Key Takeaways
- Core idea: Water softening reduces calcium and magnesium hardness so water is less likely to form scale in pipes, heaters, fixtures, and treatment equipment.
- Engineering use: Softening is used in buildings, utilities, industrial facilities, boilers, cooling systems, and treatment trains where hardness affects performance or maintenance.
- What controls it: Influent hardness, peak flow, target finished-water hardness, resin capacity, regeneration strategy, sodium or potassium use, and waste disposal shape the design.
- Practical check: A softener does not replace filtration or disinfection, and salt-based systems must be evaluated for sodium addition, brine waste, and local discharge limits.
Table of Contents
Introduction
Water softening is the process of reducing calcium and magnesium hardness in water to limit scale buildup, improve soap performance, and protect plumbing, heaters, membranes, boilers, and other water-using equipment. In engineering practice, softening is not just a comfort upgrade; it is a treatment decision controlled by water chemistry, equipment sensitivity, operating cost, maintenance, and waste handling.
Hard Water vs Soft Water: Why Softening Matters
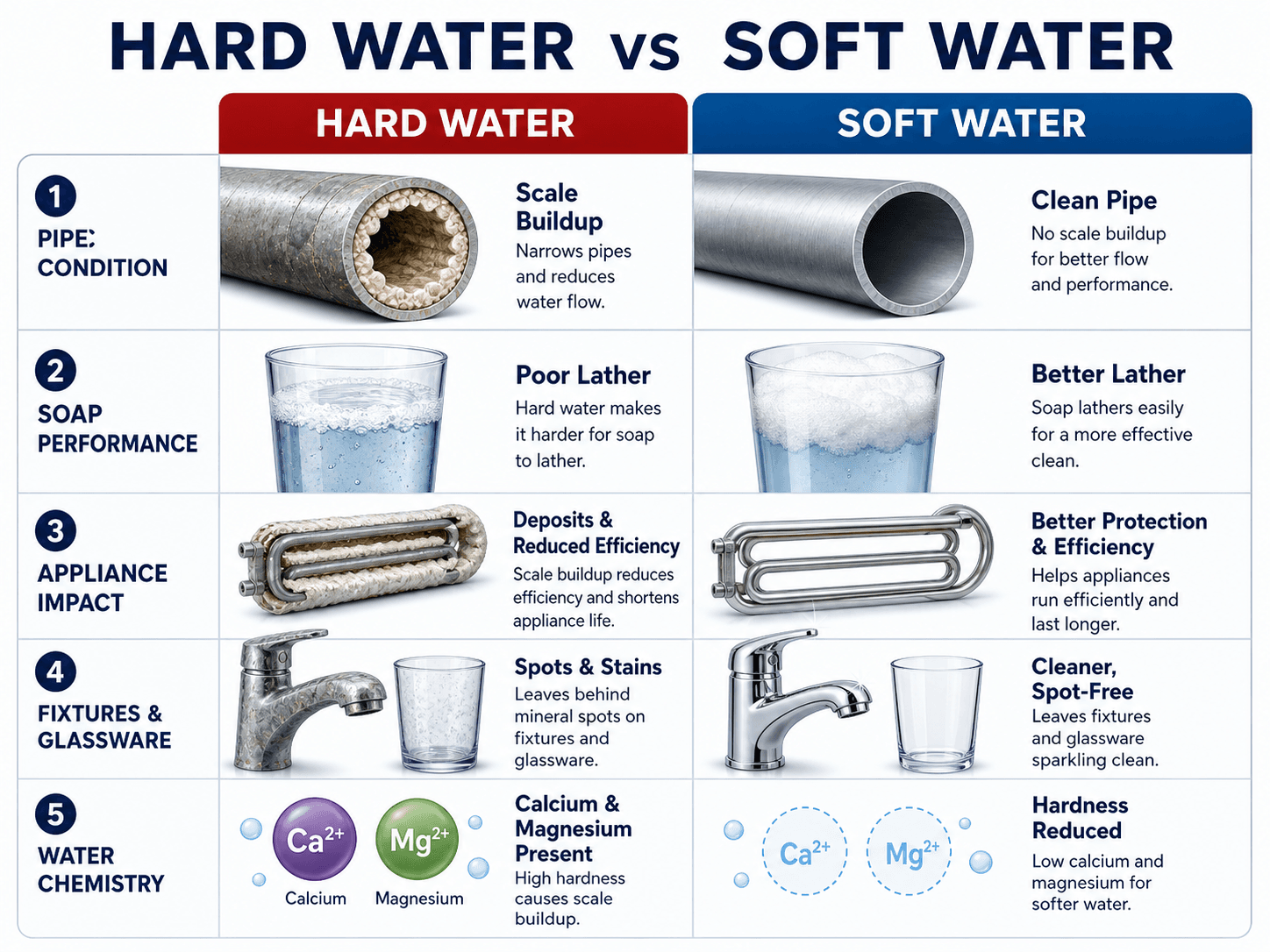
Start by comparing the problem and the outcome: hard water creates deposits and operating issues, while softened water reduces hardness-related scale. The treatment method used to reach that outcome depends on water chemistry and project goals.
What Is Water Softening?
Water softening is a water treatment process used to reduce hardness, which is mainly caused by dissolved calcium ions \(Ca^{2+}\) and magnesium ions \(Mg^{2+}\). Hardness is commonly reported as milligrams per liter as calcium carbonate, written as mg/L as \(CaCO_3\), or as grains per gallon in residential and light commercial systems.
The goal is not always to make water chemically pure. The goal is to reduce the hardness that causes scale, interferes with soap, fouls heat-transfer surfaces, clogs fixtures, and increases maintenance on downstream equipment. In a water resources engineering context, softening may be part of a centralized treatment plant, a building-scale treatment system, an industrial pretreatment step, or a small point-of-entry system.
Hardness is a dissolved water chemistry issue, not a turbidity issue. A sediment filter can remove suspended particles, but it will not remove dissolved calcium and magnesium hardness unless another treatment mechanism is added.
What Causes Hard Water?
Hard water forms when water dissolves minerals from soil, rock, limestone, dolomite, gypsum, or other geologic materials. Groundwater is often harder than surface water because it has longer contact time with mineral-bearing formations. Surface water can also be hard depending on watershed geology, reservoir chemistry, source blending, and seasonal changes.
Temporary and Permanent Hardness
Temporary hardness is associated with bicarbonate alkalinity and can often be reduced by heating or lime softening because calcium carbonate precipitates. Permanent hardness is associated with ions such as sulfate, chloride, or nitrate salts of calcium and magnesium, and it usually requires ion exchange, membrane treatment, or chemical precipitation designed for that chemistry.
How Hardness Is Reported
Reporting hardness as \(CaCO_3\) gives engineers a common basis for comparing calcium and magnesium contributions. This matters because calcium and magnesium have different atomic weights but similar practical effects on scale, soap performance, and treatment demand.
| Hardness level | Typical range as \(CaCO_3\) | Practical meaning |
|---|---|---|
| Soft | 0–60 mg/L | Softening is usually not needed for scale control unless downstream equipment is highly sensitive. |
| Moderately hard | 61–120 mg/L | Some fixture spotting, soap demand, or heating-scale issues may appear depending on use and temperature. |
| Hard | 121–180 mg/L | Softening may be justified where scale, water heaters, appliances, membranes, or process equipment are important. |
| Very hard | More than 180 mg/L | Softening is often evaluated because scaling, detergent use, and maintenance impacts can become significant. |
When Is Water Softening Needed?
Water softening is usually considered when hardness is causing a measurable performance, maintenance, or treatment problem. The decision should be based on a water test, not just appearance, because white deposits can come from hardness, evaporation, silica, corrosion products, or other dissolved solids.
| Situation | Is softening usually helpful? | Engineering reason |
|---|---|---|
| White scale on fixtures and showerheads | Often | Scale indicates dissolved minerals are precipitating as water evaporates or changes temperature. |
| Water heater or boiler deposits | Yes | Heating accelerates hardness precipitation and reduces heat-transfer efficiency. |
| Hardness above 180 mg/L as \(CaCO_3\) | Often | Very hard water commonly creates scale and maintenance problems, especially when heated. |
| Membrane or RO pretreatment | Often | Hardness can scale membrane surfaces unless controlled by softening, antiscalant, recovery limits, or other pretreatment. |
| Drinking water taste complaint only | Maybe | Softening may not solve taste, odor, chlorine, sulfur, or organic-related complaints. |
| Microbial contamination | No | Softening does not disinfect water; microbial issues require an appropriate disinfection or treatment process. |
If the main problem is scale, test hardness. If the main problem is odor, color, particles, bacteria, or chemical contamination, softening alone is usually the wrong first solution.
What Does Water Softening Remove?
A standard ion exchange softener is designed primarily for hardness removal. It is not a universal water purifier. This distinction is important because a softener can make hard water behave better in plumbing and appliances while still leaving other water quality problems untreated.
| Constituent or issue | Removed by typical ion exchange softening? | Important note |
|---|---|---|
| Calcium | Yes | Calcium is one of the primary hardness ions captured by softening resin. |
| Magnesium | Yes | Magnesium is another primary hardness ion and is also exchanged on the resin. |
| Iron | Sometimes, limited | Some ferrous iron may be exchanged, but iron can foul resin and often needs pretreatment. |
| Manganese | Sometimes, limited | Manganese can cause staining and resin fouling; dedicated pretreatment may be needed. |
| Sediment and turbidity | No | Suspended particles require filtration or clarification, not softening alone. |
| Bacteria and viruses | No | Microbial safety requires disinfection or other approved treatment methods. |
| Chlorine taste or odor | No meaningful removal | Activated carbon or another treatment process is typically used for taste and odor control. |
| Nitrate, arsenic, lead, and many dissolved contaminants | No, not by standard softening | These require contaminant-specific treatment and testing. |
How Ion Exchange Water Softening Works
Ion exchange softening is the most common method for residential, commercial, and many small industrial systems. Hard water flows through a pressure vessel filled with resin beads. The resin begins in a sodium or potassium form and has exchange sites that prefer divalent hardness ions such as calcium and magnesium.
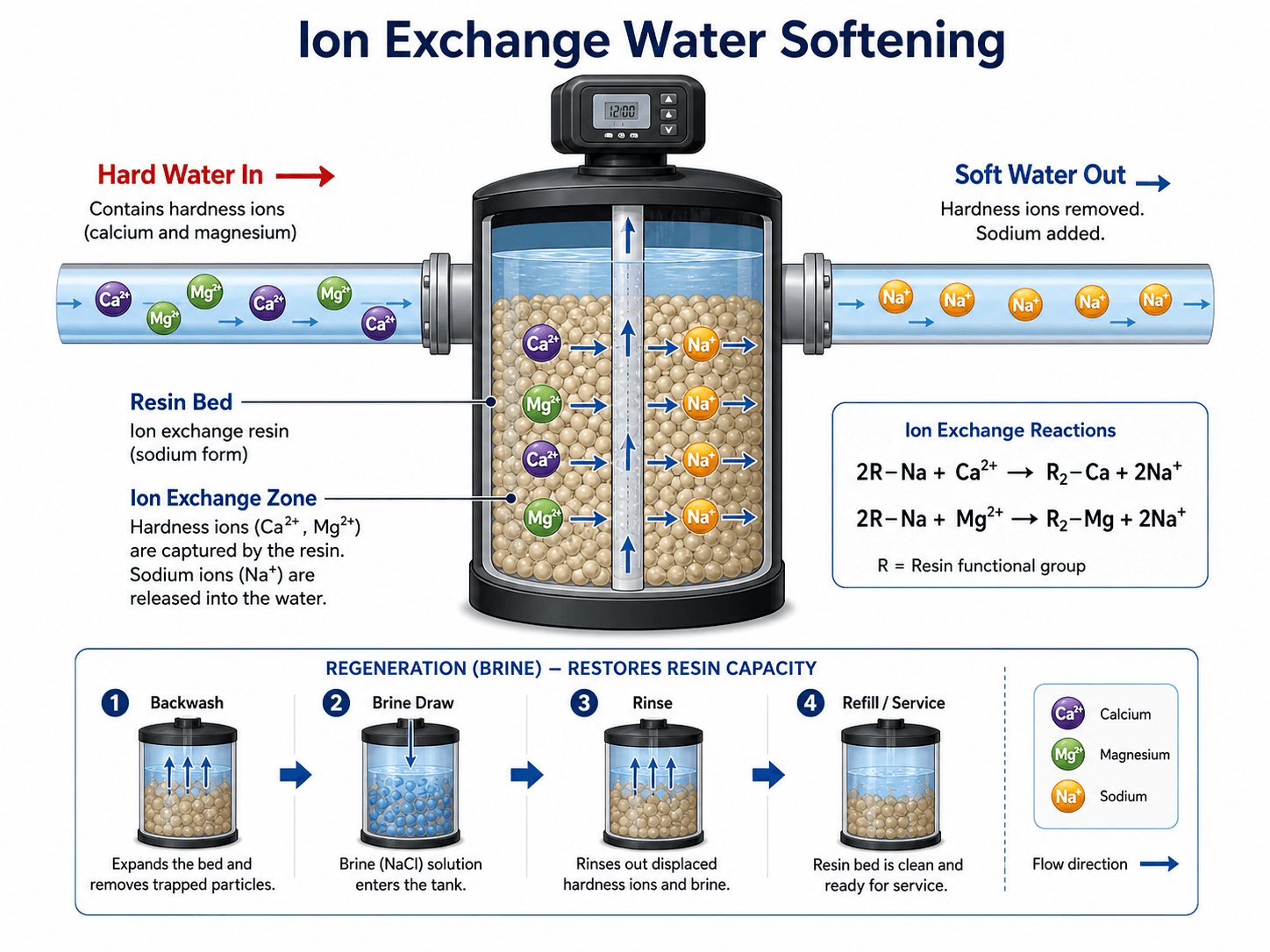
- R The active exchange site on the resin bead.
- Ca²⁺ Calcium hardness ion captured by the resin.
- Mg²⁺ Magnesium hardness ion captured by the resin.
- Na⁺ Sodium ion released into the softened water when sodium-form resin is used.
Service Cycle
During the service cycle, hard water passes through the resin bed and hardness ions attach to the resin. The treated water leaving the vessel has much lower hardness until the resin nears exhaustion. If the softener is undersized, poorly regenerated, or overloaded by peak flow, hardness can break through before the next regeneration cycle.
Regeneration Cycle
Regeneration restores the resin by flushing it with a strong brine solution. The high sodium or potassium concentration reverses the exchange, displaces calcium and magnesium from the resin, and sends the waste brine to drain. A complete regeneration cycle normally includes backwash, brine draw, slow rinse, fast rinse, and return to service.
Does Softened Water Add Sodium?
Sodium-form ion exchange softening adds sodium to the treated water because calcium and magnesium are exchanged for sodium. The amount of sodium added depends on the hardness removed. Potassium chloride can be used instead of sodium chloride in some systems, but it is typically more expensive and still produces a regeneration waste stream.
Types of Water Softening Systems
The phrase water softening can refer to several different system types. Some remove hardness directly, while others only reduce the tendency for scale to attach to surfaces. For engineering accuracy, the system type should match the performance goal.
| System type | Actually softens water? | Best use | Key limitation |
|---|---|---|---|
| Single-tank ion exchange softener | Yes | Common residential and small commercial hardness removal. | May provide hard water during regeneration unless bypassed or scheduled carefully. |
| Twin-tank ion exchange softener | Yes | Higher-demand systems where continuous soft water is needed. | Higher first cost and more equipment complexity. |
| Demand-initiated regeneration softener | Yes | Systems that regenerate based on actual water use instead of only a timer. | Still requires salt or potassium and reliable control settings. |
| Potassium chloride regenerated softener | Yes | Applications where sodium addition is undesirable. | Potassium chloride is usually more expensive and still creates waste brine. |
| Lime softening | Yes | Municipal and industrial systems with mixing, clarification, filtration, and pH control. | Requires chemical feed, solids handling, and stabilization after treatment. |
| Nanofiltration or reverse osmosis | Yes | Broader dissolved-solids removal, membrane treatment, and high-quality process water. | Requires pretreatment and produces a concentrate stream. |
| Salt-free conditioner | Usually no | Scale-control applications where hardness removal is not required. | Often changes scale behavior rather than removing calcium and magnesium. |
Water Softening Methods Compared
Engineers choose a softening method based on flow rate, raw water chemistry, target finished-water quality, solids handling, brine or concentrate disposal, operator skill, cost, and whether the system must remove other dissolved constituents at the same time.
| Method | How it works | Best fit | Main limitation |
|---|---|---|---|
| Ion exchange softening | Exchanges calcium and magnesium for sodium or potassium on resin beads. | Homes, commercial buildings, small systems, pretreatment, and equipment protection. | Requires regenerant chemical use and produces a brine waste stream. |
| Salt-free scale conditioning | Attempts to reduce scale attachment or change mineral behavior without fully removing hardness. | Applications where scale control is desired but measured hardness reduction is not required. | Often does not remove calcium and magnesium, so it should not be treated as equivalent to softening. |
| Lime softening | Raises pH so hardness precipitates as calcium carbonate and magnesium hydroxide solids. | Municipal and industrial systems with operators, mixing, settling, sludge handling, and pH control. | Requires chemical feed, solids separation, sludge management, and finished-water stabilization. |
| Nanofiltration | Uses pressure-driven membranes to remove many divalent ions, including hardness. | Hardness reduction where broader mineral control is needed but full RO-level removal may not be required. | Requires pretreatment, fouling control, pressure, and concentrate management. |
| Reverse osmosis | Uses tighter membranes to remove hardness and a broader range of dissolved ions. | High-quality process water, desalting, reuse, or point-of-use drinking water treatment. | Higher energy use, pretreatment needs, scaling risk, and concentrate disposal. |
If the project objective is lower measured hardness, use a true softening or membrane process. If the objective is only less visible scale, a conditioning approach may be considered, but it should be verified against the actual performance requirement.
Water Softening vs Filtration, RO, and Disinfection
Water softening is often confused with other treatment processes. The difference matters because each process targets a different water quality problem. A complete treatment train may use several processes together, but a softener alone should not be expected to solve every water issue.
| Treatment process | Main purpose | Removes hardness? | Typical role |
|---|---|---|---|
| Sediment filtration | Removes suspended particles. | No | Protects equipment from particles and visible solids. |
| Activated carbon | Improves taste, odor, chlorine, and some organic chemical removal. | No meaningful hardness removal | Often used for aesthetic water quality or chemical polishing. |
| Ion exchange softening | Removes calcium and magnesium hardness. | Yes | Protects plumbing, heaters, boilers, appliances, and process equipment from scale. |
| Reverse osmosis | Removes a broad range of dissolved ions and total dissolved solids. | Yes | Used for high-quality water, point-of-use treatment, desalting, or process water. |
| UV or chemical disinfection | Inactivates or controls microorganisms. | No | Used for microbial safety, not hardness control. |
Where Softening Fits in a Water Treatment Train
Water softening is usually one part of a larger treatment strategy. In a drinking water plant, softening may occur after preliminary treatment and before filtration, or it may be integrated with chemical precipitation and solids removal. In a building or industrial system, softening may be placed upstream of boilers, heaters, humidifiers, membranes, or process equipment.
- Before membranes: Softening or antiscalant control may reduce hardness-related scaling on membrane surfaces.
- Before boilers and heaters: Softening reduces deposits that interfere with heat transfer and increase maintenance.
- After clarification and filtration: Cleaner feed water can protect resin beds from suspended solids and fouling.
- Before distribution: Finished-water stabilization may still be needed so softened water does not become corrosive or unstable.
For broader treatment context, water softening connects closely with water treatment processes, coagulation in water treatment, flocculation in water treatment, and the overall water supply chain.
Key Design and Operating Factors
A softening system should not be selected from hardness alone. Engineers also consider demand profile, peak flow, contact time, resin capacity, regeneration setting, water temperature, pressure loss, competing ions, pretreatment, residual hardness target, and waste disposal.
| Factor | Why it matters | Engineering implication |
|---|---|---|
| Influent hardness | Controls resin loading, chemical demand, scaling potential, and regeneration frequency. | Higher hardness usually requires more capacity, more frequent regeneration, blending, or a different treatment method. |
| Peak flow rate | High flow can reduce contact time and increase the risk of hardness leakage through the bed. | Vessel size, resin depth, service flow rate, and parallel units must match real demand. |
| Iron and manganese | These constituents can foul resin, stain fixtures, and complicate regeneration. | Pretreatment may be needed before softening, especially for groundwater systems. |
| Brine disposal | Regeneration waste contains salts and displaced hardness ions. | Discharge limits, sewer rules, septic impacts, and environmental constraints may control feasibility. |
| Finished-water target | Very low hardness is not always the best operating target. | Some systems blend softened and unsoftened water to balance scale control, taste, corrosion, and operating cost. |
Before approving a softening system, confirm influent hardness, peak flow, resin service rate, regeneration trigger, waste discharge route, bypass or blending strategy, and downstream equipment tolerance.
Operation and Maintenance Considerations
A softening system is an operating asset, not a set-and-forget device. Even a well-sized softener can perform poorly if salt bridges form, brine draw is weak, resin fouls, a bypass valve leaks, or regeneration settings do not match actual water use.
| Maintenance item | What to check | Why it matters |
|---|---|---|
| Salt or potassium level | Confirm the brine tank has enough regenerant and no salt bridge. | Without proper brine strength, resin capacity is not restored. |
| Hardness breakthrough | Test treated water hardness periodically. | Hardness in the effluent means the resin is exhausted, fouled, or not regenerating correctly. |
| Regeneration frequency | Compare control settings to actual water use and hardness loading. | Too little regeneration causes breakthrough; too much wastes salt and water. |
| Resin condition | Watch for fouling from iron, manganese, organics, or suspended solids. | Fouled resin loses exchange capacity and may need cleaning or replacement. |
| Bypass and valve operation | Confirm valves are not leaking hard water around the softener. | A small bypass leak can make a properly functioning unit appear to fail. |
Worked Example: Converting Hardness Units
Water hardness is often reported in mg/L as \(CaCO_3\), while residential softener sizing often uses grains per gallon. A useful conversion is:
Suppose a water sample has a total hardness of 180 mg/L as \(CaCO_3\). The approximate hardness in grains per gallon is:
This is hard water in practical use. For a building, the next question is not simply whether the water is “hard,” but whether the hardness is causing scale, maintenance, soap-use problems, heater inefficiency, membrane fouling, or process-water issues.
Water Softening Method Selection Checklist
Use this checklist as a practical screening tool before selecting a softening approach. It helps separate projects that need true hardness removal from projects that only need scale control, pretreatment, or a broader water treatment train.
Start with a representative water test, identify the treatment objective, confirm whether hardness must be removed or only controlled, screen for iron and fouling risks, check brine or concentrate disposal, then choose the lowest-complexity method that meets the finished-water target.
| Check or decision | What to look for | Why it matters |
|---|---|---|
| Confirm the actual hardness | Use a recent water test reported as mg/L as \(CaCO_3\), grains per gallon, or both. | Softener sizing and treatment selection depend on measured hardness, not just visible scale. |
| Define the treatment objective | Scale reduction, boiler protection, membrane pretreatment, fixture appearance, soap performance, or process-water quality. | Different objectives can justify different finished-water targets and different technologies. |
| Check for fouling constituents | Iron, manganese, turbidity, organics, biological growth, or suspended solids. | These can shorten resin life, reduce exchange capacity, or require pretreatment. |
| Review waste handling | Brine discharge, concentrate disposal, sewer restrictions, septic impacts, and owner requirements. | A technically effective softener may still be a poor choice if waste handling is unacceptable. |
| Consider blending | Partially softened water or blending with unsoftened water. | Blending can reduce salt use, maintain some mineral content, and improve finished-water stability. |
Engineering Judgment and Field Reality
Real softening systems fail most often because the design assumed ideal water, steady demand, or perfect regeneration. Actual demand is variable, operators may adjust settings, salt may bridge in a brine tank, resin can foul, and groundwater chemistry can change seasonally or after a source blend is modified.
A good design accounts for the whole operating environment. That includes peak demand, hydraulic loading rate, pressure loss, maintenance access, salt storage, brine disposal, bypass piping, sampling taps, hardness monitoring, and what happens when the unit is offline.
The most reliable softening systems are not always the ones that chase zero hardness. They are the ones with a clear finished-water target, realistic regeneration settings, manageable waste discharge, and enough monitoring to catch hardness breakthrough before equipment is damaged.
When This Breaks Down
The simple explanation of water softening breaks down when water chemistry, operating conditions, or project constraints make hardness removal more complicated than a single resin vessel. These cases require more careful testing and treatment train design.
- High iron or manganese: Resin can foul, pressure loss can increase, and regeneration may not restore full capacity.
- High turbidity or suspended solids: Particles can clog the resin bed and create channeling, especially if pretreatment is weak.
- High flow variation: Short periods of peak demand can cause hardness leakage even when average flow appears acceptable.
- Strict discharge limits: Brine waste may make salt-based softening difficult or unacceptable.
- Corrosion-sensitive systems: Very low hardness does not automatically mean corrosive water, but aggressive finished water can occur when pH, alkalinity, dissolved carbon dioxide, chloride, sulfate, and pipe materials are not controlled.
Common Mistakes and Practical Checks
Many water softening problems come from treating hardness as a single number instead of part of a complete water chemistry and operations problem. The checks below help avoid common misinterpretations.
- Confusing softening with filtration: Filters remove particles; softening targets dissolved hardness ions.
- Ignoring peak flow: A unit sized only for average flow may leak hardness during high-demand periods.
- Assuming all conditioners are softeners: Scale-control devices may not remove calcium and magnesium.
- Forgetting brine waste: Regeneration discharge can control feasibility, especially where sewer or septic constraints apply.
- Skipping pretreatment: Iron, manganese, organics, and solids can foul resin and reduce service life.
Do not specify a softener from hardness alone. At minimum, also review flow rate, water use pattern, iron and manganese, sodium concerns, finished-water target, and brine disposal.
Useful References and Design Context
Water softening decisions should be based on water chemistry, treatment goals, and practical operating constraints. For drinking water and home treatment context, government guidance is useful because it explains how treatment technologies fit into broader water quality decisions without selling a product.
- EPA drinking water treatment guidance: EPA overview of drinking water treatment technologies provides a useful technical overview of treatment methods, including ion exchange and other drinking water treatment approaches relevant to hardness control.
- Project-specific criteria: Local plumbing rules, sewer discharge restrictions, owner standards, water quality goals, and equipment manufacturer requirements may control whether ion exchange, lime softening, membrane treatment, blending, or another method is appropriate.
- Engineering use: Engineers use references like this to connect softening to source-water chemistry, pretreatment needs, monitoring, operating cost, waste handling, and finished-water quality targets.
Frequently Asked Questions
Water softening is the process of reducing dissolved calcium and magnesium hardness in water. In most small systems, this is done with ion exchange resin that captures hardness ions and releases sodium or potassium ions into the treated water.
No. Water softening mainly targets hardness caused by calcium and magnesium. It does not replace filtration, disinfection, corrosion control, or treatment for contaminants such as pathogens, nitrate, many organic chemicals, or dissolved metals unless the system includes other treatment processes.
True water softening removes or exchanges hardness ions. Water conditioning usually refers to scale-control devices that may change how minerals behave, but they often do not physically remove calcium and magnesium from the water.
Water softening is usually considered when hardness causes scale, poor soap performance, fixture spotting, or equipment maintenance problems. The decision depends on hardness concentration, water use, temperature, treatment goals, brine disposal limits, and the sensitivity of downstream equipment.
Summary and Next Steps
Water softening is a hardness-control process used to reduce calcium and magnesium so water is less likely to form scale, damage equipment, waste energy, or interfere with soap and process performance. The most common approach is ion exchange, but lime softening, membrane treatment, blending, and scale-control methods may also be considered depending on the project.
The best softening decision starts with water testing, a clear finished-water target, realistic flow demand, fouling review, regeneration strategy, and waste-handling check. In real systems, field constraints such as peak flow, iron, manganese, brine disposal, resin fouling, and corrosion control often matter as much as the hardness number itself.
Where to go next
Continue your learning path with related Turn2Engineering resources.
-
Water Treatment Processes
See how softening fits alongside coagulation, flocculation, sedimentation, filtration, and disinfection in a complete treatment train.
-
Coagulation in Water Treatment
Learn how destabilizing fine particles fits into the early stages of water treatment before downstream separation processes.
-
Flocculation in Water Treatment
Understand how gentle mixing builds settleable floc after coagulation and supports clarification and filtration.