Key Takeaways
- Core idea: An adiabatic process has zero heat transfer across the system boundary, so \( Q = 0 \).
- Engineering use: Engineers use adiabatic assumptions to analyze engines, compressors, turbines, nozzles, gas expansion, and ideal thermodynamic cycles.
- What controls it: Pressure, volume, temperature, the heat capacity ratio \( \gamma \), process speed, insulation, and irreversibility determine how well the ideal model fits.
- Practical check: Adiabatic does not mean constant temperature; compression usually heats the gas, while expansion usually cools it.
Table of Contents
Introduction
An adiabatic process is a thermodynamic process where no heat is transferred into or out of a system. Since \( Q = 0 \), changes in internal energy come from work instead of heat. In mechanical engineering, this concept explains why rapid compression heats a gas and rapid expansion cools it.
Adiabatic Process Diagram
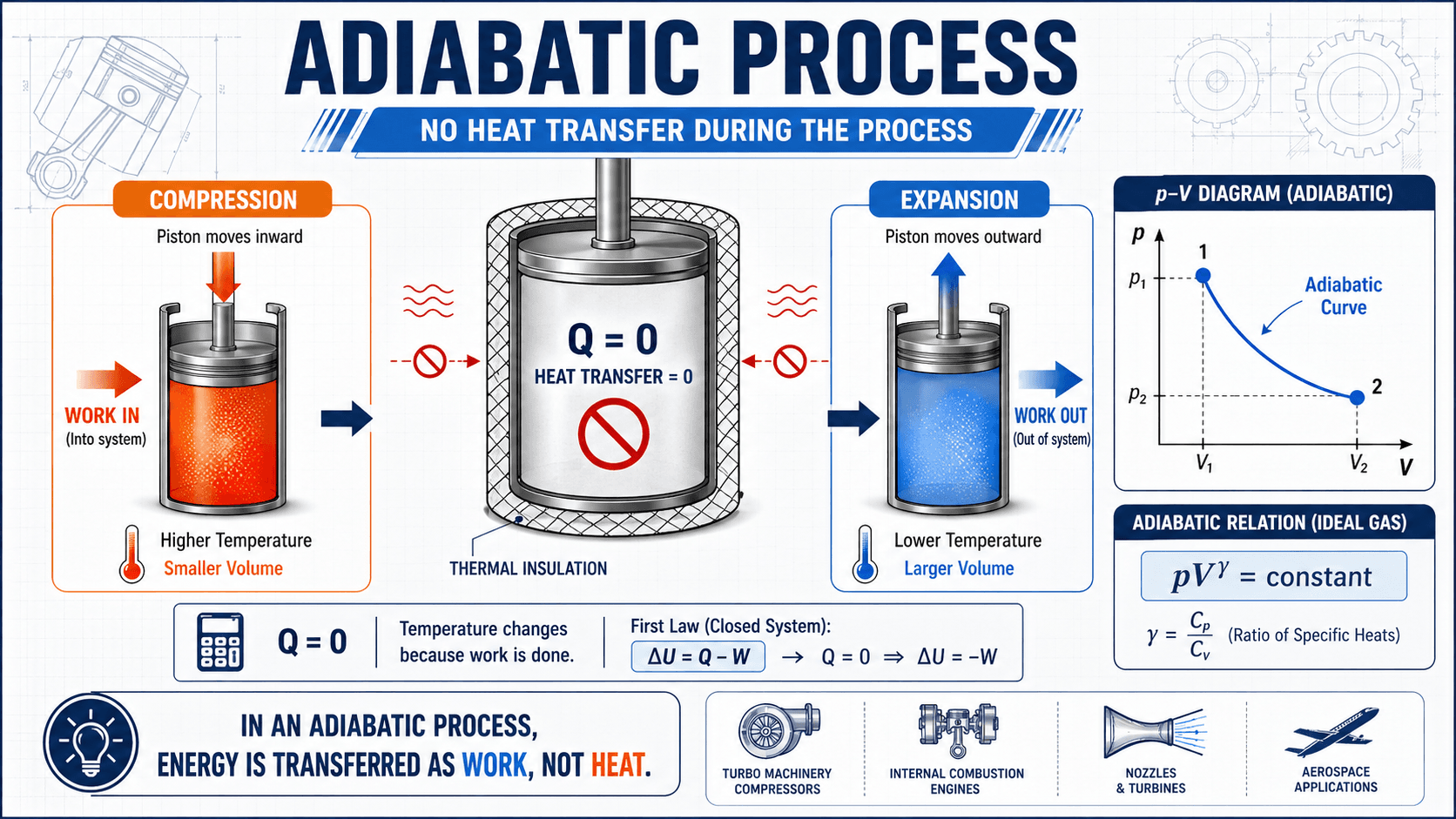
Notice the difference between heat and work first. The process is called adiabatic because heat transfer is zero, not because nothing changes. Pressure, volume, temperature, and internal energy can all change when the system is compressed or allowed to expand.
What is an Adiabatic Process?
An adiabatic process describes a thermodynamic path where heat does not cross the system boundary. The boundary may be well insulated, or the process may happen so quickly that there is not enough time for meaningful heat transfer. The defining condition is:
This does not mean the system has no energy interaction. Work can still be done by the system or on the system. For an ideal gas, that work changes the gas internal energy, which usually changes temperature. A piston compressing air rapidly is a classic example: heat has little time to escape, so the work input mostly shows up as a temperature rise.
Adiabatic is a boundary condition on heat transfer, not a statement that temperature stays constant. If the gas temperature stays constant, the process is isothermal, not adiabatic.
How an Adiabatic Process Works
The first law of thermodynamics connects heat, work, and internal energy. In an adiabatic process, the heat term drops out, so the energy balance depends on the work interaction and the chosen sign convention. For a closed system where \( W \) is work done by the system, a common form is:
Adiabatic Compression
During adiabatic compression, work is done on the gas. The gas volume decreases, pressure rises, and internal energy increases. For an ideal gas, increasing internal energy usually means increasing temperature. This is why compressor discharge air is hot and why diesel engines can heat air enough to support ignition.
Adiabatic Expansion
During adiabatic expansion, the gas does work on its surroundings. Energy leaves the gas as work, internal energy decreases, and the gas temperature falls. This behavior is important in turbines, nozzles, expanding gas flows, and atmospheric air parcels that cool as they rise.
Why the Process Can Be Treated as No-Heat-Transfer
Real equipment is rarely perfectly insulated. Engineers still use adiabatic models when heat transfer is small compared with work and internal energy change. A fast piston stroke, a short turbine passage, or a rapid nozzle expansion may be close enough to adiabatic for a first-pass calculation, even though small heat losses still exist.
Where Adiabatic Processes Are Used in Engineering
Adiabatic analysis is common in mechanical engineering because many energy systems involve rapid compression or expansion. The model helps engineers estimate temperature changes, pressure ratios, work requirements, efficiency limits, and equipment discharge conditions before adding detailed heat-transfer and loss models.
- Compressors: Estimate discharge temperature and compression work when heat removal is limited during the compression stage.
- Turbines: Approximate expansion through blades and compare ideal shaft work against real turbine performance.
- Nozzles and diffusers: Analyze high-speed gas flow where kinetic energy, pressure, and temperature change quickly.
- Internal combustion engines: Model compression and expansion strokes in ideal Otto and Diesel cycle analysis.
- Atmospheric processes: Understand air parcel warming during descent and cooling during ascent.
Before assuming a process is adiabatic, ask whether the process is fast, insulated, or dominated by work transfer. If heat transfer is comparable to the work term, the adiabatic assumption may give the wrong temperature or power estimate.
Key Factors That Control an Adiabatic Process
The ideal adiabatic model depends on both thermodynamic properties and physical conditions. In classroom problems, the process is often reversible and the gas is often ideal. In real equipment, friction, leakage, heat loss, pressure drop, and nonuniform flow can move the result away from the ideal relationship.
| Factor | Why it matters | Engineering implication |
|---|---|---|
| Heat transfer across the boundary | Adiabatic analysis requires \( Q = 0 \), or heat transfer small enough to neglect. | Poor insulation or long process time can make the actual path partly non-adiabatic. |
| Heat capacity ratio \( \gamma \) | The ideal-gas pressure-volume relationship depends on \( \gamma = C_p/C_v \). | Air is often approximated as \( \gamma \approx 1.4 \), but high temperatures and real-gas effects can change the value. |
| Compression or expansion ratio | Larger volume or pressure changes create larger temperature changes. | High pressure ratios can create hot compressor discharge temperatures or cold expansion outlet temperatures. |
| Reversibility | Friction, turbulence, shocks, and mixing generate entropy even if heat transfer is zero. | A real adiabatic process is not automatically isentropic. |
| Process speed | Fast processes often allow less time for heat transfer. | Rapid piston strokes and nozzle flows are more likely to be treated as adiabatic than slow tank heating or cooling processes. |
Adiabatic Process Equations
For a reversible adiabatic process involving an ideal gas with constant specific heats, the common relationships connect pressure, volume, and temperature. These equations are powerful, but they rely on ideal assumptions that must be checked before use.
The most recognized form is \( PV^{\gamma} = \text{constant} \). It shows that pressure changes more sharply with volume in a reversible adiabatic process than in an isothermal ideal-gas process. That is why the adiabatic curve is usually steeper on a pressure-volume diagram.
- P Absolute pressure, commonly in pascals, kilopascals, bar, or psi absolute.
- V Volume for a closed-system ideal gas problem, commonly in cubic meters or cubic feet.
- T Absolute temperature, usually in kelvin or degrees Rankine for thermodynamic equations.
- γ Heat capacity ratio \( C_p/C_v \); about 1.4 for air under many moderate-temperature ideal-gas assumptions.
Work for a Reversible Adiabatic Ideal Gas Process
For a closed system with a reversible adiabatic ideal-gas path, work can be related to the endpoint states. With the convention that positive work is work done by the gas:
If the gas expands, work done by the gas is typically positive. If the gas is compressed, this expression may produce a negative value because work is being done on the gas. Always confirm the sign convention used by the textbook, software, or engineering calculation.
Adiabatic Process Sanity Check
Use this checklist before applying adiabatic equations to a real or textbook thermodynamics problem. It helps separate the true adiabatic assumption from other assumptions that are often bundled with it.
Start with the boundary: is heat transfer negligible? Then identify the system type, sign convention, gas model, reversibility assumption, and known state points. Only after those checks should you select \( Q = 0 \), \( PV^\gamma = \text{constant} \), or a steady-flow energy balance.
| Check or decision | What to look for | Why it matters |
|---|---|---|
| Is heat transfer actually negligible? | Fast process, insulated boundary, or heat transfer small compared with work. | This is the defining adiabatic condition; without it, \( Q = 0 \) is not justified. |
| Is the process reversible? | Low friction, no shock, no strong turbulence, and no uncontrolled mixing. | \( PV^\gamma = \text{constant} \) requires a reversible adiabatic ideal-gas path, not just \( Q = 0 \). |
| Are temperatures absolute? | Use kelvin or Rankine, not Celsius or Fahrenheit, in gas-law ratios. | Using relative temperature scales can produce physically wrong ratios. |
| Is \( \gamma \) appropriate? | Check gas composition and temperature range. | The result is sensitive to \( \gamma \), especially for large compression or expansion ratios. |
| Does the result make physical sense? | Compression should generally raise temperature; expansion should generally lower it. | A result showing the opposite often indicates a sign, ratio, or unit error. |
Worked Example: Adiabatic Compression of Air
Suppose air is compressed rapidly in a piston-cylinder from \( V_1 = 1.0 \, \text{m}^3 \) to \( V_2 = 0.25 \, \text{m}^3 \). The initial temperature is \( T_1 = 300 \, \text{K} \), and air is modeled as an ideal gas with \( \gamma = 1.4 \). If the compression is treated as reversible and adiabatic, the final temperature is estimated from:
Engineering Interpretation
The final temperature is much higher because work was added to the gas and little heat escaped during the compression. This is why compressors require thermal management, why discharge piping can become hot, and why ideal compression models are useful for estimating upper-bound temperature trends.
What the Example Assumes
The calculation assumes ideal-gas behavior, constant \( \gamma \), a reversible path, and no heat transfer. A real compressor may have heat loss, leakage, pressure drop, mechanical losses, valve losses, and nonuniform flow. Those effects usually require efficiency corrections or a more detailed model.
Adiabatic vs Isothermal Process
Adiabatic and isothermal processes are often confused because both can describe gas compression or expansion. The difference is the controlled condition: adiabatic controls heat transfer, while isothermal controls temperature.
| Comparison | Adiabatic Process | Isothermal Process |
|---|---|---|
| Defining condition | No heat transfer: \( Q = 0 \) | Constant temperature: \( \Delta T = 0 \) |
| Temperature behavior | Usually changes during compression or expansion | Held constant by heat transfer |
| Typical ideal-gas relation | \( PV^\gamma = \text{constant} \) | \( PV = \text{constant} \) |
| P-V curve | Generally steeper for an ideal gas | Generally less steep for the same starting state |
| Engineering example | Rapid compressor stroke or turbine expansion | Slow compression with enough heat exchange to hold temperature nearly constant |
A useful mental check is this: if temperature is constant, heat transfer usually must occur to remove or supply energy. If heat transfer is zero, temperature is usually free to change as work changes the system energy.
Engineering Judgment and Field Reality
In real mechanical systems, adiabatic behavior is usually an approximation. A compressor casing conducts heat, a turbine blade exchanges heat with hot gas, a nozzle may have wall losses, and an engine cylinder loses heat to surrounding metal. The question is not whether heat transfer is exactly zero; the question is whether heat transfer is small enough for the purpose of the analysis.
Engineers often start with an adiabatic model to understand the main pressure-temperature-work relationship, then add real-world corrections such as isentropic efficiency, polytropic efficiency, heat loss, leakage, pressure drop, or measured performance data. This sequence keeps early calculations simple without pretending the equipment is ideal.
The adiabatic assumption is most useful when it is treated as a modeling choice, not a physical guarantee. If a slow process has large surface area, high temperature difference, or long residence time, heat transfer may dominate enough that the adiabatic model becomes misleading.
When This Breaks Down
The simple adiabatic ideal-gas equations stop being reliable when the assumptions behind them no longer match the physical process. This is especially important when moving from textbook piston problems to real compressors, turbines, nozzles, and high-speed flow systems.
- Significant heat transfer: Insufficient insulation, long process time, or large temperature difference can make \( Q \neq 0 \).
- Irreversibility: Friction, turbulence, shock waves, throttling, and mixing can make the process adiabatic but not isentropic.
- Real-gas behavior: High pressure, very low temperature, or gas composition changes can make ideal-gas equations inaccurate.
- Variable specific heats: At high temperatures, assuming constant \( C_p \), \( C_v \), and \( \gamma \) may create noticeable error.
- Open-system effects: Turbines, compressors, and nozzles often require steady-flow energy equations rather than a simple closed-system piston model.
Common Mistakes and Practical Checks
Most adiabatic process mistakes come from mixing concepts that sound similar but represent different physical conditions. The safest approach is to write the governing condition first, identify the sign convention second, and only then choose the equation.
- Confusing adiabatic with isothermal: Adiabatic means no heat transfer; isothermal means constant temperature.
- Using \( PV = \text{constant} \) for adiabatic compression: That relation is for an isothermal ideal-gas process, not a reversible adiabatic one.
- Forgetting absolute temperature: Temperature ratios must use kelvin or Rankine.
- Assuming adiabatic means isentropic: Adiabatic plus reversible is isentropic; adiabatic alone is not enough.
- Ignoring sign convention: Work done by the gas and work done on the gas have opposite signs in many textbooks and software tools.
Do not use the reversible adiabatic equations unless the problem actually supports both adiabatic and reversible assumptions. A real process can have \( Q = 0 \) and still lose useful work through irreversibility.
Useful Engineering References and Design Context
Adiabatic processes are usually taught through thermodynamics textbooks and then applied through equipment-specific design methods. For practical work, engineers commonly pair the ideal equations with manufacturer data, measured performance, and efficiency-based models.
- Engineering thermodynamics textbooks: Standard thermodynamics texts cover the first law, ideal-gas relationships, reversible adiabatic paths, entropy, and cycle analysis.
- Compressor and turbine performance data: Equipment curves and manufacturer data help translate ideal adiabatic trends into real power, temperature, and efficiency estimates.
- Cycle analysis references: Otto, Diesel, Brayton, Rankine, refrigeration, and Carnot cycle materials show how adiabatic compression and expansion fit into larger energy systems.
Frequently Asked Questions
An adiabatic process is a thermodynamic process where no heat crosses the system boundary. The system can still do work or have work done on it, so pressure, volume, temperature, and internal energy may change even though heat transfer is zero.
Yes. Temperature commonly changes during an adiabatic process because work changes the internal energy of the gas. Adiabatic compression usually increases temperature, while adiabatic expansion usually decreases temperature.
Adiabatic means no heat transfer occurs, while isothermal means temperature remains constant. An isothermal process can involve heat transfer to hold temperature steady, but an adiabatic process has zero heat transfer and usually changes temperature.
No. A process is isentropic only when it is both adiabatic and reversible. Real adiabatic processes can still have friction, turbulence, shock losses, mixing, or other irreversibilities that increase entropy.
Summary and Next Steps
An adiabatic process is defined by no heat transfer across the system boundary. The process can still change pressure, volume, temperature, and internal energy because work may be exchanged with the surroundings.
The most important practical checks are to separate adiabatic from isothermal, confirm whether the process can be treated as reversible, use absolute temperature, and remember that real equipment often needs efficiency corrections beyond the ideal \( PV^\gamma = \text{constant} \) model.
Where to go next
Continue your learning path with related Turn2Engineering resources.
-
Isothermal Process
Compare adiabatic behavior with the constant-temperature process that is most often confused with it.
-
Thermodynamic Cycles
See how adiabatic compression and expansion fit into engines, refrigerators, turbines, and power cycles.
-
Heat Transfer
Learn how real thermal energy movement affects the validity of the adiabatic assumption.