Key Takeaways
- Core idea: Flocculation gently mixes water so destabilized particles collide and form larger clumps called floc.
- Engineering use: It usually follows coagulation and prepares water for sedimentation, clarification, and filtration.
- What controls it: Mixing intensity, detention time, pH, alkalinity, coagulant dose, temperature, turbidity, organic matter, and basin hydraulics affect floc growth.
- Practical check: Good floc must be large, dense, and strong enough to settle or filter without being broken apart by excess mixing.
Table of Contents
Introduction
Flocculation in water treatment is the gentle mixing process that brings destabilized fine particles together into larger clumps called floc. These larger flocs are easier to remove by sedimentation, clarification, or filtration. Flocculation usually follows coagulation and directly affects turbidity removal, filter performance, and overall treatment reliability.
Where Flocculation Fits in Water Treatment
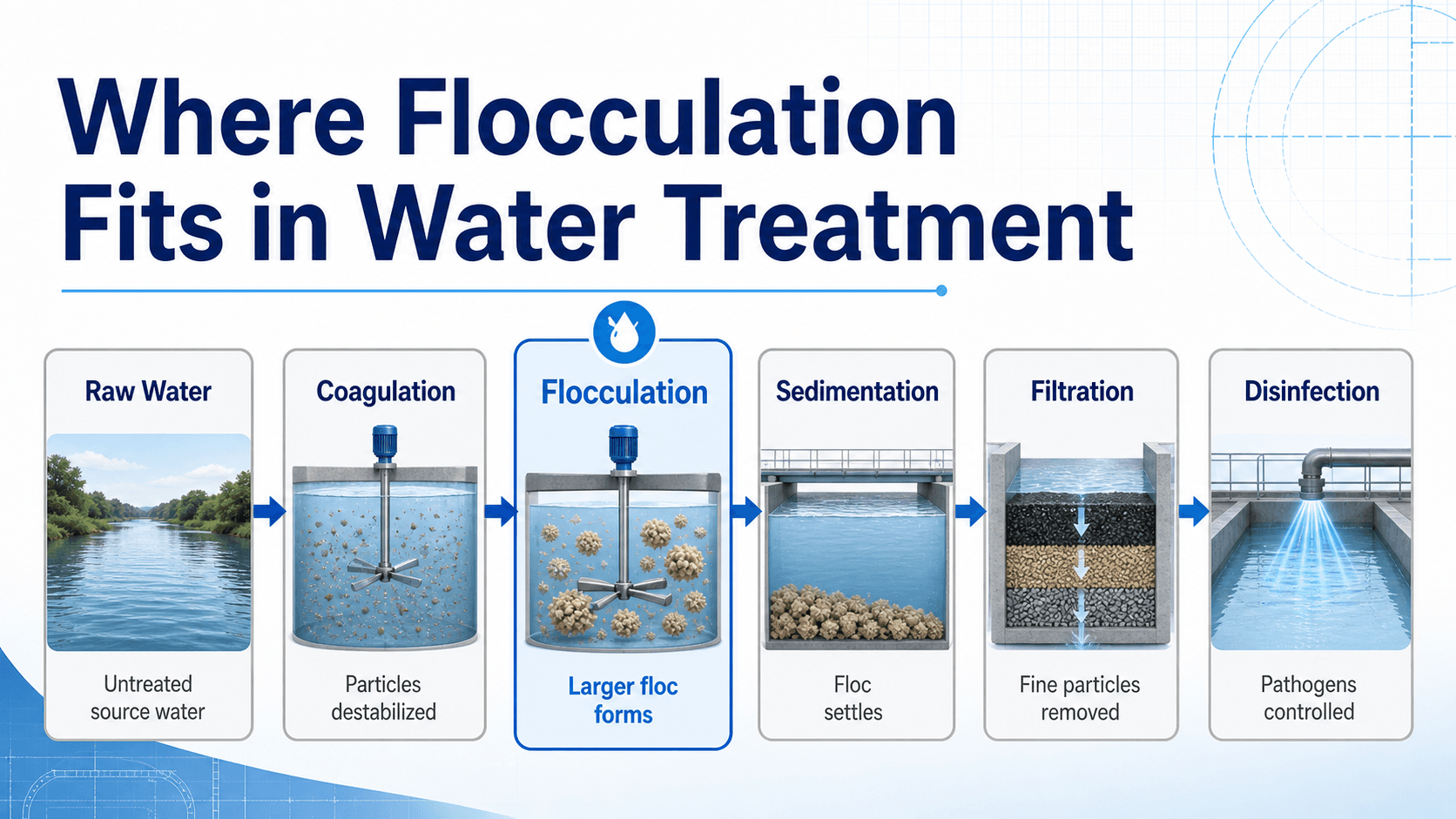
The most important idea is sequence: coagulation prepares the particles, flocculation grows them, sedimentation removes the larger solids, and filtration captures fine particles that remain.
What Is Flocculation in Water Treatment?
Flocculation is the slow-mixing step used to grow small destabilized particles into larger, removable floc. The process is especially important in conventional drinking water treatment because many suspended particles and colloids are too small to settle quickly on their own.
A floc is a loose cluster of particles, coagulant precipitate, organic matter, clay, silt, algae fragments, and other suspended solids that have joined together during mixing. Good floc is large enough to settle or filter, dense enough to drop out in a clarifier, and strong enough to survive the flow path into the next treatment unit.
Flocculation does not fix poor coagulation by itself. It grows particles that have already been chemically destabilized. If coagulant dose, pH, or alkalinity are wrong, the flocculation basin may only produce small, weak, or inconsistent floc.
Why Flocculation Is Important in Water Treatment
Flocculation is important because many suspended particles are too small to settle or filter efficiently on their own. By growing those particles into larger floc, the process improves sedimentation, reduces filter loading, lowers turbidity, and helps downstream disinfection work more reliably.
Poor flocculation often shows up later in the plant as high settled water turbidity, shorter filter runs, faster filter headloss, more frequent backwashing, or unstable finished-water quality. For a broader look at the full treatment train, see water treatment processes.
Coagulation vs Flocculation
Coagulation and flocculation are often discussed together, but they are not the same step. Flocculation and coagulation work as a paired process: coagulation destabilizes fine particles, while flocculation grows those particles into removable floc.
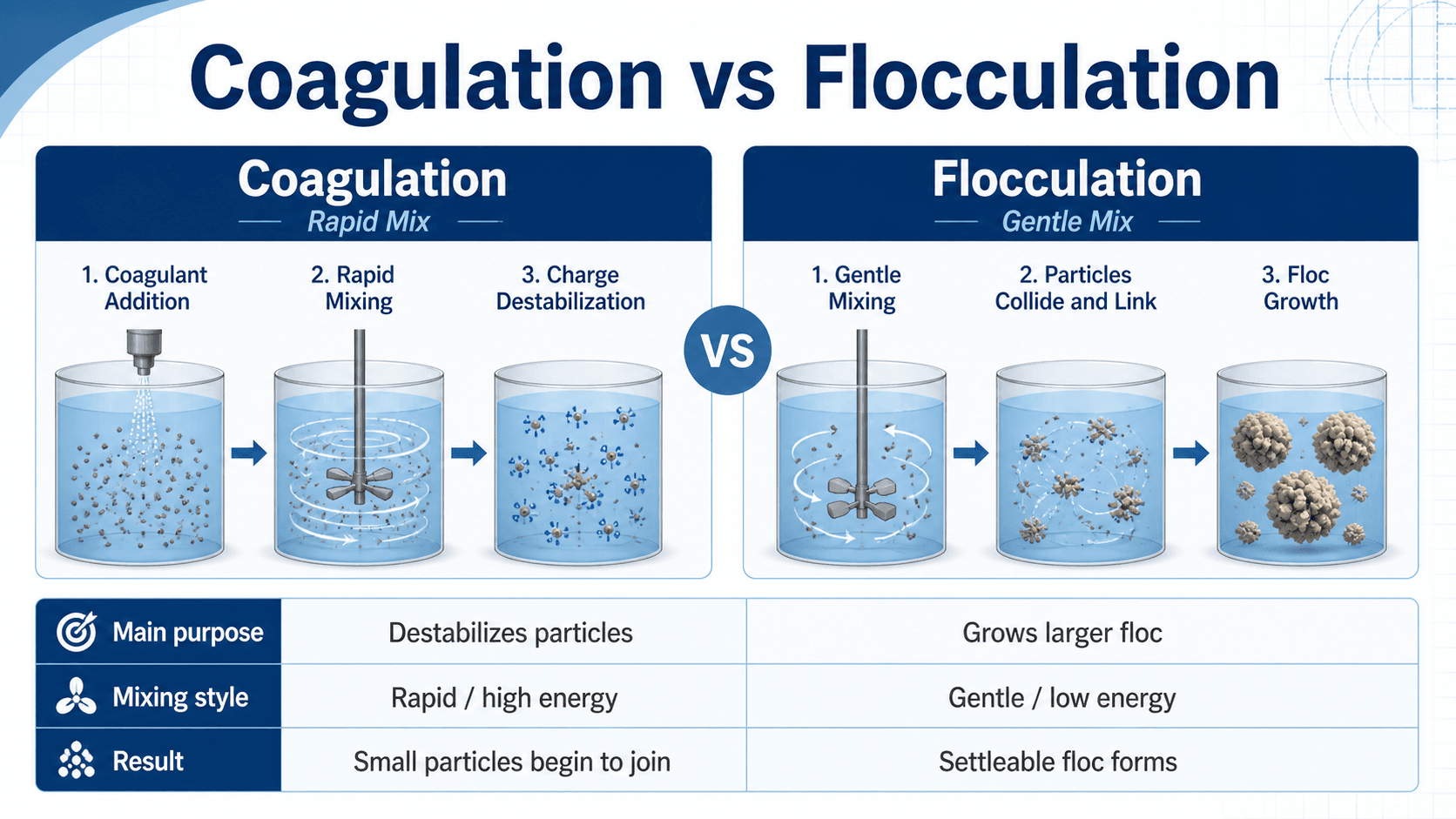
| Topic | Coagulation | Flocculation |
|---|---|---|
| Main purpose | Destabilize fine particles and colloids. | Grow destabilized particles into larger floc. |
| Mixing style | Rapid, high-energy mixing. | Slow, gentle, controlled mixing. |
| Typical location | Rapid mix basin or inline mixer. | Flocculation basin, baffled channel, or staged mixing basin. |
| Chemical role | Coagulant dose, pH, and alkalinity control particle destabilization. | Polymer aids may be used, but the main goal is controlled particle contact and floc growth. |
| Common failure sign | Particles remain stable and do not form good floc. | Floc is small, weak, sheared, floating, or slow-settling. |
How Flocculation Works
Flocculation works by creating controlled particle collisions. After coagulation reduces the stability of fine particles, gentle mixing gives those particles repeated opportunities to contact each other, attach, and form larger aggregates. As floc grows, it becomes easier to remove by gravity settling or filtration.
Step 1: Destabilized particles enter the flocculation basin
Water enters flocculation after rapid mix. At this point, the particles should already be chemically destabilized. If coagulation was ineffective, the basin may not produce strong floc no matter how well the mixers operate.
Step 2: Gentle mixing creates collisions
Mixing must be strong enough to move particles into contact but gentle enough to avoid breaking the floc that is forming. This balance is why flocculation is often staged, with mixing energy reduced as floc grows larger.
Step 3: Floc grows and becomes removable
As particles collide, attach, and bridge together, floc grows larger and more visible. The goal is not simply to make large particles; the goal is to make floc that can survive basin flow and then settle or filter efficiently.
Flocculation Basin Design and Operation
A flocculation basin is designed to provide enough detention time and controlled mixing for floc growth. The basin may use mechanical paddle mixers, vertical shaft mixers, hydraulic baffles, serpentine channels, or multiple compartments that gradually reduce mixing intensity.

Staged or tapered mixing
Tapered mixing is common because newly destabilized particles need enough energy to collide, while larger floc needs gentler conditions to avoid shearing. A basin with the same aggressive mixing throughout can form floc and then break it apart before sedimentation.
Detention time and short-circuiting
Detention time gives floc time to grow, but actual performance depends on hydraulics. Dead zones, short-circuiting, poorly placed baffles, and uneven flow distribution can reduce effective contact time even when the basin volume appears adequate.
Mechanical reliability
Mixers, drive units, shafts, paddles, baffles, valves, and access platforms all matter in practice. A flocculation basin is not just a tank; it is a controlled hydraulic and mechanical environment for floc growth.
What Controls Floc Formation?
Flocculation performance depends on both chemistry and hydraulics. The best basin design cannot overcome poor coagulant chemistry, and the best chemical dose can still fail if mixing energy, detention time, or flow distribution are wrong.
| Factor | Why it matters | Engineering implication |
|---|---|---|
| Mixing intensity | Controls particle collisions and floc breakage. | Too little mixing slows floc growth; too much mixing shears floc apart. |
| Detention time | Provides time for particles to collide, attach, and grow. | High flow or short-circuiting can reduce effective flocculation time. |
| Coagulant dose | Controls whether particles are properly destabilized before flocculation. | Underdosing, overdosing, or unstable feed can produce weak or inconsistent floc. |
| pH and alkalinity | Affect coagulant chemistry and precipitate formation. | Poor pH conditions can reduce floc strength and settling behavior. |
| Water temperature | Affects viscosity, reaction rates, and particle collision behavior. | Cold water can slow floc growth and require operational adjustment. |
| Turbidity | Affects particle concentration and collision frequency. | Very low or rapidly changing turbidity can make floc formation harder to stabilize. |
| Natural organic matter | Can increase coagulant demand and affect floc quality. | May require dose changes, pH optimization, enhanced coagulation, or additional treatment. |
| Basin hydraulics | Controls flow path, residence time, and shear zones. | Dead zones, short-circuiting, and turbulent transitions can reduce performance. |
Chemicals Used During Coagulation and Flocculation
Flocculation depends on the chemistry established during coagulation. Common coagulants include aluminum sulfate, ferric chloride, ferric sulfate, and polyaluminum chloride. Polymers may be added as flocculant aids to improve particle bridging, floc strength, or settling behavior. For a broader look at treatment chemistry, see chemical treatment.
Chemical selection depends on raw water turbidity, pH, alkalinity, temperature, natural organic matter, target turbidity, sludge handling, and downstream filtration performance. A chemical that works well for one source water may perform poorly when the season or raw water quality changes.
| Chemical or aid | Typical role | Practical note |
|---|---|---|
| Aluminum sulfate | Common coagulant for particle destabilization. | Performance depends strongly on pH and alkalinity. |
| Ferric chloride or ferric sulfate | Iron-based coagulants used for turbidity and organic matter control. | Can affect sludge quantity, pH, color, and corrosion-control considerations. |
| Polyaluminum chloride | Pre-hydrolyzed coagulant used in some plants for stable performance. | May perform well across certain raw water conditions but still needs jar testing or pilot testing. |
| Polymers | Flocculant aid for bridging particles and strengthening floc. | Overdosing can cause poor settling, filter problems, sticky floc, or operational instability. |
| Lime, caustic, or acid | pH and alkalinity adjustment. | Used when coagulation chemistry needs correction before flocculation can perform well. |
What Good Floc Looks Like
Operators often judge flocculation visually and through downstream measurements. Good floc should grow steadily through the basin, settle well in a jar test or clarifier, and reduce the solids load on filters. Poor floc may be too small, too weak, too fragile, or too buoyant to remove effectively.

| Floc condition | What it suggests | Downstream effect |
|---|---|---|
| Large, dense floc | Chemistry and mixing are generally working together. | Improved settling and lower filter loading. |
| Pin floc | Floc is too small, often from weak destabilization, low collision opportunity, or poor chemistry. | Poor settling and greater particle carryover to filters. |
| Sheared floc | Floc formed but was broken by excessive mixing, turbulent transitions, or high shear. | Higher settled water turbidity and shorter filter runs. |
| Floating floc | Floc may be low-density, algae-related, gas-bound, or affected by source water conditions. | Clarifier carryover and unstable finished-water treatment. |
Flocculation Troubleshooting Guide
Poor flocculation usually appears as a visible floc problem, a settled water turbidity problem, or a filtration problem. The troubleshooting workflow should start upstream with coagulation chemistry, then move through mixing, basin hydraulics, and downstream performance.
Check raw water change → verify coagulant feed → review pH and alkalinity → observe floc growth → check mixer speed and basin hydraulics → compare settled water turbidity → review filter headloss and run time.
| Field observation | Likely cause | What to check |
|---|---|---|
| Floc stays very small | Low coagulant dose, wrong pH, insufficient collisions, or low turbidity. | Jar test, pH, alkalinity, coagulant feed rate, and rapid mix performance. |
| Floc forms then breaks apart | Excessive mixing, high shear, turbulent transitions, or poor staged mixing. | Mixer speed, basin baffles, flow rate, valve turbulence, and basin outlet conditions. |
| Floc settles slowly | Low-density floc, algae, poor chemistry, or inadequate floc strength. | Coagulant type, polymer aid, raw water organic matter, and settling test results. |
| Filter clogs quickly | Clarifier carryover, weak floc, poor settling, or high solids loading. | Settled water turbidity, filter influent turbidity, headloss rate, and backwash frequency. |
| Floating floc appears | Algae, gas bubbles, low-density solids, or source-water changes. | Source water conditions, preoxidation, basin surface conditions, and clarifier skimming needs. |
| Floc quality changes through the day | Raw water variability, unstable chemical feed, or flow pacing problems. | Flow rate, dose pacing, chemical feed calibration, turbidity trends, and operator logs. |
Jar Testing and Field Adjustment
Jar testing is one of the most practical ways to evaluate coagulation and flocculation together. A jar test simulates rapid mix, slow mix, and settling so operators can compare chemical dose, pH adjustment, polymer aid, floc size, settling behavior, and water clarity before making full-scale changes.
What jar testing helps evaluate
- Coagulant type and dose response.
- pH and alkalinity adjustment needs.
- Polymer or flocculant aid performance.
- Rapid mix and slow mix timing.
- Floc size, strength, and settling behavior.
- Clarified water appearance and turbidity trends.
How to interpret the result
The best jar is not always the one with the biggest floc. Engineers and operators look for floc that forms consistently, settles well, leaves clear supernatant, and does not require an impractical chemical dose or unstable pH condition.
Engineering Judgment and Field Reality
Flocculation performance changes with raw water conditions. A chemical dose and mixer setting that works during stable source water conditions may not work after a storm, algae bloom, reservoir turnover, cold-weather shift, or rapid turbidity change. This is why operators track trends instead of relying only on a fixed setting.
Downstream units often reveal upstream flocculation problems. High settled water turbidity, short filter runs, rapid filter headloss, frequent backwashing, or unstable disinfectant demand can all point back to poor floc formation or poor clarification.
Flocculation is controlled collision, not random stirring. The goal is enough motion to grow floc, but not enough shear to destroy it before sedimentation.
When This Breaks Down
The simplified explanation of flocculation breaks down when chemistry, hydraulics, or source water conditions prevent stable floc growth. In those cases, gentle mixing alone cannot produce good settling or filtration performance.
- Wrong coagulant dose: Particles may stay stable, restabilize, or form weak floc.
- Poor pH or alkalinity: Coagulant chemistry may not produce strong floc under the actual water chemistry.
- Inadequate rapid mix: Coagulant may not disperse quickly enough before slow mixing begins.
- Excessive flocculation energy: Floc may form and then shear apart before sedimentation.
- Short-circuiting: Some water may pass through the basin without enough contact time.
- Cold water: Higher viscosity and slower reactions can reduce floc growth.
- Algae or organic matter: Source-water changes can create buoyant, weak, or inconsistent floc.
Common Mistakes and Practical Checks
The most common mistake is treating flocculation as a simple mixing basin instead of a controlled process that depends on upstream chemistry and downstream performance. Good flocculation requires the right chemical conditions, enough time, appropriate mixing energy, and a basin flow path that avoids damaging the floc.
- Using rapid mixing for too long: High-energy mixing can break forming floc instead of growing it.
- Ignoring pH and alkalinity: Coagulant chemistry can fail even when the mixer is operating correctly.
- Only judging by floc size: Large floc is not useful if it is too weak, buoyant, or fragile to settle.
- Overlooking basin hydraulics: Short-circuiting and dead zones reduce the effective flocculation time.
- Waiting for filters to show the problem: Filter headloss and short runs often indicate an upstream flocculation or clarification issue.
Do not increase mixing energy just because floc is small. Small floc may be caused by poor coagulation chemistry, low alkalinity, low particle concentration, or insufficient detention time rather than weak mixer power.
Useful References and Design Context
Flocculation is usually reviewed as part of the broader drinking water treatment train. Public references are useful for understanding the general process order, while full-scale design and operation depend on raw water quality, pilot testing, utility standards, state requirements, and operator experience.
- CDC drinking water treatment overview: CDC overview of how drinking water treatment works explains the common drinking water treatment sequence and places flocculation between coagulation and sedimentation.
- Project-specific criteria: Local requirements, source water data, pilot studies, chemical supplier testing, utility standards, and operating history can control the final flocculation approach.
- Engineering use: Engineers and operators use flocculation guidance together with jar testing, turbidity data, filter performance, and clarifier behavior to tune the treatment process.
Frequently Asked Questions
Flocculation in water treatment is the gentle mixing process that brings destabilized fine particles together into larger clumps called floc. These larger flocs are easier to remove by sedimentation, clarification, or filtration.
Coagulation is the rapid-mix chemical step that destabilizes fine particles. Flocculation is the slower, gentler mixing step that lets those destabilized particles collide and grow into larger floc that can settle or be filtered.
Gentle mixing creates enough particle contact for floc growth without applying so much shear that the floc breaks apart. Too little mixing produces small floc, while too much mixing can damage floc before sedimentation.
Flocculation usually depends on coagulants added during coagulation, such as aluminum sulfate, ferric chloride, ferric sulfate, or polyaluminum chloride. Polymers may also be used as flocculant aids to improve particle bridging, floc strength, and settling behavior.
Poor flocculation can cause small or weak floc, higher settled water turbidity, clarifier carryover, shorter filter runs, higher filter headloss, more frequent backwashing, and less stable downstream treatment performance.
Summary and Next Steps
Flocculation in water treatment is the controlled slow-mixing step that grows destabilized particles into larger floc. It works between coagulation and sedimentation, and it has a direct effect on turbidity removal, clarification, filter loading, and downstream treatment reliability.
Strong flocculation depends on chemical conditions, mixing intensity, detention time, water temperature, raw water quality, basin hydraulics, and operational control. The best practical check is not simply whether floc appears, but whether it settles well, survives the basin, and reduces the load on sedimentation and filtration.
Where to go next
Continue with the related resources below to connect flocculation with plant components, drinking water plant operation, and broader water resources engineering topics.