Key Takeaways
- Core idea: Water treatment processes are arranged steps that remove particles, microorganisms, dissolved chemicals, taste, odor, and other water quality concerns.
- Engineering use: Engineers combine physical, chemical, and biological processes into a treatment train based on source water quality and finished water goals.
- What controls it: Turbidity, organic matter, pathogens, hardness, pH, alkalinity, flow variation, temperature, and residuals handling strongly affect process selection.
- Practical check: A treatment train is only as reliable as its weakest controlled step; poor pretreatment can overload filtration and make disinfection less dependable.
Table of Contents
Introduction
Water treatment processes are the physical, chemical, and biological steps used to improve water quality before it is distributed, reused, discharged, or returned to the environment. In water resources engineering, the goal is not simply to make water look clear; the treatment train must remove the right contaminants, protect downstream systems, and perform reliably under changing source water conditions.
Water Treatment Processes Diagram
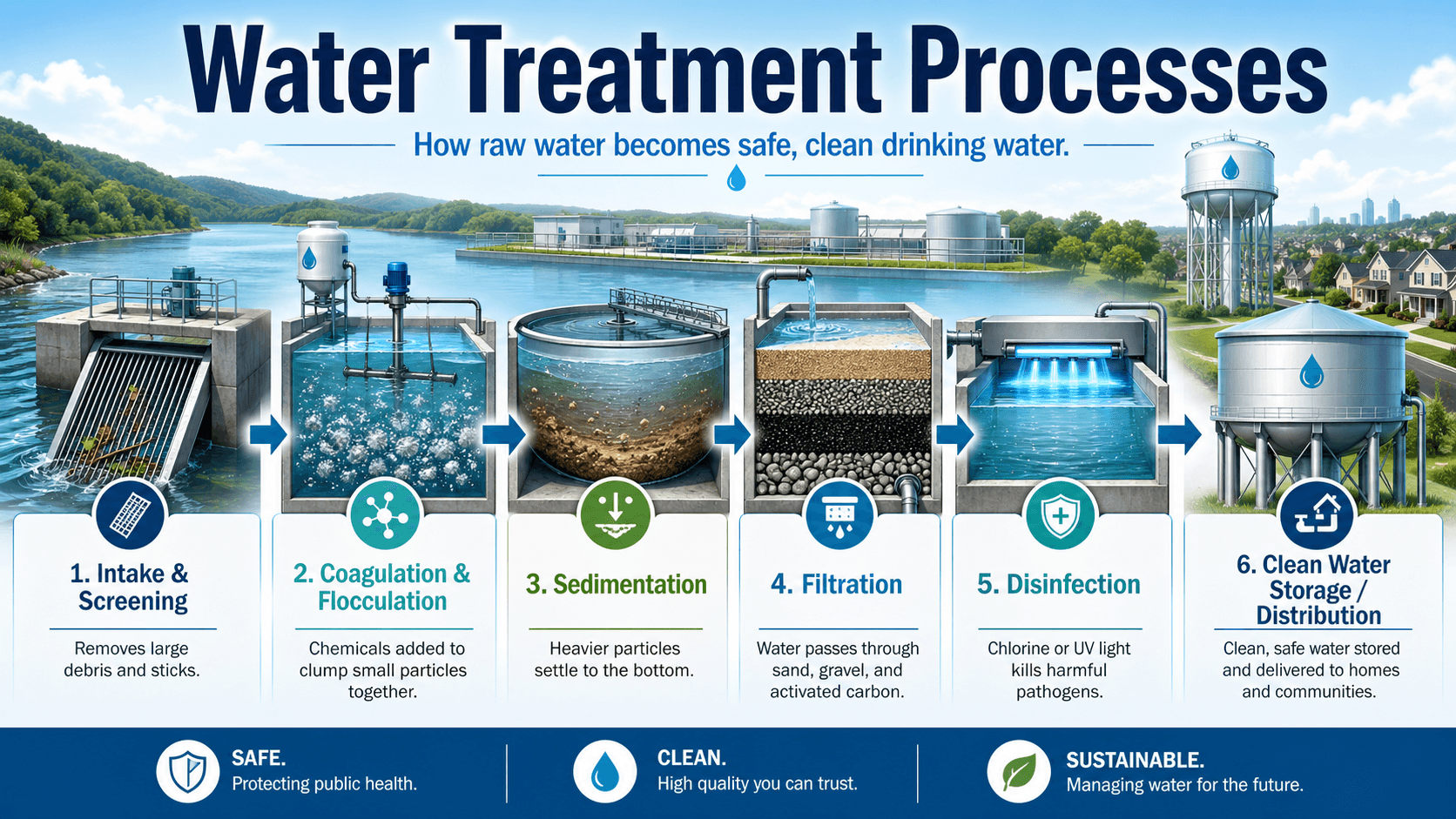
Notice that treatment is not one single unit process. Screening protects equipment, clarification reduces solids, filtration polishes water, and disinfection is most reliable after upstream processes reduce turbidity and organic demand.
What are Water Treatment Processes?
Water treatment processes are engineered steps used to change raw water into water that is suitable for drinking, industrial use, irrigation, reuse, environmental discharge, or another defined purpose. These processes may remove suspended solids, pathogens, nutrients, metals, hardness, dissolved organic matter, taste and odor compounds, salts, or trace contaminants.
The important engineering concept is the treatment train. A treatment train is a sequence of unit processes selected so that each step improves the performance of the next. For example, coagulation and flocculation help small particles form larger flocs, sedimentation removes a large portion of those flocs, filtration removes finer particles, and disinfection inactivates microorganisms after the water has already been clarified.
How Water Treatment Processes Work Together
Water treatment works by matching contaminant type to removal mechanism. Large debris is physically screened. Suspended particles are settled or filtered. Colloids may need chemical destabilization. Microorganisms require disinfection or membrane removal. Dissolved ions, hardness, and some trace chemicals may require adsorption, ion exchange, membranes, softening, or chemical oxidation.
Physical treatment removes what can be separated
Physical processes include screening, grit removal, sedimentation, flotation, filtration, membranes, and sometimes aeration. These steps are often the first barrier because they reduce solids loading, protect pumps and valves, improve clarity, and lower the burden on downstream chemical and disinfection processes.
Chemical treatment changes water chemistry
Chemical processes include coagulation, pH adjustment, softening, oxidation, corrosion control, adsorption, and disinfection. Operators must control dose, mixing energy, contact time, alkalinity, pH, temperature, and reaction demand because the correct chemical process can fail if the water chemistry is outside the working range.
Biological treatment uses microorganisms
Biological treatment is common in wastewater treatment and some advanced water treatment systems. Microorganisms convert or remove organic matter, ammonia, nitrogen, and other biodegradable constituents. The process depends on detention time, dissolved oxygen, temperature, nutrient balance, sludge age, and protection from toxic shocks.
Typical Treatment Train from Source Water to Finished Water
A treatment train is selected after reviewing raw water quality, peak and average flow, seasonal variation, target use, residuals handling, reliability requirements, and monitoring needs. The exact layout differs for drinking water, wastewater, industrial process water, stormwater reuse, desalination, and groundwater treatment, but the engineering logic is similar.
| Treatment stage | Primary purpose | Engineering implication |
|---|---|---|
| Intake and screening | Remove trash, leaves, sticks, rags, grit, and large debris. | Protects pumps, valves, basins, membranes, and downstream equipment from clogging and abrasion. |
| Coagulation and flocculation | Destabilize small particles and grow them into larger settleable flocs. | Requires proper chemical dose, rapid mixing, gentle flocculation, pH control, and jar testing or process monitoring. |
| Sedimentation or clarification | Remove settleable solids and floc before filtration. | Detention time, overflow rate, sludge removal, and short-circuiting strongly affect performance. |
| Filtration | Remove fine particles that remain after clarification. | Filter loading rate, media condition, backwash cycle, turbidity spikes, and headloss must be controlled. |
| Disinfection | Inactivate bacteria, viruses, protozoa, and other pathogens. | Depends on disinfectant type, contact time, pH, temperature, turbidity, and disinfectant demand. |
| Finished water stabilization | Adjust pH, alkalinity, corrosion potential, residual disinfectant, taste, odor, or hardness. | Helps protect distribution systems, storage tanks, plumbing, and end users after treatment is complete. |
Do not evaluate a treatment process only by its individual removal percentage. Check how the upstream and downstream processes interact, because a weak clarification step can increase filter headloss and reduce disinfection reliability.
What Controls Treatment Process Selection?
The right treatment process depends on what must be removed, how variable the source water is, and how the treated water will be used. A groundwater source with iron and hardness needs a different approach than a surface water source with algae, turbidity, pathogens, and storm-driven sediment spikes.
| Factor | Why it matters | Engineering implication |
|---|---|---|
| Turbidity and particle size | Particles shield microorganisms and overload filters. | Drives coagulation, flocculation, sedimentation, filtration, and backwash design. |
| Organic matter | Organic compounds create taste, odor, color, and disinfectant demand. | May require optimized coagulation, activated carbon, oxidation, or precursor control. |
| Pathogen risk | Bacteria, viruses, and protozoa require reliable barriers. | Controls disinfection strategy, contact time, filtration performance, and monitoring intensity. |
| pH and alkalinity | Chemical reactions, corrosion control, coagulation, and disinfection efficiency depend on pH. | May require chemical adjustment before or after major treatment steps. |
| Hardness, iron, manganese, and dissolved ions | Dissolved constituents may not be removed by ordinary settling or filtration. | May require softening, oxidation, ion exchange, membranes, or specialized adsorption media. |
| Temperature and seasonal change | Cold water slows reactions and settling; storms can change turbidity quickly. | Requires flexible dosing, operational response, and safety margin during seasonal extremes. |
Core Physical, Chemical, and Biological Processes
Most treatment systems combine several removal mechanisms. The table below gives a practical engineering view of when each process is commonly used and what it controls.
| Process | Best suited for | Common limitation |
|---|---|---|
| Screening and grit removal | Large debris, grit, rags, and equipment protection. | Does not remove dissolved contaminants or fine particles. |
| Coagulation and flocculation | Colloids, turbidity, color, and some organic matter. | Requires dose control, pH range, adequate mixing, and sludge handling. |
| Sedimentation | Settleable solids and floc removal. | Short-circuiting, density currents, and poor sludge removal reduce performance. |
| Granular media filtration | Fine suspended particles after clarification. | Needs backwashing and can breakthrough if overloaded. |
| Activated carbon | Taste, odor, organic compounds, and some trace contaminants. | Media exhaustion must be monitored and managed. |
| Membrane filtration | Particles, microorganisms, salts, or dissolved constituents depending on membrane type. | Fouling, energy use, concentrate disposal, and pretreatment needs can control feasibility. |
| Biological treatment | Biodegradable organic matter, ammonia, and nutrient removal. | Sensitive to temperature, oxygen, toxicity, loading changes, and solids control. |
| Disinfection | Pathogen inactivation and distribution protection. | Less effective when turbidity, organic demand, or contact time is poorly controlled. |
Water Treatment Process Selection Workflow
A strong process selection workflow starts with the water, not with a preferred technology. Engineers first define the source water and finished water goal, then select the simplest reliable treatment train that can meet the target under normal, peak, and upset conditions.
Define source water quality → identify target contaminants → set finished water goals → select primary removal barriers → check pretreatment needs → evaluate residuals and sludge → test seasonal and peak flow scenarios → confirm monitoring and operator control points.
| Check or decision | What to look for | Why it matters |
|---|---|---|
| Start with contaminant form | Is the concern particulate, dissolved, biological, organic, inorganic, or aesthetic? | The contaminant form determines whether settling, filtration, adsorption, membranes, oxidation, or biological treatment is appropriate. |
| Check source variability | Storm turbidity, algae blooms, seasonal temperature, drought concentration, or groundwater chemistry shifts. | A process that works at average conditions may fail during the exact conditions that control design. |
| Verify pretreatment | Screening, coagulation, pH adjustment, antiscalant, or solids removal before sensitive processes. | Membranes, filters, UV systems, and granular media can fail early when pretreatment is weak. |
| Account for residuals | Sludge, brine, spent media, backwash water, and chemical residuals. | A treatment process is not complete until the waste stream is handled safely and economically. |
| Define control points | Turbidity, pH, disinfectant residual, headloss, flow, dose, contact time, and microbial indicators. | Operators need measurable indicators to keep the process inside its reliable operating window. |
Example: Matching a Treatment Train to Source Water
Consider a surface water source that is generally clear during dry weather but experiences high turbidity after storms, seasonal algae growth, moderate natural organic matter, and pathogen risk. A single filter or disinfectant step would not be a resilient design because the source water changes across the year.
Reasonable treatment logic
Screening removes debris at the intake. Coagulation and flocculation help control storm-driven turbidity and some organic matter. Sedimentation reduces the solids load before filtration. Granular media filtration polishes the water. Disinfection provides the pathogen barrier, and finished water stabilization helps manage corrosion, residual disinfectant, and distribution system water quality.
Engineering interpretation
The design is not only about average removal efficiency. The important question is whether the treatment train remains stable when raw water turbidity increases quickly, algae affect filter run time, or organic matter increases disinfectant demand. That is why process selection includes monitoring, operations, sludge handling, and seasonal response planning.
Engineering Judgment and Field Reality
Treatment processes often look clean in diagrams, but real plants must handle changing water quality, aging equipment, operator response time, sludge buildup, chemical supply issues, instrumentation drift, and maintenance constraints. A process that is technically appropriate can still be a poor fit if it is too complex for the site, too sensitive to upstream variation, or difficult to monitor.
Many water treatment problems are not caused by choosing the wrong process category. They are caused by losing control of the process window: incorrect chemical dose, inadequate mixing, filter breakthrough, short detention time, fouled membranes, poor backwash, or underestimated residuals.
When This Breaks Down
Simplified descriptions of water treatment can break down when they imply that one process solves all water quality problems. In practice, each process has a working range, and water quality can change faster than a fixed treatment train can respond.
- High turbidity events: Storms can overload clarification and filtration if coagulant dose, mixing, and sludge removal are not adjusted.
- Cold water: Lower temperature can slow chemical reactions, floc formation, settling, and biological activity.
- High organic demand: Natural organic matter can consume disinfectant and contribute to taste, odor, color, and disinfection byproduct concerns.
- Membrane fouling: Membranes may remove very fine particles or dissolved constituents, but poor pretreatment can make fouling the controlling design problem.
- Residuals bottlenecks: Sludge, brine, spent media, and backwash water can limit the practicality of a treatment process.
Common Mistakes and Practical Checks
The most common mistakes occur when treatment is treated as a fixed recipe instead of a controlled engineering system. Engineers should check whether the process is appropriate for the raw water, resilient during upset conditions, and manageable by the people operating it.
- Choosing technology before defining the contaminant: The removal mechanism must match the contaminant form and concentration.
- Ignoring seasonal water quality: Average conditions can hide storm events, algae blooms, drought concentration, and temperature effects.
- Underestimating pretreatment: Filters, membranes, UV, and advanced systems usually need upstream protection.
- Forgetting waste streams: Treatment creates sludge, concentrate, spent media, backwash water, or chemical residuals that must be handled.
- Assuming clear water is safe water: Pathogens and dissolved contaminants may remain even when turbidity appears low.
Do not evaluate a treatment train by appearance alone. Clear water can still have microbial, chemical, hardness, corrosion, taste, odor, or dissolved contaminant problems.
Relevant Manuals, Standards, and Design References
Water treatment design is shaped by source water data, public health goals, local requirements, utility standards, operator capability, and recognized engineering references. The exact references depend on the project type and jurisdiction, but the following sources commonly support process selection and review.
- U.S. EPA drinking water guidance: Provides regulatory and technical context for drinking water contaminants, treatment requirements, disinfectants, and public health protection.
- AWWA standards and manuals: Commonly used for water treatment practice, utility operations, filtration, disinfection, distribution water quality, and treatment plant design context.
- WEF wastewater treatment references: Used for wastewater process design, biological treatment, solids handling, nutrient removal, and plant operations.
- State and local design criteria: Define approval requirements, monitoring expectations, treatment credits, residuals handling, and project-specific compliance needs.
Frequently Asked Questions
The main water treatment processes commonly include screening, coagulation, flocculation, sedimentation, filtration, disinfection, chemical adjustment, and sometimes advanced processes such as activated carbon, membranes, ion exchange, biological treatment, or advanced oxidation. The exact sequence depends on raw water quality and the required finished water standard.
Water treatment processes are arranged in a treatment train because each step prepares the water for the next one. Removing grit and large solids protects downstream equipment, clarification reduces particle load, filtration polishes the water, and disinfection is more reliable when turbidity and organic demand have already been controlled.
Engineers choose treatment processes by comparing source water quality, target contaminants, flow range, seasonal variability, residuals handling, energy demand, chemical use, operator skill, monitoring requirements, and regulatory goals. A process that works well for one source may perform poorly if the water has different turbidity, organics, hardness, pathogens, or dissolved contaminants.
Water treatment processes often fail when raw water conditions change faster than the plant can respond, chemical dosing is poorly controlled, filters are overloaded, short-circuiting reduces detention time, disinfection demand is underestimated, or maintenance is deferred. Many failures are process-control problems rather than a single broken piece of equipment.
Summary and Next Steps
Water treatment processes are the engineered barriers that improve water quality by removing solids, pathogens, dissolved contaminants, organic matter, hardness, taste, odor, and other concerns. The best treatment approach depends on the source water, target use, contaminant type, and reliability required.
The key practical lesson is that treatment processes work as a system. Screening, clarification, filtration, disinfection, stabilization, and advanced treatment must be selected and controlled together so the finished water remains reliable during normal operation, seasonal variation, and upset conditions.
Where to go next
Continue your learning path with related Turn2Engineering resources.
-
Water Quality Management
Learn how pollutants, monitoring, standards, and protection strategies shape water treatment goals.
-
Water Resources Modeling
See how engineers simulate water systems, pollutant transport, and treatment-related scenarios.
-
Water Supply Chain
Understand how source water, treatment, storage, distribution, and end users connect in a complete supply system.