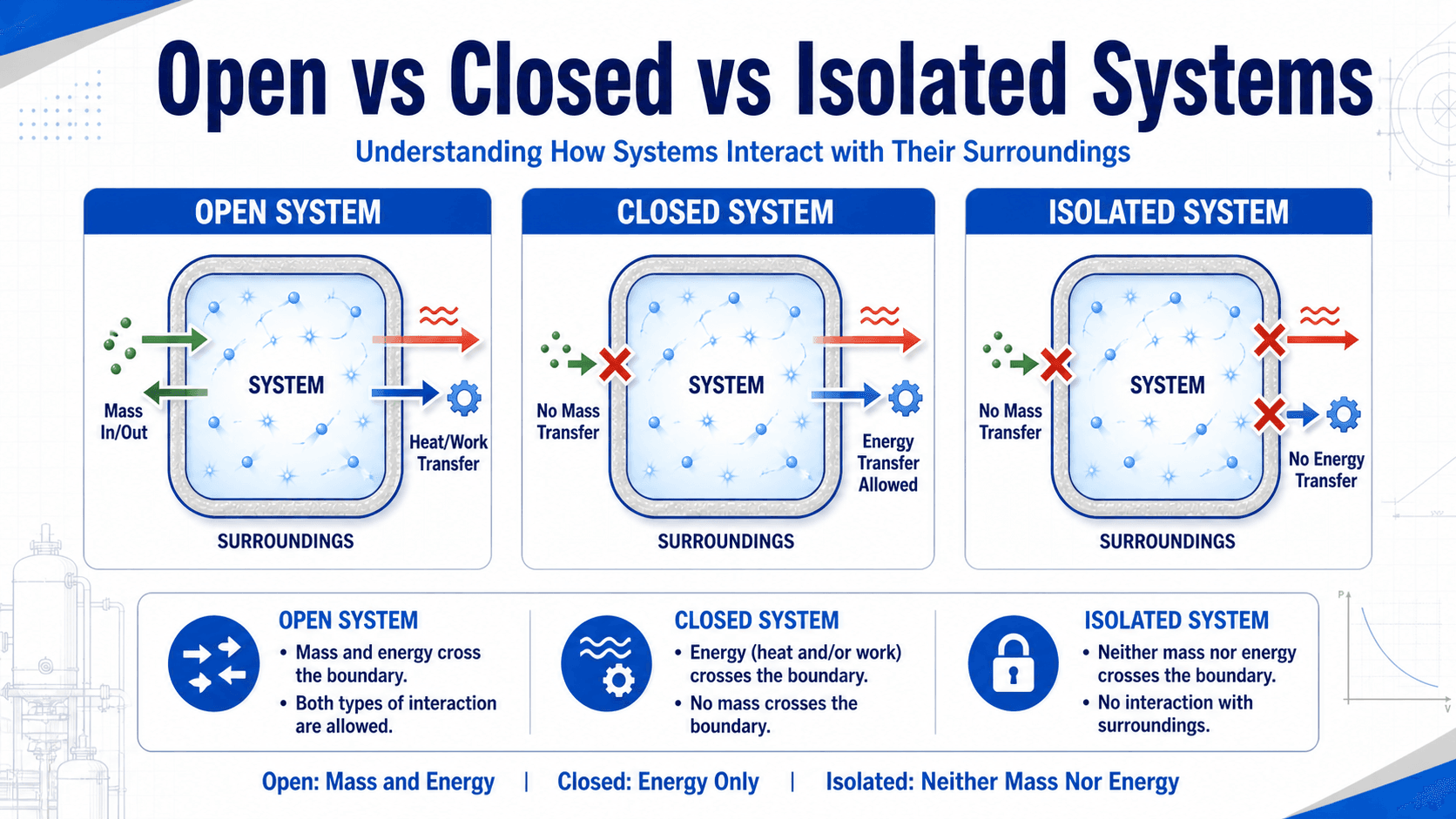
The fastest way to classify a thermodynamic system is to ask what crosses the boundary. If neither matter nor energy crosses, the model is isolated.
What Is an Isolated System?
An isolated system is a system separated from its surroundings by a boundary that does not permit mass transfer, heat transfer, or work transfer. In thermodynamics, this is the most restrictive system classification because the system cannot exchange matter or energy with anything outside the boundary.
The word “isolated” does not mean that nothing happens inside the system. Molecules can move, temperatures can equalize, chemical reactions can occur, and energy can change form internally. The key point is that the total mass and total energy of the system remain fixed because nothing crosses the boundary.
This distinction is especially important when comparing isolated systems with closed systems and open systems. A closed system blocks mass transfer but may still exchange heat or work. An open system can exchange both mass and energy.
How an Isolated System Boundary Works
The boundary is the defining feature of any thermodynamic system. For an isolated system, the boundary is idealized so that no mass, heat, or work crosses it. In a diagram, this is often shown with blocked arrows for mass flow, heat flow, and mechanical work.
| Interaction | Can it cross an isolated boundary? | Engineering meaning |
|---|---|---|
| Mass transfer | No | No fluid, gas, vapor, particles, or material enter or leave the system. |
| Heat transfer | No | The boundary is treated as perfectly insulated, so \(Q = 0\). |
| Work transfer | No | No shaft work, boundary work, electrical work, or other external work crosses the boundary. |
| Internal energy redistribution | Yes, internally | Energy may transform or redistribute inside the system even though total energy is constant. |
System Boundary vs. Physical Wall
A thermodynamic boundary is a modeling choice, not always a literal wall. In engineering problems, the boundary may be drawn around a tank, a calorimeter, a gas volume, or a combined system plus surroundings. If the chosen boundary prevents all external mass and energy interactions, the model can be treated as isolated.
Ideal Isolation vs. Real Isolation
Perfect isolation is an ideal assumption. Real insulation slows heat transfer but does not eliminate it. Real seals reduce mass leakage but may not be perfect. Real equipment may also have small mechanical, electrical, or thermal interactions that become important over long time periods.
Energy and Entropy in an Isolated System
Isolated systems are important because they connect the first and second laws of thermodynamics in a clean way. The first law says total energy is conserved. The second law says entropy in an isolated system cannot decrease.
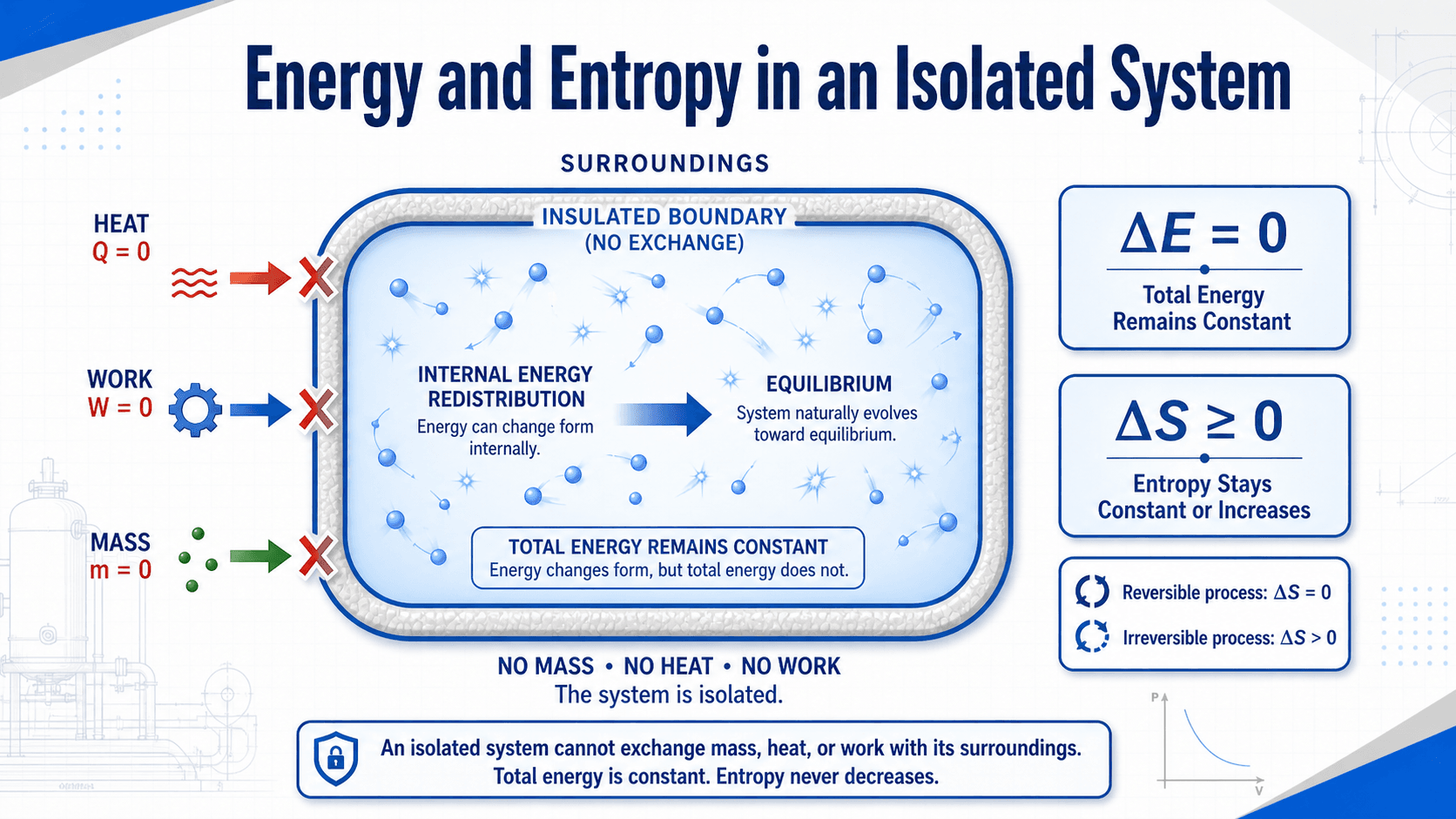
For a closed-system energy balance, the simplified first-law form is commonly written as:
For an isolated system, heat transfer and work transfer across the boundary are both zero:
This does not mean every form of energy inside the system is frozen. Kinetic energy can dissipate into internal energy, temperature differences can smooth out, and chemical or mechanical energy can convert internally. The total energy of the isolated system remains constant.
The entropy statement is equally important:
- \(Q\) Heat transferred across the system boundary. For an isolated system, \(Q = 0\).
- \(W\) Work transferred across the system boundary. For an isolated system, \(W = 0\).
- \(\Delta E\) Change in total system energy, including internal, kinetic, and potential energy. For an isolated system, \(\Delta E = 0\).
- \(\Delta S\) Change in entropy. For an isolated system, entropy remains constant for an ideal reversible process and increases for an irreversible process.
Examples of Isolated Systems
The best examples of isolated systems are idealized engineering models. Real systems can approach isolated behavior for a limited time, but they usually leak some heat, allow some small interaction, or depend on assumptions that break down over longer durations.
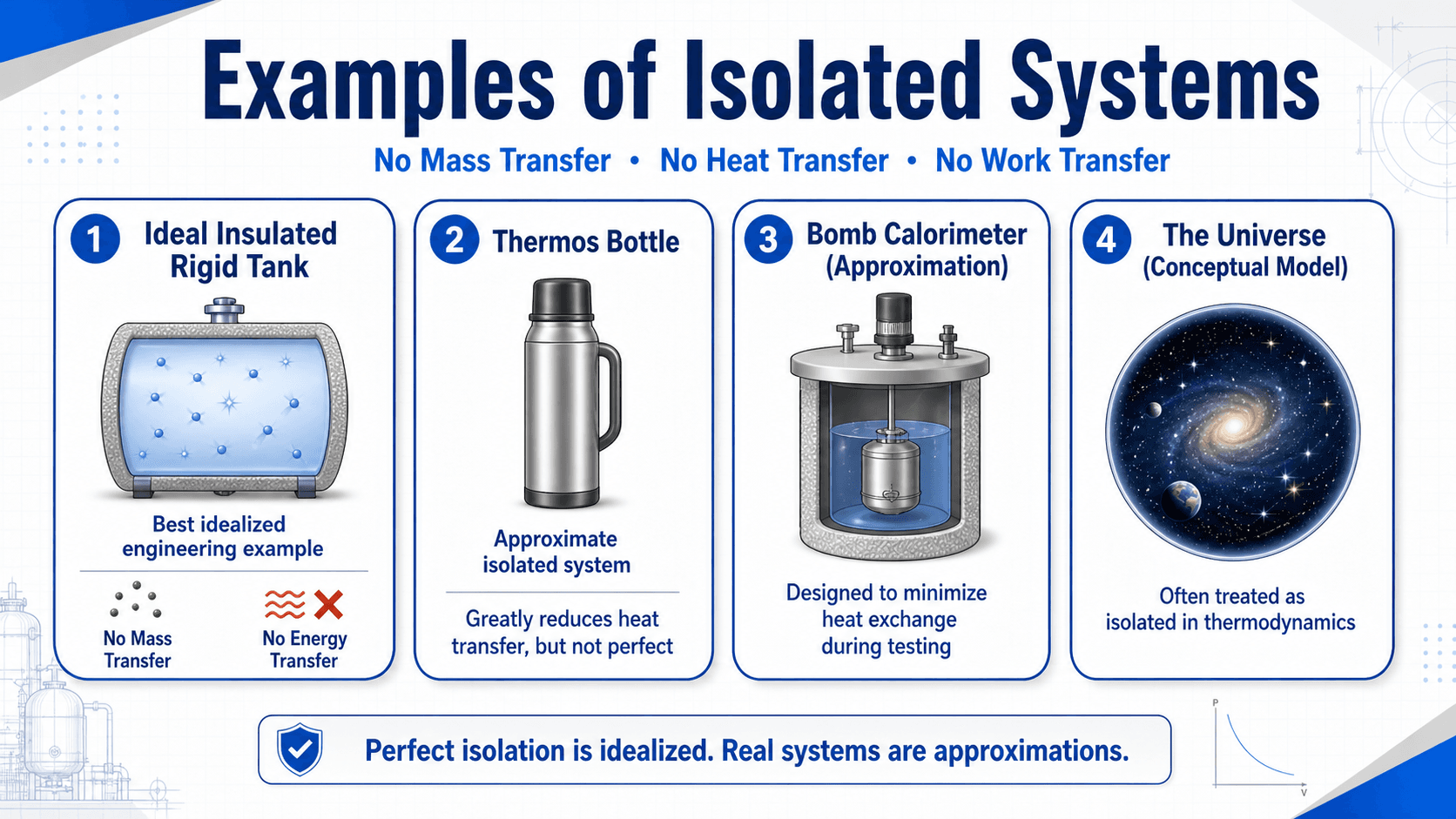
| Example | How isolated is it? | What the example teaches |
|---|---|---|
| Ideal insulated rigid tank | Ideal model | No mass crosses, no boundary work occurs, and perfect insulation prevents heat transfer. |
| Thermos bottle | Approximation | Mass is sealed in and heat transfer is greatly reduced, but not eliminated. |
| Bomb calorimeter | Approximation during testing | The device is designed to minimize heat exchange while measuring energy release. |
| The universe | Conceptual model | Often treated as isolated because there is no external surroundings in the thermodynamic model. |
A thermos is a useful example for beginners, but the best engineering example is usually an ideal insulated rigid tank because the mass, heat, and work assumptions can be stated explicitly.
Isolated vs. Closed vs. Open Systems
Many mistakes in thermodynamics come from mixing up system types. The classification depends on what crosses the boundary, not on the shape of the object or whether it is physically sealed.
| System type | Mass crosses boundary? | Energy crosses boundary? | Typical example |
|---|---|---|---|
| Open system | Yes | Yes | Turbine, compressor, pump, nozzle, boiler, heat exchanger |
| Closed system | No | Yes | Sealed piston-cylinder, sealed tank with heat transfer |
| Isolated system | No | No | Ideal insulated rigid tank, idealized universe model |
Why This Comparison Matters
If you classify the system incorrectly, the energy balance will be wrong. An open-system problem may need mass-flow terms and enthalpy. A closed-system problem does not include mass flow, but heat and work can still matter. An isolated-system problem removes external heat, work, and mass interactions, leaving only internal changes.
How Engineers Use Isolated-System Assumptions
Engineers use isolated systems as ideal models when outside interactions are negligible compared with the internal behavior being studied. This is common in introductory thermodynamics, laboratory testing, insulated equipment analysis, and rapid-process approximations.
- Estimating final equilibrium temperature after two materials are placed in an insulated container.
- Modeling short-duration processes where heat transfer is small because the process happens quickly.
- Understanding entropy generation caused by mixing, friction, free expansion, or internal irreversibility.
- Creating simplified control-mass problems where no mass enters or leaves the chosen boundary.
- Checking whether an energy balance should include external heat, work, or mass-flow terms.
Before treating a system as isolated, ask whether heat transfer, shaft work, boundary work, electrical work, leakage, or mass flow is small enough to ignore for the time scale and accuracy required.
Isolated System Classification Checklist
Use this checklist when deciding whether a thermodynamics problem can reasonably be modeled as an isolated system. The goal is not to prove the real object is perfectly isolated, but to decide whether the isolated-system assumption is appropriate for the analysis.
Start with the boundary. If mass crosses it, the system is not isolated. If heat crosses it, the system is not isolated. If external work crosses it, the system is not isolated. If all three are negligible for the problem, an isolated-system model may be reasonable.
| Check or decision | What to look for | Why it matters |
|---|---|---|
| Mass boundary | Valves, vents, leaks, flow inlets, flow outlets, phase escape, or material addition. | Any mass crossing the boundary disqualifies the system from being isolated. |
| Heat boundary | Temperature difference, insulation quality, exposed surface area, test duration, radiation, conduction paths. | Heat leakage may be small for short tests but important for long-duration behavior. |
| Work interaction | Moving pistons, rotating shafts, electrical power, springs, stirring devices, or deforming boundaries. | Work crossing the boundary means the system is not isolated even if it is insulated. |
| Time scale | Rapid event versus long-term storage or slow thermal drift. | A system may behave nearly isolated for seconds but not for hours or days. |
| Accuracy needed | Conceptual estimate, classroom problem, lab measurement, or design-quality calculation. | The higher the required accuracy, the less forgiving the isolated assumption becomes. |
Isolated System vs. Adiabatic System
An adiabatic system is not automatically an isolated system. Adiabatic only means no heat transfer across the boundary. The system may still exchange work, and depending on how the boundary is defined, it may also involve mass transfer.
| Concept | Heat transfer? | Work transfer? | Mass transfer? |
|---|---|---|---|
| Adiabatic system or process | No | May occur | Depends on system definition |
| Isolated system | No | No | No |
A well-insulated piston-cylinder can be adiabatic while still doing boundary work as the piston moves. That makes it adiabatic, but not isolated. To be isolated, the same system would also need no work transfer and no mass transfer.
Engineering Judgment and Field Reality
In real engineering work, isolated systems are usually approximations. Insulation has finite thermal resistance, seals can leak, instruments can disturb the system, and equipment may exchange small amounts of energy through supports, wiring, shafts, or test fixtures.
Experienced engineers focus on whether those interactions are negligible for the question being answered. A heat leak that is irrelevant during a five-second test may dominate a 24-hour storage calculation. A tiny shaft input may be negligible in a classroom example but important in precision calorimetry.
The isolated-system assumption is strongest when the system is well insulated, sealed, rigid, short-duration, and analyzed at a level where small losses are not controlling the result.
When This Breaks Down
The isolated-system model breaks down when external interactions are large enough to affect the result. This usually happens because the boundary is not truly impermeable, not truly insulated, not rigid, or not isolated from external work.
- Heat transfer becomes important over long durations, especially when a temperature difference exists between the system and surroundings.
- Mass leakage, venting, evaporation, or condensation across the chosen boundary changes the system mass.
- Moving boundaries, rotating shafts, electrical leads, magnetic devices, or stirrers introduce work transfer.
- Measurement devices disturb the system by adding thermal mass, heat conduction paths, or electrical input.
- The system boundary is drawn too narrowly, excluding surroundings that should have been included in the isolated model.
Common Mistakes and Practical Checks
Most isolated-system mistakes come from treating “sealed,” “insulated,” “adiabatic,” and “isolated” as if they mean the same thing. They do not. Each term describes a different boundary condition.
- Calling every sealed container isolated: A sealed container blocks mass transfer, but heat and work may still cross the boundary.
- Calling every insulated system isolated: Insulation reduces heat transfer, but work or mass transfer may still occur.
- Assuming internal energy cannot change form: Total energy is constant, but internal redistribution can still occur.
- Ignoring entropy generation: Irreversible internal processes can increase entropy even when no energy enters or leaves.
- Using isolated assumptions for long storage periods: Small heat leaks can become significant over time.
The most important check is whether energy crosses the boundary. If heat or work crosses, the system is not isolated even if no mass crosses.
Useful References and Thermodynamics Context
Isolated systems are usually taught through the first and second laws of thermodynamics, especially energy conservation and entropy. A strong reference should help readers connect the system-boundary concept to entropy and irreversibility.
- OpenStax University Physics: OpenStax University Physics Volume 2 section on entropy provides a useful educational reference for entropy, state functions, reversible processes, and the second-law behavior that supports isolated-system analysis.
- Engineering textbooks: Mechanical engineering thermodynamics courses commonly use isolated systems to introduce energy conservation, entropy generation, and final equilibrium states.
- Engineering use: The reference value is not a code requirement; it helps readers understand why isolated systems conserve total energy while entropy cannot decrease for spontaneous irreversible processes.
Frequently Asked Questions
An isolated system is a thermodynamic system that exchanges neither mass nor energy with its surroundings. In ideal form, no matter, heat, or work crosses the system boundary, although energy may still redistribute internally.
A closed system does not exchange mass with its surroundings, but it can exchange energy as heat or work. An isolated system is more restrictive because it exchanges neither mass nor energy across the boundary.
A thermos bottle is an approximate isolated system, not a perfect one. It greatly reduces heat transfer and blocks mass transfer when sealed, but small heat leaks still occur over time through the lid, walls, and radiation.
Entropy in an isolated system cannot decrease. It remains constant for an ideal reversible process and increases for irreversible processes such as mixing, friction, unrestrained expansion, and heat flow from hot regions to cold regions.
No. Adiabatic means no heat transfer, but work or mass transfer may still occur depending on the system. Isolated means no heat transfer, no work transfer, and no mass transfer across the boundary.
Summary and Next Steps
An isolated system is the strictest thermodynamic system type because no mass, heat, or work crosses the boundary. It is an ideal model used to study conservation of total energy, entropy generation, internal irreversibility, and equilibrium.
The practical engineering skill is knowing when the assumption is reasonable. If leakage, heat transfer, work transfer, or measurement disturbance affects the result, the model should be revised to a closed or open system instead.
Where to go next
Continue your learning path with related Turn2Engineering resources.
-
Closed Systems
Learn the related system type where mass is fixed but energy may still cross the boundary as heat or work.
-
Open Systems
Compare isolated systems with control volumes that allow both mass flow and energy transfer.
-
Thermodynamics
Explore the broader thermodynamics hub for system boundaries, energy balances, entropy, and engineering applications.