Key Takeaways
- Core idea: Entropy describes energy dispersal, possible microscopic arrangements, and the direction real thermodynamic processes tend to move.
- Engineering use: Engineers use entropy to evaluate irreversibility, lost work potential, heat engine limits, refrigeration performance, and cycle efficiency.
- What controls it: Temperature, heat transfer path, phase change, mixing, expansion, pressure loss, friction, and boundary assumptions all affect entropy change.
- Practical check: A local entropy decrease can happen, but the combined entropy change of the system and surroundings must satisfy the Second Law.
Table of Contents
Introduction
Entropy is a thermodynamic property that measures how dispersed energy is and how many microscopic arrangements a system can have. In mechanical engineering, entropy is used to judge process direction, irreversibility, heat engine limits, refrigeration performance, and how much useful work is lost when real equipment departs from ideal behavior.
Visual Guide to Entropy
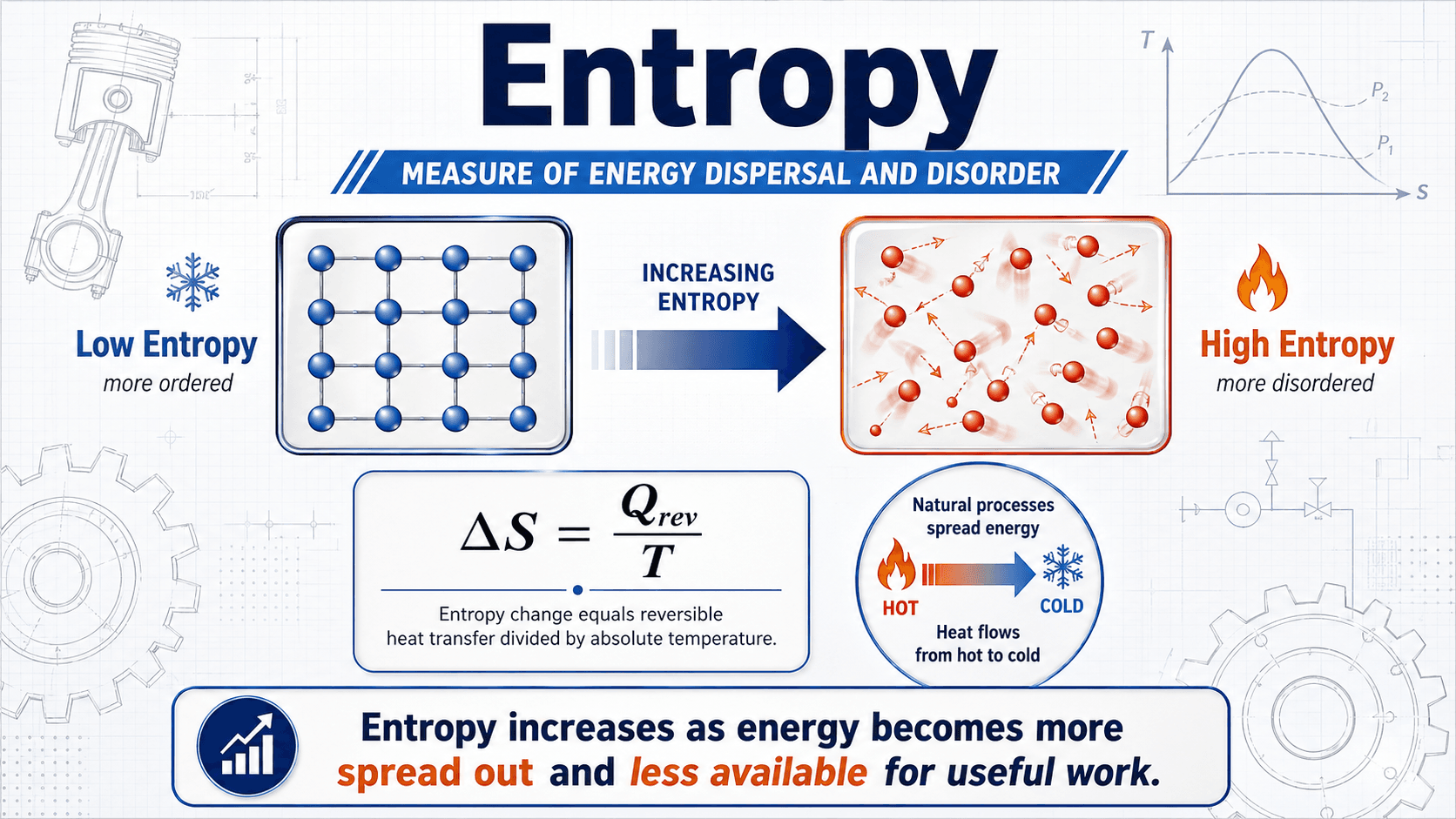
Notice that entropy is not just “messiness.” For engineers, the useful question is whether energy is becoming less available to produce work because of heat transfer, mixing, friction, pressure loss, expansion, or other irreversible effects.
What is Entropy?
Entropy, usually represented by \(S\), is a thermodynamic state property. It does not depend on the path used to reach a state, but the entropy change between two states helps describe whether a process is physically possible, reversible, or irreversible. Higher entropy generally means energy is more spread out, matter is more mixed, or the system has more possible microscopic arrangements.
The common phrase “entropy means disorder” can help beginners, but it is incomplete. In engineering analysis, entropy is more useful when viewed as a measure of energy quality and irreversibility. Two systems can have the same total energy, but the system with higher entropy usually has less ability to produce useful work.
Entropy explains why a warm room cannot spontaneously concentrate its thermal energy into one hot corner, why pressure drops reduce useful energy, and why no real heat engine can convert all supplied heat into work.
How Entropy Works in Thermodynamics
Entropy works by tracking how energy and matter distribute themselves as a process occurs. In an isolated system, natural processes move toward states with greater total entropy. That does not mean every object becomes more disordered at every moment; it means the combined system and surroundings follow the direction allowed by the Second Law of Thermodynamics.
Energy dispersal
Heat naturally flows from a hotter body to a colder body because that transfer spreads thermal energy over more available microscopic states. The hot body loses entropy, the cold body gains entropy, and the combined entropy change is positive for a real heat-transfer process across a finite temperature difference.
Microstates and probability
At the molecular level, a macrostate is the visible condition of a system, such as pressure, temperature, and volume. A microstate is one possible molecular arrangement that produces that visible condition. States with many possible microstates are more probable, which is why gases spread into available volume and mixed systems do not usually separate without external work.
Irreversibility
Real processes generate entropy through friction, turbulence, unrestrained expansion, heat transfer across a finite temperature difference, throttling, shock waves, electrical resistance, and mixing. Those effects do not violate energy conservation; they degrade energy quality and reduce the amount of energy that can be converted into useful work.
Why Entropy Matters in Mechanical Engineering
Entropy is central to thermal system design because it connects the ideal world of reversible processes to the real world of compressors, turbines, heat exchangers, engines, pumps, nozzles, boilers, condensers, HVAC equipment, and refrigeration cycles. When engineers compare real equipment against ideal performance, entropy generation often explains the gap.
- Heat engines: Entropy shows why only part of the supplied heat can become useful work in systems such as steam turbines, gas turbines, and internal combustion engines.
- Refrigeration and heat pumps: Entropy helps evaluate compressor losses, throttling losses, evaporator performance, and condenser heat rejection.
- Heat exchangers: Entropy generation increases when heat is transferred across large temperature differences or with excessive pressure drop.
- Power cycles: Entropy is used with temperature-entropy diagrams to visualize heat addition, heat rejection, and deviations from ideal cycle behavior.
If a design claims high efficiency, check where entropy is being generated: pressure losses, friction, throttling, imperfect heat transfer, leakage, nonideal compression, and uncontrolled mixing are often the hidden causes of lost performance.
Key Factors That Control Entropy Change
Entropy change depends on the process path used for evaluation, the initial and final thermodynamic states, and the boundary used to define the system. The same physical event can look different depending on whether the engineer analyzes the working fluid, the device, the surroundings, or the combined universe.
| Factor | Why it matters | Engineering implication |
|---|---|---|
| Absolute temperature | Entropy change from heat transfer depends on temperature, not just heat quantity. | Heat transferred at lower temperature usually creates a larger entropy change for the same amount of heat. |
| Heat transfer path | Reversible and irreversible heat transfer produce different entropy accounting. | Large temperature differences in heat exchangers increase entropy generation and reduce useful work potential. |
| Pressure loss | Friction and flow resistance convert useful mechanical energy into internal energy. | Ducts, piping, valves, nozzles, and heat exchangers should be checked for pressure-drop penalties. |
| Mixing and diffusion | Mixed fluids have more possible molecular arrangements than separated fluids. | Mixing is usually irreversible, so separation requires external work or a dedicated process. |
| Phase change | Melting, boiling, condensation, and freezing change molecular freedom and energy distribution. | Entropy change during phase change is important in boilers, condensers, evaporators, and thermal storage. |
| System boundary | A system can lose entropy while its surroundings gain more. | Always define the system, surroundings, and control volume before judging the Second Law result. |
Entropy Equations and Units
Entropy is measured in joules per kelvin \((\text{J/K})\), kilojoules per kelvin \((\text{kJ/K})\), or on a mass basis as \(\text{kJ/(kg·K)}\). In thermodynamics tables, specific entropy is often listed as \(s\), while total entropy is listed as \(S\).
This equation defines entropy change using a reversible heat-transfer path. The subscript “rev” matters because entropy is a state property, but heat transfer is path dependent. Engineers often evaluate entropy change by imagining a reversible path between the same two states, even if the actual process is irreversible.
- \(\Delta S\) Change in total entropy, commonly in \(\text{J/K}\) or \(\text{kJ/K}\).
- \(\delta Q_{\text{rev}}\) Differential heat transfer for an equivalent reversible path.
- \(T\) Absolute temperature in kelvin or rankine; entropy equations must not use Celsius or Fahrenheit temperature differences as absolute temperature.
- \(s\) Specific entropy, usually in \(\text{kJ/(kg·K)}\), used in steam tables, refrigerant tables, and cycle analysis.
| Situation | Common entropy relation | Use case |
|---|---|---|
| Constant-temperature reversible heat transfer | \(\Delta S = Q_{\text{rev}} / T\) | Idealized reservoirs, phase-change approximations, and basic entropy calculations. |
| Phase change at constant temperature | \(\Delta S = \Delta H_{\text{phase}} / T\) | Melting, boiling, condensation, freezing, evaporators, boilers, and condensers. |
| Incompressible substance with constant heat capacity | \(\Delta S = mc \ln(T_2/T_1)\) | Liquid water, solids, and approximate heating or cooling calculations. |
| Ideal gas, constant temperature | \(\Delta S = nR \ln(V_2/V_1)\) | Idealized gas expansion or compression at constant temperature. |
| System plus surroundings | \(\Delta S_{\text{univ}} = \Delta S_{\text{sys}} + \Delta S_{\text{surr}}\) | Second Law checks for spontaneous processes and real equipment losses. |
Entropy Sanity Check for Engineering Problems
Entropy problems become easier when the analysis starts with the right boundary and a realistic expectation for the sign of entropy change. Use this sanity check before trusting a calculation, table lookup, or software result.
Define the system boundary → identify whether mass crosses the boundary → list heat transfer, work, mixing, pressure loss, and phase change → calculate or look up entropy change → add surroundings if needed → check whether the total result satisfies the Second Law.
| Check or decision | What to look for | Why it matters |
|---|---|---|
| Define the boundary first | Closed system, open system, isolated system, surroundings, or control volume. | The same process can show different entropy changes depending on what is included. |
| Use absolute temperature | Kelvin or rankine in entropy equations involving \(T\). | Using Celsius or Fahrenheit as absolute temperature produces physically wrong results. |
| Separate property change from entropy generation | Property tables may show \(\Delta s\), while losses create entropy generation. | A device can have small property change while still destroying useful work through irreversibility. |
| Check the expected sign | Expansion, heating, melting, boiling, and mixing usually increase entropy of the material. | A sign error often indicates reversed states, wrong heat direction, or a bad system boundary. |
| Add the surroundings when judging spontaneity | Compute or reason through \(\Delta S_{\text{sys}} + \Delta S_{\text{surr}}\). | A system entropy decrease is possible, but the total entropy change must satisfy the Second Law. |
Worked Example: Heat Transfer Between Two Reservoirs
Consider 100 kJ of heat transferred from a hot reservoir at 500 K to a cold reservoir at 300 K. The hot reservoir loses entropy, and the cold reservoir gains entropy. The energy is conserved, but the process still generates entropy because heat crosses a finite temperature difference.
Interpretation
The hot reservoir became less entropic, but the cold reservoir gained more entropy than the hot reservoir lost. The positive total entropy change confirms that the heat transfer direction is physically allowed and irreversible. In an actual heat exchanger, this same idea explains why smaller temperature differences can reduce entropy generation but often require more heat-transfer area and higher equipment cost.
Connection to useful work
The entropy generated in this transfer represents lost opportunity to produce work. A reversible engine operating between the two temperatures would extract some work while moving heat. Direct heat transfer across the temperature difference gives up that opportunity, which is why entropy is closely tied to exergy destruction and real efficiency losses.
Entropy vs Enthalpy
Entropy and enthalpy are both thermodynamic properties, but they answer different engineering questions. Enthalpy is especially useful for tracking energy flow in open systems. Entropy is especially useful for tracking irreversibility, process direction, and the quality of energy.
| Concept | Entropy | Enthalpy |
|---|---|---|
| Symbol | \(S\) or specific entropy \(s\) | \(H\) or specific enthalpy \(h\) |
| What it helps describe | Energy dispersal, irreversibility, and process direction. | Energy carried with flowing fluids, heat addition, and shaft-work devices. |
| Common units | \(\text{kJ/K}\) or \(\text{kJ/(kg·K)}\) | \(\text{kJ}\) or \(\text{kJ/kg}\) |
| Typical engineering use | Second Law checks, exergy loss, cycle limits, and \(T\)-\(s\) diagrams. | Energy balances, steam tables, refrigerant tables, turbines, compressors, and heat exchangers. |
In many thermal systems, engineers use both properties together. For example, turbine performance may be evaluated using enthalpy drop for work output and entropy change to measure how close the process is to ideal isentropic behavior.
Engineering Judgment and Field Reality
Textbook entropy problems often use clean assumptions: ideal gases, internally reversible paths, steady flow, negligible pressure drop, or constant heat capacity. Real equipment rarely behaves that cleanly. Measurements may be noisy, properties may vary with temperature and pressure, and the largest entropy sources may come from practical details that are easy to overlook.
- Heat exchangers: A design with excellent heat recovery can still perform poorly if fouling, maldistribution, or pressure drop creates avoidable irreversibility.
- Compressors: Entropy rise often reflects nonideal compression, heat transfer, mechanical losses, leakage, and off-design operation.
- Turbines: Entropy increase indicates that not all available pressure and temperature drop became useful shaft work.
- Throttling valves: Enthalpy may remain approximately constant, but entropy increases because useful pressure potential is destroyed.
In real plant troubleshooting, a rising entropy trend can be a clue, not the final diagnosis. It should be paired with measured pressures, temperatures, mass flow, equipment curves, fouling checks, leakage checks, and instrument calibration.
When This Breaks Down
Simple entropy explanations are useful, but they break down when the system is highly nonideal, chemically reacting, strongly transient, extremely small in scale, or outside the assumptions behind common property models. Engineers should know when a basic equation is only a teaching approximation.
- Nonideal fluids: Steam, refrigerants, and real gases often require property tables, equations of state, or software instead of simple ideal-gas equations.
- Rapid transient events: Startups, shutdowns, blowdowns, and relief events may require unsteady control-volume analysis.
- Chemical reactions: Combustion and reacting flows need species accounting, reaction thermodynamics, and sometimes equilibrium chemistry.
- Small-scale systems: At microscopic scales, statistical fluctuations can make simplified macroscopic intuition less reliable.
- Bad measurements: Entropy calculations are sensitive to temperature, pressure, and property-state accuracy, so poor instrumentation can mislead the analysis.
Common Mistakes and Practical Checks
Entropy is often misunderstood because it sits between physical intuition, mathematical property tables, and Second Law reasoning. Most mistakes come from treating entropy like a vague idea instead of a state property with specific units, boundaries, and assumptions.
- Saying entropy is only disorder: Disorder is a loose analogy; energy dispersal, microstates, and irreversibility are usually more useful in engineering.
- Ignoring the surroundings: A system can decrease in entropy while the surroundings increase more.
- Using the wrong temperature scale: Entropy equations involving \(T\) require absolute temperature.
- Confusing heat with entropy: Heat is energy in transfer; entropy is a property that changes based on heat-transfer path and temperature.
- Assuming isentropic means real: Isentropic processes are ideal references; real compressors, turbines, nozzles, and pumps generate entropy.
Do not judge a process by system entropy alone. For Second Law reasoning, define the system and surroundings, then check the total entropy change for the combined universe.
Useful References and Design Context
Entropy is a core thermodynamics concept rather than a standalone design code. In engineering practice, it is usually applied through thermodynamics textbooks, property databases, handbooks, equipment standards, and design software.
- Thermodynamics textbooks: Used to define entropy, entropy generation, reversible processes, \(T\)-\(s\) diagrams, exergy, and Second Law efficiency.
- Steam and refrigerant property tables: Used to find specific entropy, enthalpy, pressure, temperature, and quality for working fluids in power and refrigeration cycles.
- ASHRAE Handbook: Useful for HVAC, refrigeration, heat-pump, and psychrometric applications where entropy-related losses influence performance.
- NIST property data: Commonly used for accurate thermophysical properties of fluids when ideal-gas or simplified assumptions are not accurate enough.
Frequently Asked Questions
Entropy is a thermodynamic property that describes how spread out energy is and how many microscopic arrangements a system can have. In practical terms, it helps explain why heat flows from hot to cold, why gases expand, why mixing happens naturally, and why real processes are not perfectly reversible.
For a reversible heat-transfer process at a constant absolute temperature, entropy change is commonly written as delta S equals Q reversible divided by T. More specific equations are used for ideal gases, phase changes, heating and cooling with heat capacity, and open-flow devices such as turbines and compressors.
The entropy of a specific system can decrease if entropy is transferred to the surroundings, such as when water freezes or vapor condenses. The important Second Law check is the combined entropy change of the system and surroundings; for a real spontaneous process, the entropy of the universe increases.
Entropy is important because it shows where useful energy is degraded by irreversibilities such as friction, heat transfer across a finite temperature difference, turbulence, throttling, mixing, and shock losses. Engineers use entropy to evaluate cycle efficiency, equipment performance, exergy destruction, and realistic limits on heat engines and refrigeration systems.
Summary and Next Steps
Entropy is a thermodynamic property that connects energy dispersal, microscopic probability, process direction, and irreversibility. For mechanical engineers, it is one of the most important ideas behind the Second Law of Thermodynamics because it explains why real systems lose useful work potential even when total energy is conserved.
The most useful workflow is to define the system boundary, identify heat transfer, work, mass flow, mixing, pressure loss, and phase change, then check entropy change for both the system and surroundings. In real equipment, entropy generation often points toward friction, throttling, heat-transfer limitations, leakage, fouling, turbulence, or off-design operation.
Where to go next
Continue your learning path with related Turn2Engineering resources.
-
Second Law of Thermodynamics
Learn how entropy controls process direction, irreversibility, and the limits of real energy conversion.
-
Reversible and Irreversible Processes
Review the difference between ideal reversible behavior and real processes that generate entropy.
-
Heat Engines
See how entropy limits thermal efficiency in engines, turbines, and power cycles.