Key Takeaways
- Core idea: Enthalpy is a thermodynamic property that combines internal energy with pressure-volume energy.
- Engineering use: It is especially useful for constant-pressure heating, steam systems, HVAC equipment, and steady-flow devices.
- What controls it: Temperature, pressure, phase, reference state, and fluid properties all affect the enthalpy value engineers use.
- Practical check: Engineers usually compare enthalpy differences, not absolute values, and must keep units and reference states consistent.
Table of Contents
Introduction
Enthalpy is a thermodynamic property that represents a system’s internal energy plus the energy associated with pressure and volume. In mechanical engineering, enthalpy is most useful when analyzing heat transfer at constant pressure and flowing-fluid equipment such as turbines, compressors, boilers, condensers, nozzles, and HVAC coils.
How Enthalpy Works
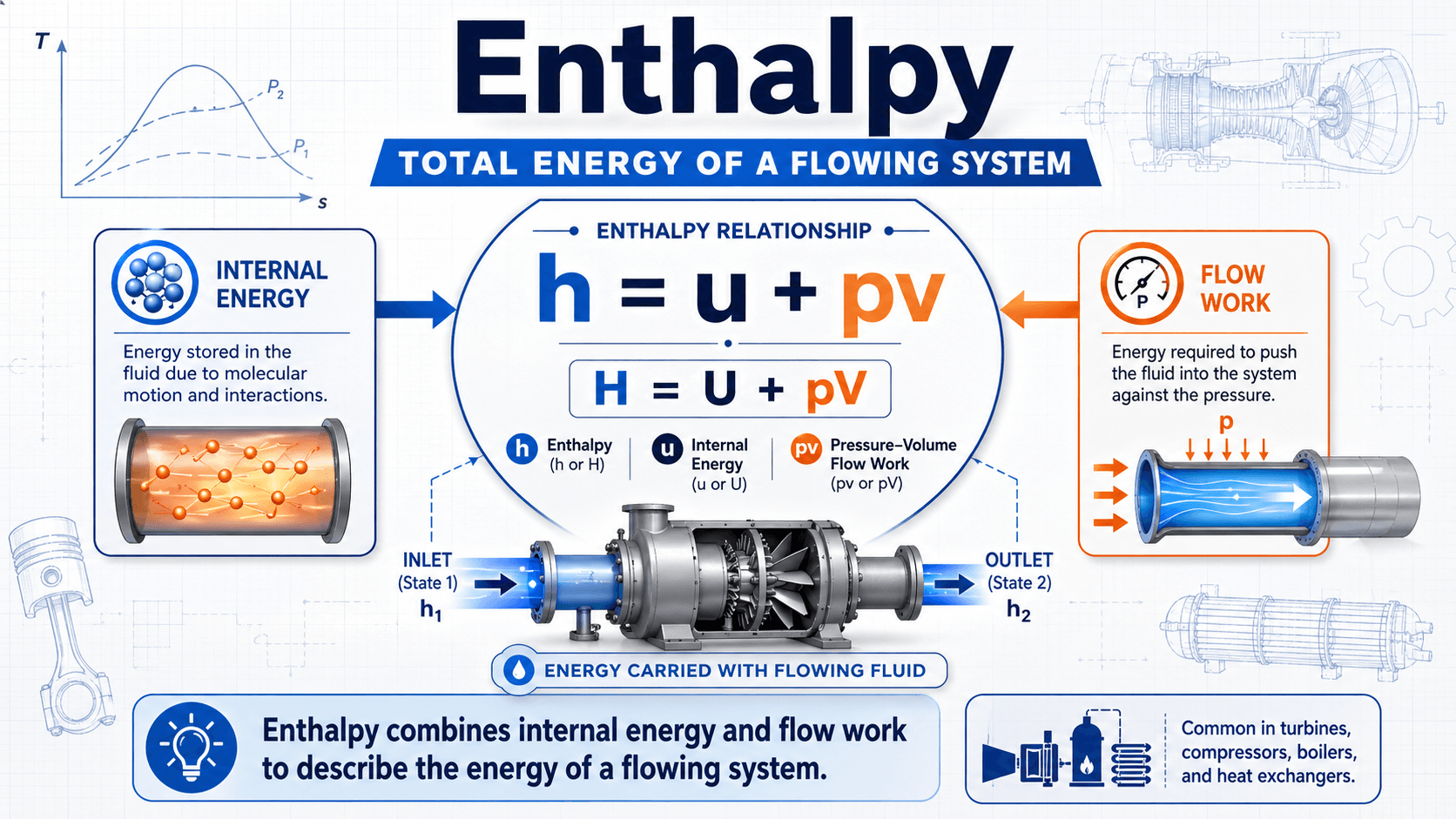
Notice that enthalpy is not just “heat.” It is a property of the state of the fluid or system. The useful engineering quantity is often the change in enthalpy between two states, because that difference helps estimate heat transfer, shaft work, and equipment performance.
What is Enthalpy?
Enthalpy, usually represented by \(H\), is a thermodynamic property defined as internal energy plus the pressure-volume product. Internal energy represents the microscopic energy stored in the system, while the \(PV\) term accounts for the energy associated with pressure and volume.
In mechanical engineering, enthalpy is valuable because many practical systems operate at nearly constant pressure or involve fluid flow. Instead of separately tracking internal energy and the work needed to push fluid into or out of a device, engineers use enthalpy as a combined property.
- \(H\) Enthalpy, commonly expressed in joules, Btu, kJ/kg, or Btu/lbm depending on the analysis.
- \(U\) Internal energy stored within the system.
- \(PV\) Pressure-volume term that becomes especially important in constant-pressure and flowing-fluid problems.
Why Enthalpy Includes Pressure and Volume
The pressure-volume term is the part that makes enthalpy difficult at first. A fluid does not only carry microscopic internal energy. When it occupies volume under pressure, or flows into a device, there is also energy associated with pushing surrounding material out of the way.
Closed systems and constant-pressure heating
In a piston-cylinder example, heat added at constant pressure may increase the internal energy of the gas and also move the piston. Enthalpy is useful because it packages those effects together. For constant-pressure processes with only pressure-volume work, the heat transfer equals the change in enthalpy.
Open systems and flow work
In a turbine, compressor, pump, nozzle, heat exchanger, or HVAC coil, mass crosses the system boundary. The entering and leaving fluid carries energy with it. Specific enthalpy, written as \(h\), lets engineers account for the energy carried per unit mass without repeatedly separating internal energy from flow work.
For equipment analysis, engineers rarely need the absolute stored energy of a fluid. They need the enthalpy difference between the inlet and outlet because that difference connects directly to heat transfer, shaft work, cooling capacity, or heating capacity.
How Engineers Use Enthalpy
Enthalpy appears throughout mechanical engineering because energy systems often involve heat transfer and moving fluids. A steam turbine, refrigeration cycle, boiler, condenser, chiller, compressor, or heat exchanger can usually be understood by comparing inlet and outlet enthalpy states.
- Steam and power cycles: Turbine work and boiler heat input are commonly estimated using steam-table enthalpy differences.
- HVAC systems: Air-side heating and cooling loads depend on the enthalpy change of moist air across coils.
- Refrigeration: Evaporators, condensers, compressors, and expansion devices are evaluated using refrigerant enthalpy states.
- Nozzles and diffusers: Enthalpy can convert into kinetic energy as velocity changes through the device.
- Heat exchangers: The heat gained or lost by a fluid stream is often tied to the mass flow rate times enthalpy change.
When an energy balance produces a heat rate, cooling load, or turbine work that seems unrealistic, check the enthalpy units first. Mixing kJ/kg with J/kg, Btu/lbm with Btu, or mass flow rate with total mass is a common source of large errors.
What Controls Enthalpy?
Enthalpy is a state property, so its value depends on the thermodynamic state of the substance. For a real fluid, that state may depend on temperature, pressure, phase, composition, and the reference state used by a property table or software package.
| Factor | Why it matters | Engineering implication |
|---|---|---|
| Temperature | For many gases, enthalpy changes strongly with temperature. | Heating, cooling, and combustion problems often use temperature change to estimate \(\Delta h\). |
| Pressure | Pressure affects phase behavior and real-fluid properties. | Steam, refrigerants, and compressed fluids require pressure-aware property data. |
| Phase | Liquid, vapor, and two-phase mixtures can have very different enthalpy values. | Boilers, condensers, evaporators, and flash processes require careful phase identification. |
| Reference state | Absolute enthalpy values depend on the chosen zero reference. | Use enthalpy differences from the same table, chart, or software source. |
| Composition | Mixtures such as humid air or combustion gas do not behave like a pure substance. | Psychrometric charts, mixture models, or property software may be needed. |
Enthalpy Change and Common Equations
Most engineering calculations use enthalpy change rather than absolute enthalpy. The change in enthalpy between two states is written as \(\Delta H\) for total enthalpy or \(\Delta h\) for specific enthalpy per unit mass.
For flowing systems, engineers usually work with specific enthalpy:
This simplified steady-flow energy balance is useful when kinetic and potential energy changes are small. It helps connect measured or tabulated enthalpy values to real equipment performance.
- \(\Delta H\) Total enthalpy change, often in J, kJ, Btu, or ft·lbf.
- \(\Delta h\) Specific enthalpy change, commonly kJ/kg or Btu/lbm.
- \(\dot{m}\) Mass flow rate, typically kg/s or lbm/hr.
- \(\dot{Q}\) Heat transfer rate, commonly W, kW, Btu/hr, or tons of refrigeration.
- \(\dot{W}\) Power transfer, such as turbine output or compressor input.
Enthalpy Sanity Check for Engineering Problems
Before trusting an enthalpy result, engineers should confirm that the state, units, property source, and energy balance all make physical sense. The checklist below is a practical way to catch errors before they become design mistakes.
Identify the system boundary → determine whether the process is closed or flowing → find the inlet and outlet states → use one consistent property source → calculate the enthalpy difference → interpret the sign and magnitude against the equipment type.
| Check or decision | What to look for | Why it matters |
|---|---|---|
| System type | Closed system, steady-flow device, or transient control volume. | The correct energy balance depends on whether mass crosses the boundary. |
| Property source | Steam table, refrigerant table, psychrometric chart, ideal gas table, or software model. | Mixing data sources can introduce inconsistent reference states. |
| State identification | Temperature, pressure, quality, superheat, subcooling, or mixture condition. | A wrong phase assumption can make the enthalpy value completely wrong. |
| Sign convention | Whether heat, work, or enthalpy change is positive into or out of the control volume. | Wrong signs can reverse whether equipment appears to produce or consume power. |
| Magnitude check | Compare the result with expected equipment capacity or known operating ranges. | Unrealistic loads often reveal unit conversion or mass flow errors. |
Worked Example: Heating Air with Specific Enthalpy
Suppose air flows through a heating coil at a mass flow rate of \(1.2 \, \text{kg/s}\). The specific enthalpy rises from \(28 \, \text{kJ/kg}\) at the inlet to \(46 \, \text{kJ/kg}\) at the outlet. If changes in kinetic and potential energy are negligible and no shaft work crosses the coil, the heating rate is estimated from the enthalpy increase.
Assumptions
This example assumes steady flow, no shaft work, negligible velocity and elevation changes, and enthalpy values taken from a consistent air-property or psychrometric source. Those assumptions are reasonable for a simple coil estimate but may not capture all real HVAC effects.
Engineering meaning
The coil adds approximately \(21.6 \, \text{kW}\) of heat to the air stream. In a real design, an engineer would also check fan heat, humidity effects, coil approach temperature, control strategy, pressure drop, and whether the selected equipment can deliver that capacity under actual operating conditions.
Engineering Judgment and Field Reality
Enthalpy tables and charts are powerful, but the numbers only help if the state points are correct. In field troubleshooting, the hardest part is often not the equation. It is confirming that temperature, pressure, flow rate, quality, humidity, or refrigerant condition actually represents the system being modeled.
For example, a steam line may have wet steam even if a simplified calculation assumes dry saturated vapor. An HVAC coil may show unexpected enthalpy change because outdoor air leakage or humidity variation is not captured. A compressor calculation may look wrong because suction superheat or discharge pressure was measured at the wrong location.
Textbook enthalpy states are clean. Real equipment has pressure losses, sensor error, fouling, nonuniform flow, heat loss to surroundings, and operating conditions that drift. Experienced engineers check the measurements before blaming the thermodynamic model.
When This Breaks Down
Simplified enthalpy calculations become less reliable when the assumptions behind the energy balance are not valid. The concept itself remains correct, but the shortcut equation may no longer represent the real system accurately.
- Large kinetic energy changes: Nozzles, diffusers, jets, and high-speed flows may require velocity terms in the energy balance.
- Large elevation changes: Pumped systems, hydro applications, or tall piping networks may require potential energy terms.
- Transient operation: Startup, shutdown, cycling equipment, and storage tanks may require time-dependent analysis.
- Two-phase uncertainty: Steam, refrigerants, and boiling or condensing fluids require accurate quality or phase data.
- Nonideal mixtures: Humid air, combustion products, and process gases may need mixture-specific property methods.
Common Mistakes and Practical Checks
Enthalpy is often introduced as “heat content,” but that shortcut can mislead readers. The more accurate view is that enthalpy is a state property, and under specific conditions its change corresponds to heat transfer.
- Calling enthalpy heat in every situation: Heat is energy in transit; enthalpy is a property of a system or flowing fluid.
- Ignoring the reference state: Absolute enthalpy values from different tables may not be directly comparable.
- Using total and specific values interchangeably: \(H\) is total enthalpy, while \(h\) is enthalpy per unit mass.
- Forgetting phase change: Latent heat effects can dominate the enthalpy change in boiling, condensing, melting, and freezing.
- Dropping terms without checking: Kinetic, potential, heat loss, and work terms may matter in certain equipment.
Do not subtract enthalpy values from two different property references unless you know the reference states are compatible. In most engineering work, use one table, one software model, or one chart for the entire calculation.
Useful References and Data Sources
Enthalpy is usually applied through property data, thermodynamics references, and equipment-specific design methods rather than a single design code. The best reference depends on the substance and application.
- Steam tables: Used for water and steam states in boilers, turbines, condensers, and power cycles.
- Refrigerant property tables and pressure-enthalpy charts: Used to analyze refrigeration and heat pump cycles.
- Psychrometric charts: Used for moist-air enthalpy in HVAC heating, cooling, humidification, and dehumidification problems.
- Thermodynamics textbooks: Used to understand the derivation of \(H = U + PV\), the steady-flow energy equation, and property relationships.
Frequently Asked Questions
Enthalpy is a thermodynamic property that combines the internal energy of a system with the pressure-volume energy needed to occupy space or move fluid into and out of a control volume. In engineering, changes in enthalpy are usually more useful than absolute enthalpy values.
Enthalpy is not the same as heat. Enthalpy is a property of the system, while heat is energy transferred because of a temperature difference. Under constant-pressure conditions with only pressure-volume work, the heat transfer equals the change in enthalpy.
Engineers use enthalpy because many real devices involve flowing fluids. Turbines, compressors, pumps, nozzles, boilers, condensers, and HVAC coils all move mass across a boundary, so enthalpy conveniently combines internal energy and flow work in one property.
A negative enthalpy change means the final state has lower enthalpy than the initial state. In a constant-pressure heat transfer or reaction problem, this usually means energy is released by the system, such as during condensation, freezing, or combustion.
Summary and Next Steps
Enthalpy is one of the most useful thermodynamic properties in mechanical engineering because it combines internal energy and pressure-volume energy into a single state property. It becomes especially important when analyzing constant-pressure heat transfer and flowing-fluid equipment.
The key is to focus on enthalpy change, not just the definition. Good engineering use requires consistent units, correct state points, reliable property data, and careful interpretation of whether the result represents heat transfer, work transfer, equipment capacity, or a change in fluid state.
Where to go next
Continue your learning path with related Turn2Engineering resources.
-
First Law of Thermodynamics
Review the energy conservation principle that connects heat, work, internal energy, and enthalpy.
-
Open Systems
Learn why enthalpy is so useful when mass flows through turbines, compressors, pumps, nozzles, and heat exchangers.
-
Heat Transfer
Connect enthalpy change to practical heating, cooling, conduction, convection, and radiation problems.