Key Takeaways
- Definition: Coulomb’s Law calculates the electric force between two charges based on charge magnitude, distance, and the surrounding medium.
- Main use: Engineers use it to estimate electrostatic attraction or repulsion in sensors, insulation, high-voltage equipment, capacitors, and electrostatic discharge problems.
- Watch for: The simple form assumes point charges, stationary charges, and a known permittivity; real conductors and dielectrics can change the field.
- Outcome: You will be able to solve for force, charge, or separation distance while checking sign convention, units, and physical assumptions.
Table of Contents
Electric force between two point charges
Coulomb’s Law relates electric force to charge magnitude, separation distance, and the permittivity of the surrounding medium.
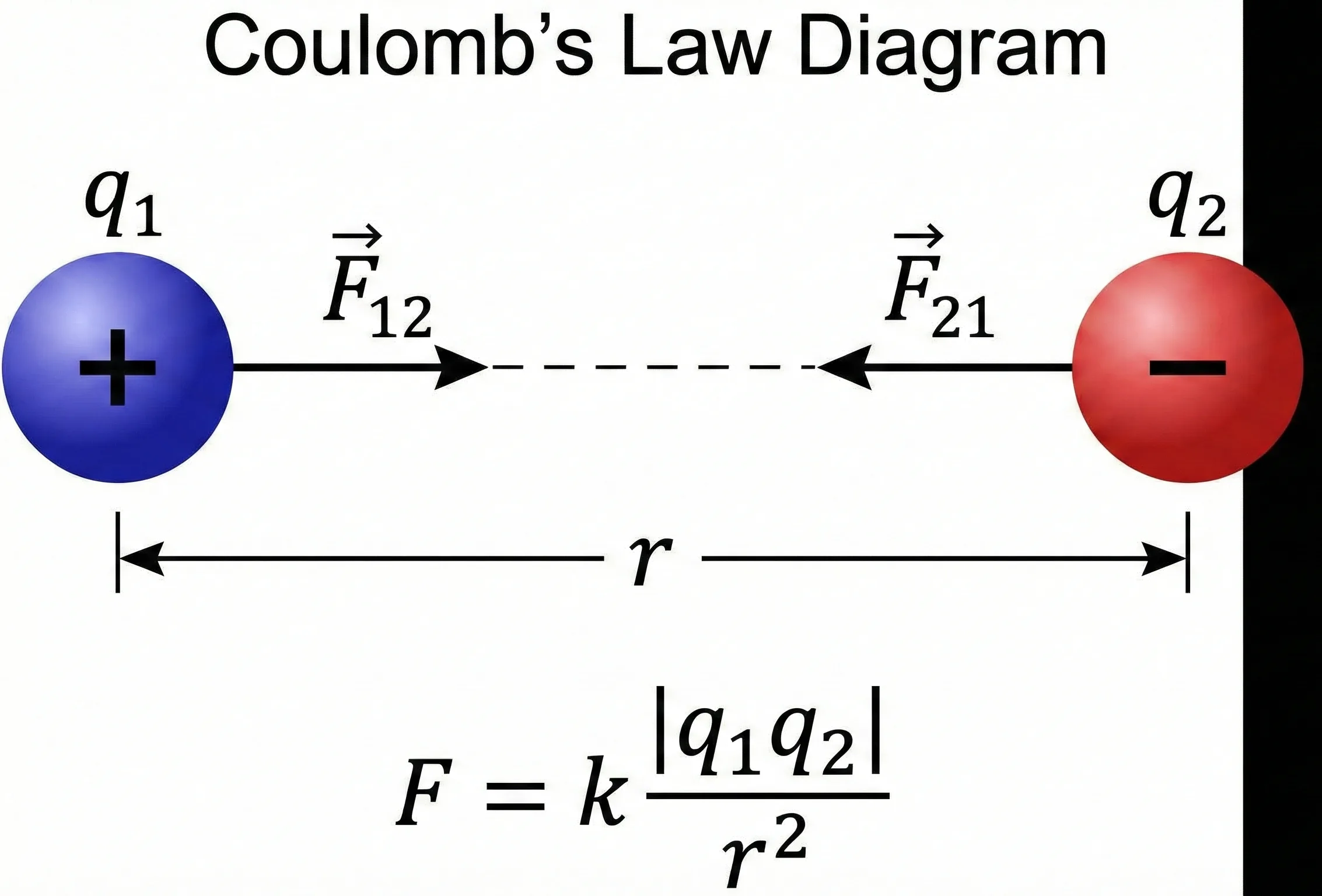
The first thing to notice is the distance term. Because distance is squared in the denominator, doubling the separation reduces the force to one-fourth, while halving the separation increases the force by four.
What is Coulomb’s Law?
Coulomb’s Law is the fundamental equation for the electrostatic force between two stationary electric charges. It tells you how strongly two charges attract or repel each other when their charge magnitudes and separation distance are known.
In engineering terms, Coulomb’s Law is the electric-force equivalent of a basic interaction model. It is not usually the final answer for a full device, but it is the starting point for understanding electric fields, capacitance, insulation stress, electrostatic discharge, charged particles, and force behavior in simple electrostatic systems.
The law is especially useful when the charges can be treated as point charges or when charged objects are far enough apart that their size and shape do not dominate the result.
The Coulomb’s Law formula
The most common scalar form of Coulomb’s Law in free space is:
This form gives the magnitude of the electrostatic force between two point charges. The force increases with larger charge magnitudes and decreases with the square of the separation distance.
Coulomb’s constant can also be written in terms of the permittivity of free space:
For a material medium instead of vacuum or air, the permittivity changes. A more general form is:
The direction of the force depends on the signs of the charges. Like charges repel, while opposite charges attract. The scalar equation above gives magnitude; the sign and direction must be interpreted from the charge arrangement.
Do not use only the force magnitude if direction matters. In vector problems, define a coordinate system, assign charge signs, and track whether the force points toward or away from the other charge.
Variables and units
Coulomb’s Law is unit-sensitive because electric charge values are often very small. Microcoulombs, nanocoulombs, and picocoulombs must be converted before using the SI form of the equation.
- \(F\) Electrostatic force between charges. SI unit: newtons (N).
- \(q_1\) First electric charge. SI unit: coulombs (C). Common practical units include µC, nC, and pC.
- \(q_2\) Second electric charge. SI unit: coulombs (C). The sign determines attraction or repulsion.
- \(r\) Center-to-center separation distance between point charges. SI unit: meters (m).
- \(k_e\) Coulomb’s constant, approximately \(8.99\times10^9\,\text{N·m}^2/\text{C}^2\) in free space.
- \(\varepsilon\) Permittivity of the medium. Higher permittivity reduces the force compared with free space.
Use coulombs and meters when using \(k_e = 8.99\times10^9\,\text{N·m}^2/\text{C}^2\). Convert \(1\,\mu\text{C}=10^{-6}\,\text{C}\), \(1\,\text{nC}=10^{-9}\,\text{C}\), and \(1\,\text{pC}=10^{-12}\,\text{C}\).
| Variable | Meaning | SI units | Common practical units | Engineering note |
|---|---|---|---|---|
| \(F\) | Electrostatic force | N | mN, µN | Use sign or vector direction separately from magnitude. |
| \(q_1, q_2\) | Electric charges | C | µC, nC, pC | Charge sign controls attraction or repulsion. |
| \(r\) | Charge separation | m | mm, cm, m | Use center-to-center distance for point-charge approximations. |
| \(k_e\) | Coulomb’s constant | N·m\(^2\)/C\(^2\) | Usually SI only | Applies to free space or air approximations. |
| \(\varepsilon\) | Permittivity | F/m | Relative permittivity \(\varepsilon_r\) | Use for dielectric materials instead of free space. |
Because \(F\propto1/r^2\), distance dominates quickly. A small error in charge spacing can create a much larger force error than the same percentage error in one charge value.
How to rearrange Coulomb’s Law
Coulomb’s Law can be rearranged to solve for force, one unknown charge, or separation distance. These rearrangements are common in physics, electrical engineering, and electrostatics problems.
When solving for distance, the square root means a four-times larger force requires only half the distance if the charges stay the same. Use this inverse-square intuition to catch unreasonable answers.
Worked example: force between two small charges
Example problem
Two small charged objects have charges \(q_1 = 3.0\,\mu\text{C}\) and \(q_2 = -2.0\,\mu\text{C}\). Their centers are separated by \(0.15\,\text{m}\). Estimate the electrostatic force magnitude and state whether the force is attractive or repulsive.
First convert charge values to coulombs:
Substitute magnitudes into Coulomb’s Law:
Simplify the result:
Since one charge is positive and the other is negative, the force is attractive. Each charge experiences the same force magnitude, but in opposite directions along the line connecting the two charges.
A force of a few newtons from microcoulomb charges can be realistic when the separation distance is small. If your result seems surprisingly large, recheck distance units and charge conversions first.
Where engineers use Coulomb’s Law
Coulomb’s Law is most useful as a first-principles model for electric force. In practical engineering, it often supports a larger electric-field, insulation, sensing, or circuit-design workflow.
- Electrostatic discharge: estimating why charge buildup can create strong forces, sparks, or sensitive-device risk.
- High-voltage design: understanding force and field trends near charged conductors, terminals, and insulation gaps.
- Capacitors and dielectrics: connecting charge, field, voltage, and material permittivity in idealized electrostatic systems.
- Sensors and actuators: analyzing electrostatic attraction in MEMS, capacitive sensors, charged particles, and small-scale devices.
- Physics and circuit fundamentals: building intuition for electric fields before applying voltage, capacitance, or current relationships.
Use Coulomb’s Law when you can approximate two interacting charges as point charges. Use electric-field methods when one charge interacts with a field. Use capacitance relationships when charge is stored on conductors with known geometry.
Assumptions and limitations
Coulomb’s Law is exact for ideal point charges at rest in a uniform medium. Real engineering objects are often conductors, dielectrics, electrodes, plates, wires, or surfaces with distributed charge, so the point-charge assumption must be checked.
- 1 The charges can be treated as point charges or as small compared with their separation distance.
- 2 The charges are stationary or moving slowly enough that magnetic and radiation effects are negligible.
- 3 The surrounding medium has a known and reasonably uniform permittivity.
- 4 Nearby conductors, grounds, shields, or dielectric boundaries do not significantly distort the field.
Neglected factors
The simple two-charge equation does not account for distributed charge, nonuniform materials, induced charge on nearby conductors, dielectric polarization, surface leakage, ionization, air breakdown, time-varying fields, or magnetic effects from moving charges.
Do not use the point-charge form directly for charged plates, complex electrode geometry, high-voltage clearances, or insulation design without checking electric-field distribution and material breakdown limits.
Engineering judgment and field reality
Real electrostatic systems rarely contain two clean point charges floating in isolation. Conductors redistribute charge, nearby grounded objects reshape fields, and dielectric materials reduce or redirect forces depending on permittivity and geometry.
In high-voltage hardware, the most important risk is often not the total force between two idealized charges. It is the local electric-field concentration at sharp edges, small gaps, contamination, humidity, or insulation defects.
Treat Coulomb’s Law as a first-pass force model. For actual hardware, follow with electric-field analysis, clearance checks, creepage considerations, dielectric strength review, or finite-element electrostatic modeling when geometry matters.
Common mistakes and engineering checks
- Forgetting charge conversions: using microcoulombs as coulombs creates a \(10^{12}\) error in the charge product.
- Using diameter instead of separation: \(r\) is the distance between charge centers or point-charge locations.
- Ignoring direction: the scalar equation gives magnitude, not the full vector force.
- Applying point-charge math to plates: charged plates, electrodes, and conductors need field or capacitance methods.
- Ignoring the medium: dielectric materials change permittivity and reduce force compared with free space.
After calculating force, double the distance mentally. If the force does not drop to about one-fourth, the inverse-square relationship was likely applied incorrectly.
| Check item | What to verify | Why it matters |
|---|---|---|
| Charge units | Convert µC, nC, or pC to C | Small charge-unit errors become huge force errors |
| Distance | Use meters and square the distance | The force changes with \(1/r^2\) |
| Direction | Check charge signs and line of action | Determines attraction, repulsion, and vector direction |
| Geometry | Confirm point-charge assumption is reasonable | Large conductors or plates require electric-field methods |
Frequently asked questions
Coulomb’s Law calculates the electrostatic force between two point charges based on charge magnitudes, separation distance, and the permittivity of the surrounding medium.
In SI units, force is in newtons, charge is in coulombs, distance is in meters, and Coulomb’s constant is in N·m\(^2\)/C\(^2\).
Like charges repel and opposite charges attract. If \(q_1q_2\) is positive, the interaction is repulsive. If \(q_1q_2\) is negative, the interaction is attractive.
The simple point-charge form becomes less accurate when charge is distributed over large objects, nearby conductors distort the field, materials polarize, or the charges move rapidly.
Summary and next steps
Coulomb’s Law is the core equation for electrostatic force between point charges. It shows that force increases with charge magnitude and decreases with the square of separation distance.
The main engineering judgment is knowing whether the point-charge assumption is reasonable. For simple isolated charges, Coulomb’s Law is a strong first estimate. For real conductors, capacitors, insulation gaps, or high-voltage hardware, electric-field distribution, dielectric behavior, and geometry must also be checked.
Where to go next
Continue your learning path with these curated next steps.
-
Prerequisite: Engineering Equations Hub
Browse foundational equations across physics, electrical engineering, mechanics, and thermodynamics.
-
Current topic: Coulomb’s Law
Use this page as your reference for electrostatic force, variables, units, assumptions, and checks.
-
Next step: Capacitance Calculator
Move from point-charge force to charge storage, voltage, capacitance, and energy in electrical components.