Key Takeaways
- Core idea: Thermodynamic equilibrium means a system has no net macroscopic driving force for change.
- Engineering use: Equilibrium states allow engineers to assign meaningful properties such as pressure, temperature, enthalpy, entropy, and internal energy.
- What controls it: Full equilibrium requires thermal, mechanical, chemical, diffusive, and phase balance where those effects apply.
- Practical check: A steady system is not automatically at equilibrium if gradients, flows, reactions, or phase changes are still present.
Table of Contents
Introduction
Thermodynamic equilibrium is the condition where a system has no net macroscopic driving force for change. In engineering thermodynamics, it means the system can be described by stable state properties because temperature, pressure, composition, and phase behavior are balanced where relevant. This matters because most property tables, state equations, and ideal process models assume equilibrium or near-equilibrium states.
Conditions Required for Thermodynamic Equilibrium
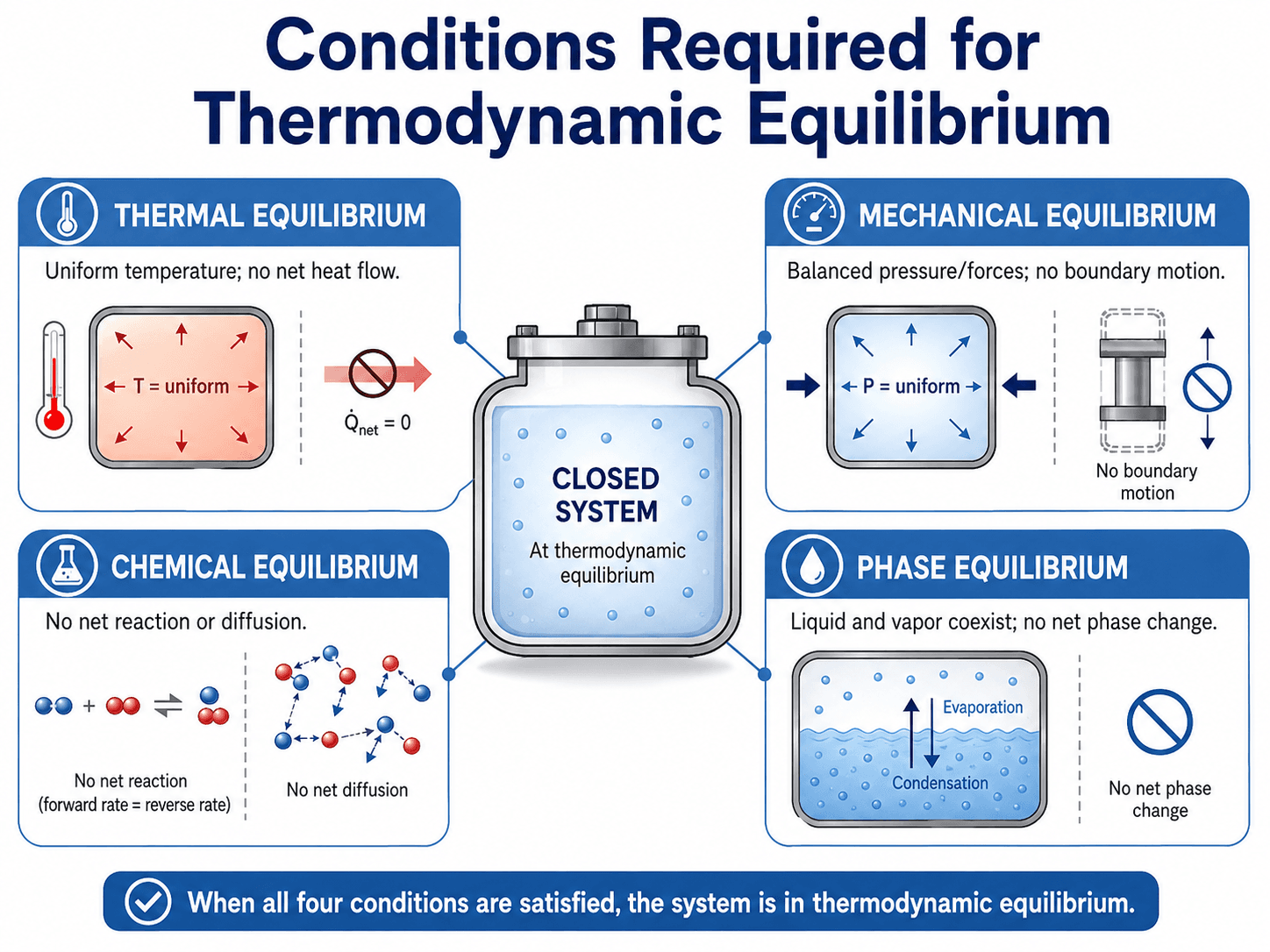
A system does not have to be perfectly isolated to be discussed in terms of equilibrium. The key question is whether net driving forces remain under the chosen boundary and constraints.
What Is Thermodynamic Equilibrium?
Thermodynamic equilibrium is a macroscopic state where the measurable properties of a system are no longer changing because there is no remaining imbalance to drive change. The system may still contain moving molecules, vibrating atoms, and microscopic collisions, but the bulk behavior is stable.
In practical thermodynamics, equilibrium is what allows a system to be assigned a clear state. If a gas has one meaningful temperature, one meaningful pressure, and a stable composition, engineers can use property tables, equations of state, and energy balances with much more confidence. If strong gradients exist inside the same system, the idea of one single state becomes less reliable.
| Related term | What it means | What it checks |
|---|---|---|
| Thermal equilibrium | Temperature is balanced | No net heat-transfer driver from temperature difference |
| Mechanical equilibrium | Pressure or force is balanced | No unbalanced boundary motion, acceleration, or pressure relaxation |
| Chemical equilibrium | Reaction tendency is balanced | No net change in composition due to reaction |
| Phase equilibrium | Phase change tendency is balanced | No net boiling, condensation, melting, freezing, or flashing |
| Thermodynamic equilibrium | All relevant equilibrium conditions are satisfied | No remaining macroscopic driving force for change |
Equilibrium is less about “nothing happening” and more about “nothing changing at the macroscopic level.” That distinction matters when analyzing piston-cylinder devices, sealed tanks, refrigerant states, heat engines, and idealized thermodynamic cycles.
The Main Equilibrium Conditions
A system reaches full thermodynamic equilibrium only when all relevant forms of equilibrium are satisfied at the same time. Some systems involve all conditions. Others, such as a simple sealed gas with no chemical reaction or phase change, may only require the applicable conditions.
| Equilibrium condition | Balanced quantity | What it means in engineering terms |
|---|---|---|
| Thermal equilibrium | Temperature | No net heat transfer is driven by a temperature difference within the system or across an interacting boundary. |
| Mechanical equilibrium | Pressure, force, or stress | No unbalanced pressure force causes bulk acceleration, piston motion, sudden expansion, or boundary movement. |
| Chemical equilibrium | Chemical potential and reaction tendency | No net reaction changes the system composition at the macroscopic scale. |
| Diffusive equilibrium | Chemical potential and species distribution | No net species migration occurs because concentration or chemical-potential gradients have been balanced. |
| Phase equilibrium | Phase potential at the given conditions | Liquid, vapor, or solid phases coexist without a net phase change in one direction. |
Thermal equilibrium
Thermal equilibrium means temperature has balanced out enough that there is no net heat transfer caused by temperature difference. Two metal blocks at the same temperature are in thermal equilibrium with each other, but that does not automatically prove chemical, phase, or mechanical equilibrium.
Mechanical equilibrium
Mechanical equilibrium means forces and pressures are balanced. A piston-cylinder at rest may be mechanically balanced if the gas pressure, piston weight, atmospheric pressure, and any applied load are in balance. If the piston is accelerating, the system is not mechanically at equilibrium.
Chemical, diffusive, and phase equilibrium
Chemical equilibrium matters when reactions can change composition. Diffusive equilibrium is often grouped with chemical equilibrium because it depends on chemical potential balance, but it is useful to name separately when mass transfer or species gradients are the main issue. Phase equilibrium matters when more than one phase is present, such as liquid water and water vapor coexisting at saturation conditions.
Which conditions matter by system type?
Not every thermodynamic problem requires the same checks. The system boundary, phase behavior, and physical process determine which equilibrium conditions matter most.
| System type | Equilibrium checks that usually matter most | Practical interpretation |
|---|---|---|
| Sealed ideal gas tank | Thermal and mechanical equilibrium | Uniform temperature and pressure usually make a single state reasonable. |
| Piston-cylinder | Thermal and mechanical equilibrium; quasi-equilibrium if moving | The piston must move slowly enough for pressure and temperature to remain well defined. |
| Liquid-vapor mixture | Thermal, mechanical, and phase equilibrium | Saturation behavior controls whether liquid and vapor coexist without net phase change. |
| Reacting gas mixture | Thermal, mechanical, chemical, and diffusive equilibrium | Composition must stop changing before a full equilibrium state is reasonable. |
| Heat exchanger | Usually not full thermodynamic equilibrium | It may be steady, but temperature differences and heat flow are required for operation. |
| Combustion chamber | Often non-equilibrium locally | Temperature, pressure, composition, and reaction progress can vary strongly through the chamber. |
Thermal Equilibrium vs Thermodynamic Equilibrium
Thermal equilibrium is only one part of thermodynamic equilibrium. It means temperature is balanced and no net heat transfer is driven by a temperature difference. Thermodynamic equilibrium is broader because it also requires mechanical, chemical, diffusive, and phase balance where those effects are relevant.
A system can be thermally balanced but still not be in full thermodynamic equilibrium. For example, a gas mixture may have a uniform temperature while a chemical reaction is still progressing. A tank may have uniform temperature but still have vapor condensing into liquid. A piston-cylinder may have uniform temperature but still be mechanically unbalanced if the piston is moving.
Thermal equilibrium asks one question: “Is temperature balanced?” Thermodynamic equilibrium asks the larger engineering question: “Are all relevant driving forces balanced?”
How a System Reaches Thermodynamic Equilibrium
A system approaches equilibrium by reducing gradients. Temperature differences drive heat transfer, pressure differences drive expansion or compression, concentration differences drive diffusion, and chemical potential differences drive reactions or phase changes. The process continues until the relevant driving force is no longer present at the macroscopic scale.
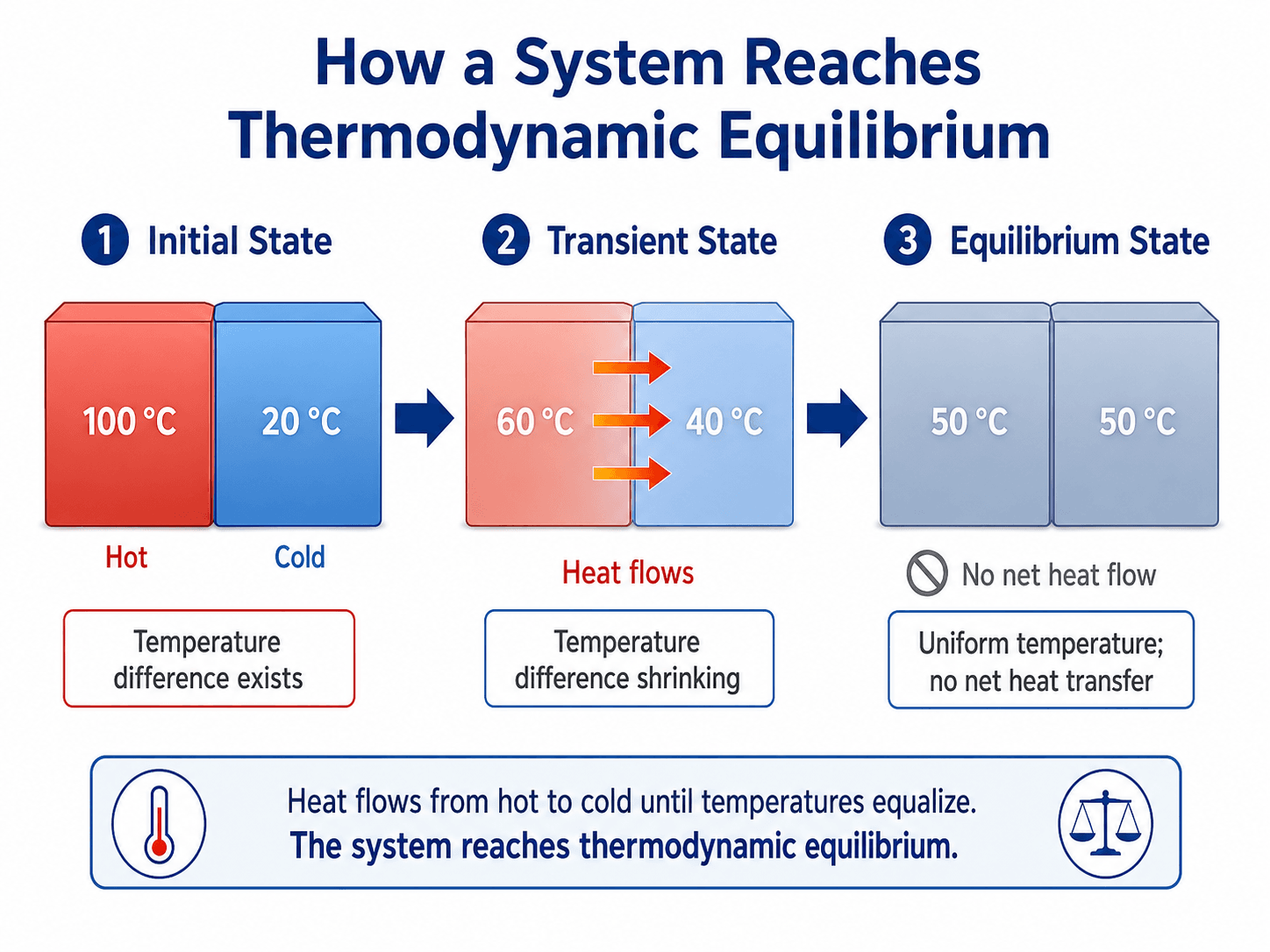
Equilibrium is the final macroscopic condition
In the block example, the initial state has a clear temperature difference. During the transient period, heat transfer reduces that difference. At the final equilibrium state, the blocks may still contain molecular motion, but the measured temperature is uniform and there is no net heat transfer between them.
Different gradients relax at different rates
Temperature, pressure, concentration, and phase changes do not always settle at the same speed. A gas may reach pressure balance quickly, while chemical composition or phase distribution may take longer. In real engineering systems, the slowest relevant relaxation process often controls whether the equilibrium assumption is reasonable.
Thermodynamic Equilibrium vs Steady State
Steady state and thermodynamic equilibrium are often confused. A steady-state system has properties at each location that do not change with time. A system at thermodynamic equilibrium has no net driving force for macroscopic change. The difference is important because many steady systems still have continuous energy or mass flow.
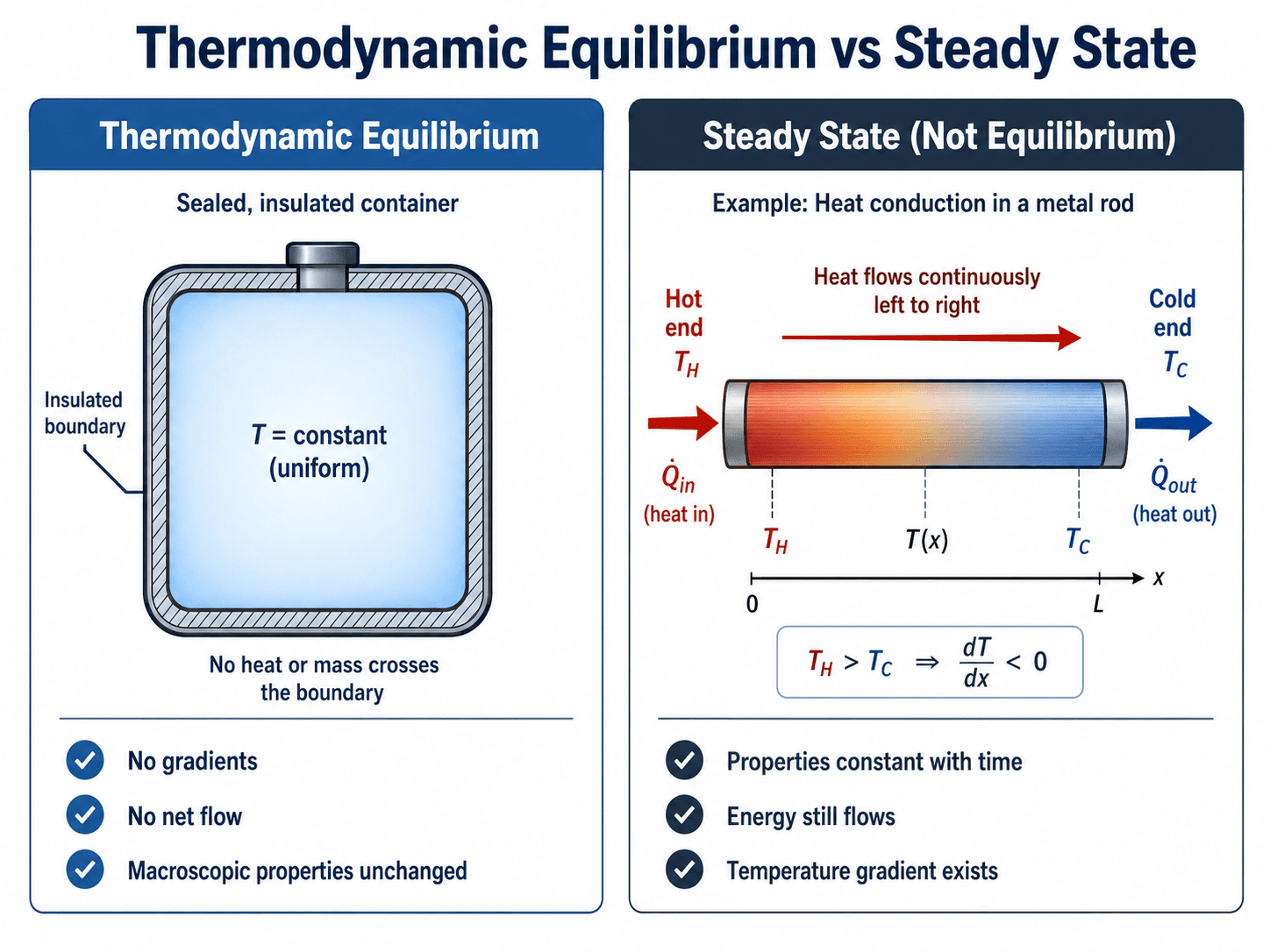
Can a system be steady but not at equilibrium?
Yes. A heat exchanger, turbine, compressor, boiler, nozzle, or cooling fin can operate at steady state while still having heat transfer, flow, pressure drop, or temperature gradients. In those cases, the operating condition may be steady, but the device as a whole is not in thermodynamic equilibrium.
| Condition | Do properties change with time? | Can flow or gradients exist? | Example |
|---|---|---|---|
| Thermodynamic equilibrium | No macroscopic change | No relevant net flow or gradient | Sealed gas at uniform temperature and pressure |
| Steady state | No change at a fixed location | Yes | Operating heat exchanger or heat-conducting rod |
| Transient process | Yes | Often yes | Hot tank cooling toward room temperature |
| Non-equilibrium process | May or may not | Usually yes | Rapid expansion, shock, mixing, combustion, or diffusion |
Why Equilibrium Matters in Engineering Thermodynamics
Engineers use equilibrium because it makes thermodynamic states measurable, repeatable, and useful for calculations. A state point on a property table, a P-v diagram, a T-s diagram, or a pressure-enthalpy chart assumes the working fluid can be treated as being at a defined state.
- Property lookup: Steam tables, refrigerant tables, and gas-property charts are based on equilibrium state properties.
- Energy balances: The First Law of Thermodynamics becomes easier to apply when inlet, outlet, and system states are well defined.
- Entropy and irreversibility: The Second Law of Thermodynamics explains why isolated systems tend toward equilibrium and why real processes generate entropy.
- Cycle modeling: Ideal engines, refrigerators, and heat pumps are commonly modeled as sequences of equilibrium or near-equilibrium states.
- System boundaries: Equilibrium checks depend on whether the model is a closed system, open system, or isolated system.
Before using a single pressure and temperature to represent a system, ask whether one bulk state is physically meaningful. If the system has strong internal gradients, rapid motion, active reaction, or phase separation, the equilibrium assumption may need to be localized, delayed, or replaced with a more detailed model.
State Properties, Entropy, and Equilibrium Criteria
Thermodynamic properties such as internal energy, enthalpy, entropy, pressure, temperature, and specific volume are state properties. They describe the condition of a system, not the path used to reach that condition. This is why equilibrium is so important: a state property is most useful when the state itself is well defined.
For an isolated system, spontaneous processes move in the direction of nondecreasing entropy. Under fixed constraints, the equilibrium state corresponds to no remaining macroscopic driving force and maximum entropy relative to the allowed states. Other constraints use different thermodynamic potentials.
| Constraint | Common equilibrium criterion | Engineering meaning |
|---|---|---|
| Isolated system | Entropy tends toward a maximum | The system evolves until no allowed spontaneous change remains. |
| Constant temperature and volume | Helmholtz free energy tends toward a minimum | Useful when temperature and volume are controlled. |
| Constant temperature and pressure | Gibbs free energy tends toward a minimum | Important for phase equilibrium, chemical equilibrium, and many reacting systems. |
This connects thermodynamic equilibrium directly to entropy, irreversibility, and the direction of real processes. For most introductory engineering calculations, the main practical point is that equilibrium lets engineers define reliable state properties.
- T Temperature; uniform temperature is required for thermal equilibrium.
- P Pressure; balanced pressure or force is required for mechanical equilibrium.
- S Entropy; useful for understanding why isolated systems tend toward equilibrium.
- G Gibbs free energy; useful for equilibrium at constant temperature and pressure.
- h, u Enthalpy and internal energy; commonly read from equilibrium property tables or charts.
Examples of Thermodynamic Equilibrium and Non-Equilibrium Systems
The easiest way to understand thermodynamic equilibrium is to compare systems with no remaining driving force against systems that still have gradients, flow, reaction, or ongoing change.
Examples of thermodynamic equilibrium
- Sealed rigid tank left undisturbed: Temperature and pressure can become uniform after transients disappear.
- Ice and liquid water at the melting point: Solid and liquid can coexist with no net melting or freezing under fixed conditions.
- Piston-cylinder at rest: A gas can be mechanically balanced when pressure forces and external loads are in balance.
Examples of non-equilibrium systems
- Operating heat exchanger: It may be steady, but heat transfer and temperature differences are required for operation.
- Rapid gas expansion after a valve opens: Large pressure gradients, turbulence, and transient motion prevent a single uniform state.
- Combustion chamber during reaction: Temperature, composition, and reaction rates vary strongly through space and time.
| System or process | Equilibrium status | Why |
|---|---|---|
| Sealed rigid tank left undisturbed | Can reach equilibrium | Temperature and pressure can become uniform after transients disappear. |
| Ice and liquid water at the melting point | Can be phase equilibrium | Solid and liquid can coexist with no net melting or freezing under fixed conditions. |
| Operating heat exchanger | Not full equilibrium | It may be steady, but heat transfer and temperature differences are required for operation. |
| Rapid gas expansion after a valve opens | Non-equilibrium | Large pressure gradients, turbulence, and transient motion prevent a single uniform state. |
| Combustion chamber during reaction | Usually non-equilibrium locally | Temperature, composition, and reaction rates vary strongly through space and time. |
Many engineering devices are intentionally not at equilibrium while operating. Turbines, compressors, heat exchangers, nozzles, and boilers depend on flow, heat transfer, pressure difference, or phase change. Engineers usually analyze these devices using inlet and outlet states rather than pretending the entire device is at full equilibrium.
Equilibrium Assumption Checklist
Use this checklist before treating a system as being at thermodynamic equilibrium or before using a single state point to represent the whole system.
First identify the system boundary. Then check for temperature gradients, pressure imbalance, composition changes, diffusion, phase changes, and time dependence. If any relevant gradient or process is still active, decide whether the system can be divided into local equilibrium regions or whether a transient/non-equilibrium model is needed.
| Check or decision | What to look for | Why it matters |
|---|---|---|
| Can one temperature represent the system? | Small internal temperature variation, no strong hot/cold zones, no active heat-transfer gradient. | A single temperature is needed for a single equilibrium state point. |
| Can one pressure represent the system? | No rapid expansion, pressure wave, accelerating piston, shock, or strong pressure gradient. | Mechanical equilibrium requires balanced forces and pressure behavior. |
| Is composition stable? | No continuing reaction, diffusion, mixing front, or species separation changing bulk composition. | Chemical and diffusive equilibrium are required when composition affects properties or heat release. |
| Is phase behavior balanced? | No net boiling, condensation, melting, freezing, flashing, or phase separation still progressing. | Phase equilibrium controls saturated liquid-vapor states and refrigerant behavior. |
| Is the process slow enough? | Process time is long compared with the time required for internal relaxation. | Slow processes can often be modeled as quasi-equilibrium paths. |
Worked Example: Is This System at Thermodynamic Equilibrium?
Consider a sealed rigid tank containing liquid water and water vapor. The tank has been left undisturbed for a long time, and measurements show a uniform temperature and pressure. Can the tank be treated as being at thermodynamic equilibrium?
Step 1: Check the boundary and time behavior
The tank is sealed and rigid, so no mass leaves the system and there is no moving boundary. If the readings are stable with time and no heating or cooling transient is occurring, the closed system assumption is reasonable.
Step 2: Check the relevant equilibrium conditions
Uniform temperature supports thermal equilibrium. Uniform pressure and a fixed boundary support mechanical equilibrium. Because liquid and vapor are both present, phase equilibrium must also be checked. The measured temperature and pressure should be consistent with saturation behavior for water.
Step 3: Interpret the result
If the liquid-vapor mixture is at saturation conditions and there is no visible net boiling, condensation, stratification, or continuing transient, the tank can usually be treated as thermodynamic equilibrium for property lookup and energy-balance calculations. If vapor is still condensing, the wall is being heated, or the temperature varies by location, the system is not yet fully equilibrated.
For a two-phase system, equal temperature alone is not enough. Confirm that pressure and temperature match the expected saturation relationship before treating liquid and vapor as being in phase equilibrium.
Quasi-Equilibrium and Local Thermodynamic Equilibrium
A quasi-equilibrium process is an idealized process that happens slowly enough that the system remains very close to equilibrium at each step. This does not mean the process is perfectly real. It means the departure from equilibrium is small enough that a sequence of state points can represent the path.
Why quasi-equilibrium is useful
Piston-cylinder compression is a common example. If the piston moves slowly, the gas has time to redistribute pressure and temperature internally, so each intermediate condition can be approximated as a defined state. That lets engineers draw meaningful process paths on property diagrams.
Why it is still an approximation
Real processes require finite differences to make anything happen. Heat transfer needs a temperature difference. Flow needs a pressure difference. Diffusion needs a concentration difference. Quasi-equilibrium is useful because it keeps those differences small enough for a simple thermodynamic model to remain accurate.
Local thermodynamic equilibrium
In some real systems, engineers use local thermodynamic equilibrium. The entire device may not be at equilibrium, but a small region is assumed close enough to equilibrium that properties such as temperature, pressure, and composition can be assigned locally. This idea is useful in heat transfer, combustion, compressible flow, two-phase flow, and other systems with spatial gradients.
Engineering Judgment and Field Reality
Real systems rarely meet perfect thermodynamic equilibrium everywhere. A steam line may have local condensation, a refrigerant coil may have two-phase zones, a compressor discharge may be hot and turbulent, and a tank may stratify by temperature. Engineers often use equilibrium assumptions because they are practical, but the assumptions must match the scale and purpose of the analysis.
A measured pressure or temperature can be misleading if the sensor is placed in a non-representative location. In real equipment, the question is often not “is the entire system perfectly at equilibrium?” but “is the measured region close enough to equilibrium for the calculation being performed?”
This is especially important in systems involving heat transfer, two-phase flow, fast transients, combustion, throttling, or mixing. In those cases, engineers may use local equilibrium assumptions, control-volume balances, empirical correlations, or detailed simulations instead of a single whole-system equilibrium state.
When This Breaks Down
The equilibrium assumption breaks down when gradients or rates are too large for the system to be represented by one stable macroscopic state. The concept is still useful, but the model must be adjusted.
- Rapid transients: Sudden valve openings, shock waves, and fast piston motion can create pressure and temperature gradients that invalidate a single-state model.
- Strong heat transfer gradients: Walls, rods, coils, and heat exchangers can be steady but not at equilibrium because heat continues to flow.
- Active chemical reaction: Combustion and reacting mixtures may have changing composition, temperature, and reaction progress.
- Two-phase non-equilibrium: Flashing, boiling, condensation, and droplet formation may not instantly match saturation-equilibrium assumptions.
- Poor mixing or stratification: Large tanks can have layers of different temperature or concentration even when the system appears still.
Common Mistakes and Practical Checks
Most mistakes come from using the word equilibrium too loosely. Equal temperature, steady operation, or a constant instrument reading may be useful information, but none of those alone proves full thermodynamic equilibrium.
- Confusing thermal equilibrium with full thermodynamic equilibrium: Equal temperature does not guarantee pressure, composition, diffusion, or phase balance.
- Assuming steady state means equilibrium: Steady heat exchangers, turbines, pumps, and flow devices are usually not at full equilibrium while operating.
- Ignoring system boundaries: A closed system, open system, and isolated system can have very different equilibrium interpretations.
- Using property tables blindly: Tables represent equilibrium properties, so the selected state should match the actual fluid condition closely enough.
- Forgetting local equilibrium: A whole device may not be at equilibrium, but small regions may still be modeled using local equilibrium if gradients are manageable.
Do not call a system “at equilibrium” simply because nothing obvious is moving. If heat, mass, reaction, diffusion, or phase change is still occurring, the system may be static-looking but not thermodynamically balanced.
Useful References and Engineering Context
Thermodynamic equilibrium is usually taught through engineering thermodynamics, statistical mechanics, heat transfer, and materials science references. For engineering learners, the most useful reference is one that connects state properties, equilibrium states, and quasi-equilibrium process modeling.
- MIT OpenCourseWare: MIT Unified Engineering Thermodynamics Chapter 2 explains thermodynamic state properties and why equilibrium matters when defining pressure, temperature, density, internal energy, enthalpy, and entropy.
- Project-specific criteria: Equipment models, laboratory assumptions, and design calculations may define how close to equilibrium a state must be for the required level of accuracy.
- Engineering use: Engineers use equilibrium references to justify property-table lookup, state-point selection, quasi-equilibrium paths, and the limits of simplified thermodynamic models.
Frequently Asked Questions
No. Thermal equilibrium only means temperature is balanced so there is no net heat transfer due to a temperature difference. Thermodynamic equilibrium is broader and also requires mechanical, chemical, diffusive, and phase equilibrium where those effects apply.
The main conditions are thermal equilibrium, mechanical equilibrium, chemical equilibrium, and phase equilibrium where applicable. Diffusive equilibrium is also important when mass transfer or concentration gradients exist, because species movement is driven by chemical potential differences.
Yes. A system can have properties that do not change with time while heat, work, or mass still flows through it. A metal rod with constant heat conduction is steady, but it is not in thermodynamic equilibrium because a temperature gradient and continuous heat flow still exist.
Thermodynamic equilibrium matters because state properties such as pressure, temperature, internal energy, enthalpy, and entropy are most clearly defined for equilibrium states. Engineers often analyze real processes by connecting equilibrium or near-equilibrium states.
No. Molecules continue moving at the microscopic level. Thermodynamic equilibrium means there is no net macroscopic change, no unbalanced driving force, and no continuing bulk process such as net heat transfer, pressure relaxation, diffusion, reaction, or phase change.
Summary and Next Steps
Thermodynamic equilibrium is the condition where a system has no net macroscopic driving force for change. It requires more than equal temperature: a system must also satisfy mechanical, chemical, diffusive, and phase equilibrium where those effects are relevant.
In engineering thermodynamics, equilibrium is the foundation for meaningful state properties, property-table lookup, process diagrams, entropy analysis, and idealized cycle modeling. The practical skill is knowing when a real system is close enough to equilibrium and when gradients, transients, reactions, or phase changes require a more careful model.
Where to go next
Continue your learning path with related Turn2Engineering resources.
-
Entropy
Learn how entropy explains irreversibility, process direction, and why isolated systems move toward equilibrium.
-
First Law of Thermodynamics
Review how heat, work, and internal energy are balanced between equilibrium state points.
-
Closed Systems
See how equilibrium assumptions are applied to sealed tanks, piston-cylinder devices, and fixed-mass energy balances.