Key Takeaways
- Core idea: A closed system has fixed mass; mass does not cross the boundary, but energy can cross as heat or work.
- Engineering use: Engineers use closed-system analysis for piston-cylinder devices, sealed tanks, rigid vessels, and other control-mass problems.
- What controls it: The analysis depends on the chosen boundary, heat transfer, work transfer, internal energy change, and sign convention.
- Practical check: Do not confuse a closed system with an isolated system; closed systems can still exchange energy.
Table of Contents
Introduction
A closed system in thermodynamics is a fixed amount of matter separated from its surroundings by a system boundary. Mass does not cross that boundary, but energy can cross as heat or work. This makes closed systems especially important for analyzing sealed tanks, piston-cylinder devices, and control-mass energy balances.
Closed System Boundary Diagram
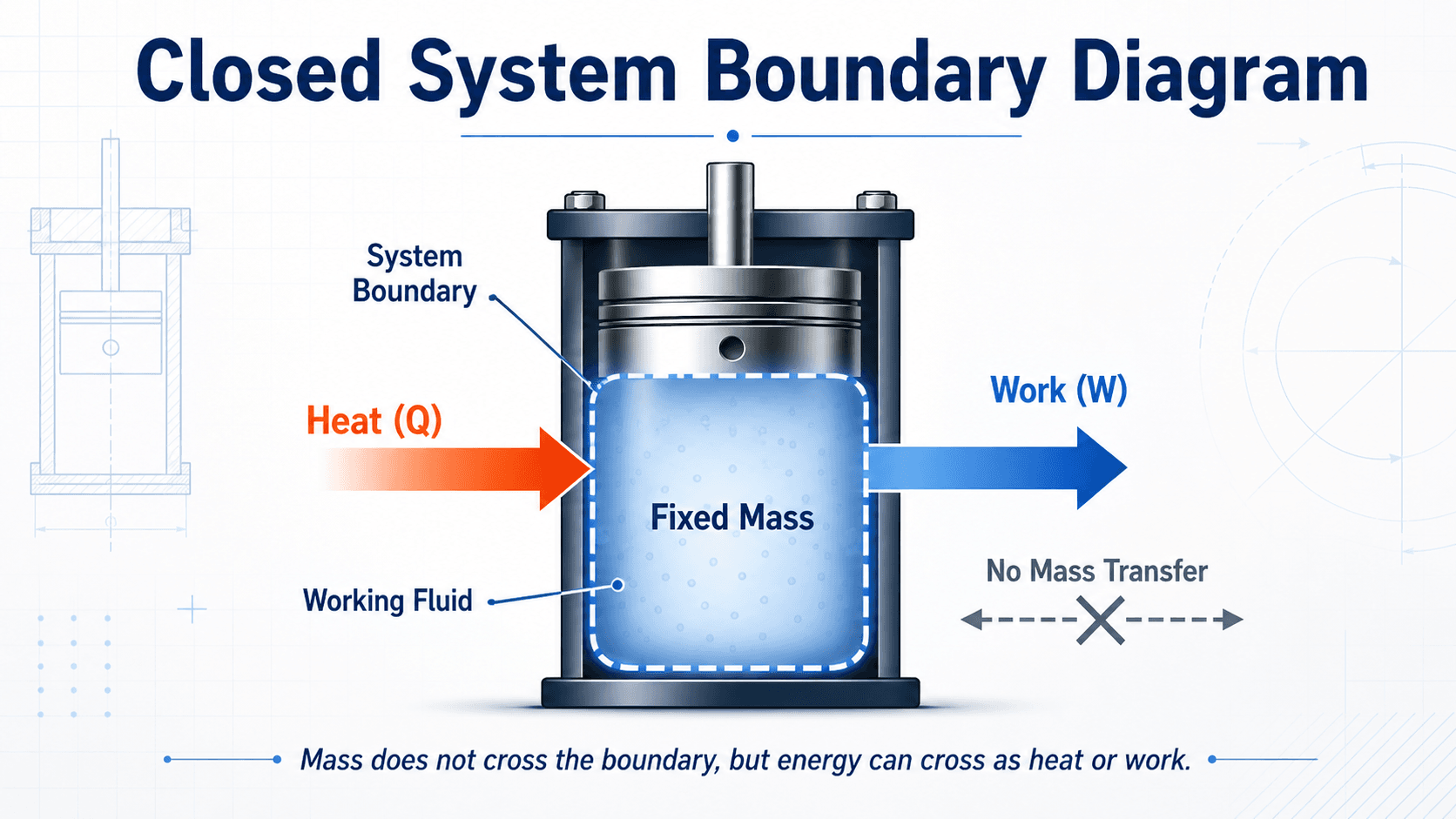
Notice that the boundary is closed only to mass. The system can still gain or lose energy through heat transfer or work transfer.
What is a Closed System?
A closed system is a thermodynamic system containing a fixed mass. The system may be heated, cooled, compressed, expanded, stirred, or electrically energized, but the same matter remains inside the chosen boundary during the process. Because the mass is fixed, closed systems are also called control masses.
The word “closed” often causes confusion because it sounds like nothing can cross the boundary. In thermodynamics, it means closed to mass transfer, not closed to energy transfer. Heat and work can still cross the boundary, and those energy transfers can change internal energy, temperature, pressure, volume, or phase.
A sealed piston-cylinder can be a closed system even when the piston moves. The moving boundary allows work transfer, but the gas mass inside the cylinder remains fixed.
Closed System vs Open System vs Isolated System
Most confusion around closed systems comes from comparing them with open and isolated systems. The classification depends on what crosses the boundary, not simply whether the container looks sealed.
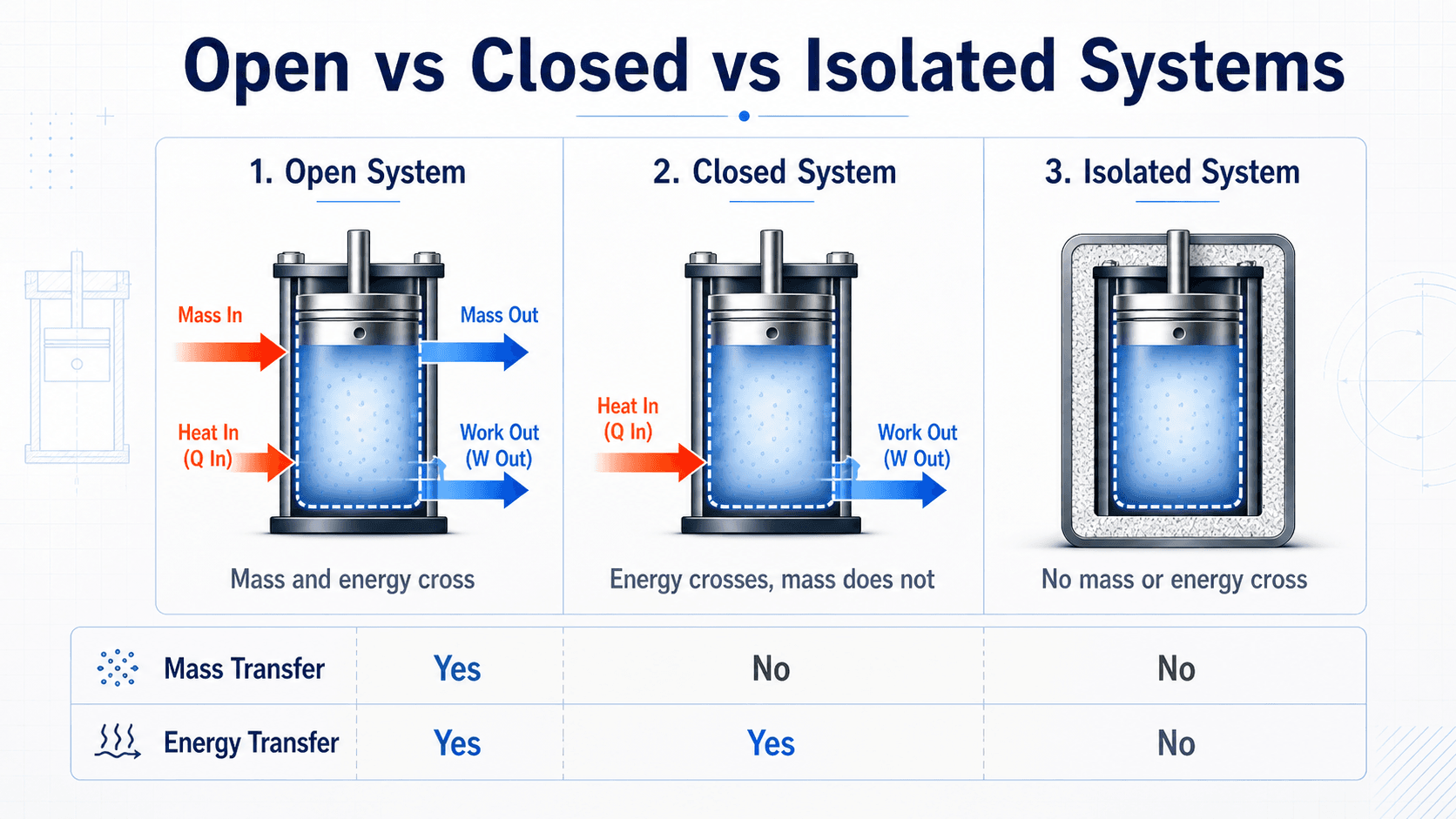
| System type | Mass crosses boundary? | Energy crosses boundary? | Common example |
|---|---|---|---|
| Open system | Yes | Yes | Turbine, compressor, pump, nozzle, heat exchanger |
| Closed system | No | Yes | Sealed piston-cylinder, sealed rigid tank |
| Isolated system | No | No | Ideal insulated sealed container |
For related system classifications, see the Turn2Engineering pages on Open Systems and Isolated Systems.
How System Boundaries Control the Analysis
Closed-system analysis begins by drawing the boundary. The boundary may be a real physical surface, such as the wall of a sealed tank, or an imaginary surface chosen for analysis. Once the boundary is chosen, the engineer asks what crosses it: mass, heat, work, or none of those.
Fixed boundary
A sealed rigid tank has a fixed boundary. Its volume does not change, so boundary work is usually zero. If heat enters the tank, the internal energy of the fixed mass changes, often increasing pressure and temperature.
Moving boundary
A piston-cylinder has a moving boundary. The gas mass can stay fixed while the piston moves outward or inward. This creates boundary work even though no mass crosses the system boundary.
Boundary choice matters
The same physical equipment can be analyzed differently depending on where the boundary is drawn. A compressor with air entering and leaving is usually an open system, but a sealed gas sample inside a piston-cylinder is usually a closed system.
Common Examples of Closed Systems
Closed systems are common in thermodynamics problems because they let engineers follow the same mass through a process. The examples below are useful because each one shows energy crossing the boundary without mass crossing it.
| Example | Why it can be a closed system | What energy transfer may occur |
|---|---|---|
| Sealed piston-cylinder | The same gas stays inside while the piston moves. | Heat transfer and moving-boundary work. |
| Sealed rigid tank | No mass enters or leaves the tank. | Heat transfer; usually no boundary work if volume is fixed. |
| Unopened bottle warming on a table | The liquid or gas remains inside the sealed bottle. | Heat transfer from the room to the contents. |
| Pressure cooker before venting | It may be treated as closed before steam escapes. | Heat transfer raises internal energy and pressure. |
The moment mass begins to enter or leave, the model may no longer be closed. A pressure cooker that vents steam, for example, becomes better represented as an open or transient control-volume problem during venting.
First Law of Thermodynamics for a Closed System
For a closed system, the first law is an energy balance on a fixed mass. Since mass does not cross the boundary, there are no mass-flow energy terms. The system energy changes because heat and work cross the boundary.
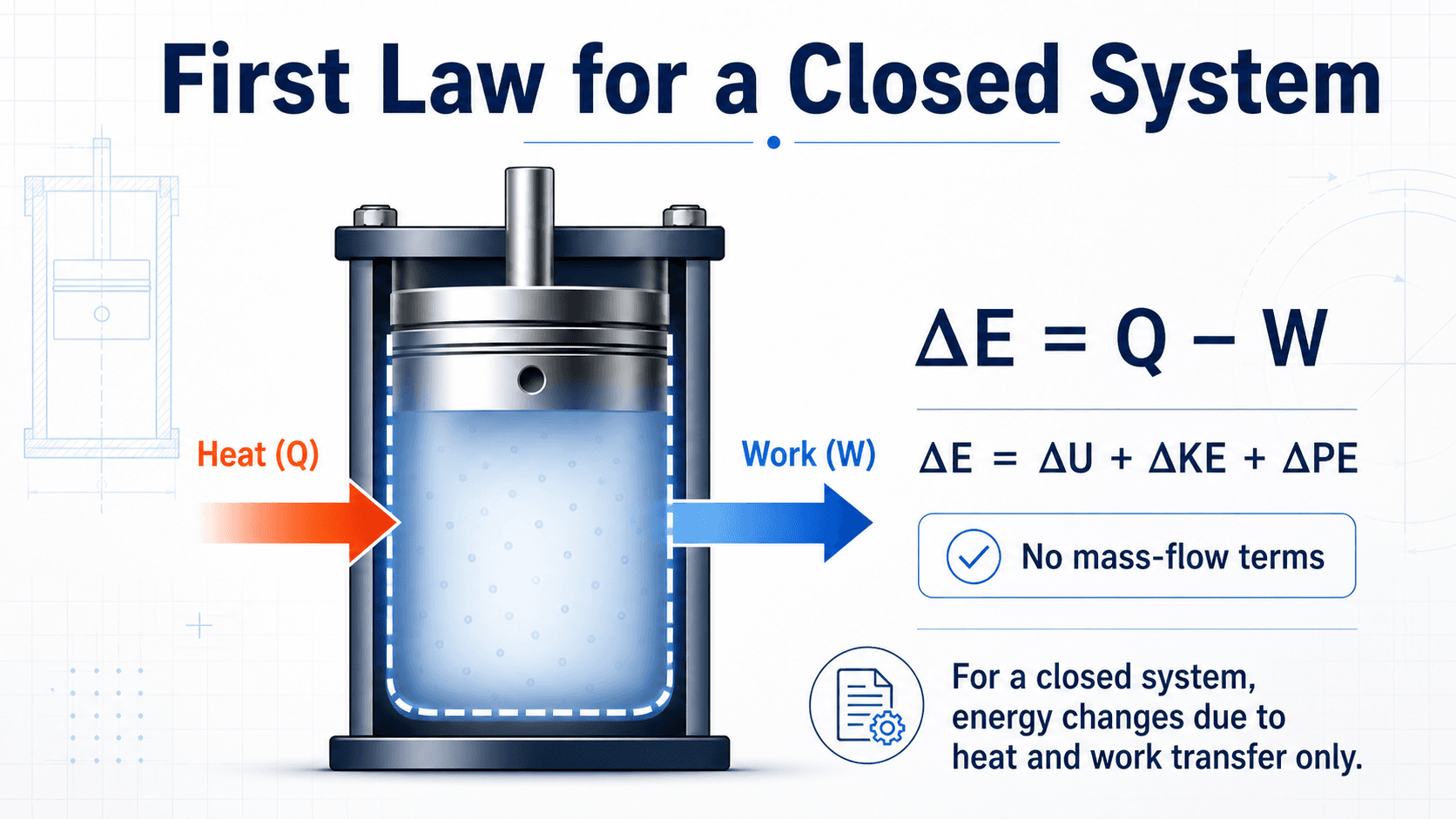
In this common sign convention, \(Q\) is positive when heat is added to the system, and \(W\) is positive when work is done by the system on the surroundings. The total energy change can be expanded as:
Many basic closed-system problems neglect kinetic and potential energy changes. When \(\Delta KE\) and \(\Delta PE\) are small, the energy balance is often simplified to:
- \(Q\) Heat transfer across the boundary, usually in J, kJ, Btu, or ft·lbf depending on the unit system.
- \(W\) Work transfer across the boundary, including boundary work, shaft work, electrical work, or other work interactions.
- \(\Delta U\) Change in internal energy of the fixed mass, often found from property tables, ideal-gas relations, or material data.
For a deeper energy-balance discussion, see the related Turn2Engineering page on the First Law of Thermodynamics.
Closed-System Process Types
Closed systems can undergo many different thermodynamic processes. The system classification tells you whether mass crosses the boundary, while the process type tells you what is held constant or how the state changes.
| Process type | Typical closed-system example | Engineering implication |
|---|---|---|
| Constant volume | Sealed rigid tank | Boundary work is usually zero because volume does not change. |
| Constant pressure | Weighted piston-cylinder | Heat transfer may cause expansion and boundary work. |
| Adiabatic | Insulated compression or expansion | No heat transfer is assumed, so work changes the system energy. |
| Isothermal | Slow expansion with temperature control | Temperature is constant; for ideal gases, internal energy may remain constant. |
| Polytropic | Gas compression or expansion approximation | The pressure-volume path is modeled with \(PV^n = C\). |
This distinction helps avoid a common mistake: “closed system” does not mean “constant volume,” “adiabatic,” or “isolated.” A closed system can be heated, cooled, expanded, compressed, or insulated depending on the actual process.
How to Identify a Closed System
Use this practical check before writing equations. Most thermodynamics mistakes happen before the math starts, when the wrong boundary or system type is chosen.
Start by drawing the boundary. If the same mass remains inside the boundary during the process, analyze it as a closed system. If mass crosses through an inlet, outlet, leak, valve, nozzle, or vent, switch to an open-system or transient control-volume analysis.
| Check or decision | What to look for | Why it matters |
|---|---|---|
| Does mass cross the boundary? | Look for inlets, outlets, vents, leaks, valves, or material flow. | If mass crosses, the system is not closed for that process. |
| Can energy cross? | Look for heat transfer, piston motion, shaft work, electrical work, or stirring. | Energy transfer is allowed in a closed system and drives the energy balance. |
| Is the boundary moving? | Look for pistons, diaphragms, balloons, or deformable walls. | A moving boundary may create boundary work even when mass is fixed. |
| Is the process idealized? | Check assumptions such as adiabatic, constant volume, frictionless, or quasi-equilibrium. | These assumptions affect the equation simplifications, not whether the mass is fixed. |
Worked Example: Sealed Piston-Cylinder
Consider a sealed piston-cylinder containing a fixed mass of gas. Heat is added slowly, and the piston rises. No gas enters or leaves, so the gas is a closed system. The process may still involve both heat transfer and work transfer.
Assumptions
Assume the gas is the system, the piston-cylinder is sealed, kinetic and potential energy changes are negligible, and work output is positive when the gas pushes the piston upward.
Energy balance
If \(20\ \text{kJ}\) of heat enters the gas and the gas does \(7\ \text{kJ}\) of work on the piston, the internal energy increase is:
Engineering meaning
The added heat does not all become stored internal energy because part of it leaves the system as work. The fixed mass gains \(13\ \text{kJ}\) of internal energy, while \(7\ \text{kJ}\) is transferred to the surroundings through piston motion.
Engineering Judgment and Field Reality
Real equipment rarely behaves like a perfectly clean textbook boundary. Seals leak, walls conduct heat, pistons have friction, and instruments measure pressure or temperature with uncertainty. A closed-system model is still useful, but the engineer must decide whether the fixed-mass assumption is reasonable for the time scale and purpose of the analysis.
The system boundary is a modeling choice. If the boundary is drawn around a whole sealed device, the analysis may look closed. If it is drawn around a component with flow through it, the same equipment may require open-system analysis.
This is especially important for refrigeration, HVAC, and power equipment. Individual devices such as turbines, compressors, pumps, nozzles, and heat exchangers are often open systems because mass flows through them, even when they are part of a larger loop.
When This Breaks Down
Closed-system analysis breaks down when the fixed-mass assumption no longer matches the process. The equations may still look simple, but the result can be wrong if the model ignores mass crossing the boundary.
- Venting or leakage: Any steam, gas, liquid, or refrigerant crossing the boundary introduces mass transfer.
- Flow devices: Turbines, compressors, pumps, nozzles, and diffusers usually require open-system analysis.
- Long-duration heat loss: A system treated as adiabatic for a short event may not be adiabatic over a longer period.
- Nonuniform states: Fast compression, stratified tanks, or poorly mixed systems may not have one uniform temperature or pressure.
Common Mistakes and Practical Checks
The most common errors with closed systems are conceptual, not mathematical. Before using the first law, check the boundary, the sign convention, and the process assumptions.
- Confusing closed with isolated: A closed system can exchange energy; an isolated system ideally cannot exchange mass or energy.
- Ignoring boundary work: A moving piston can do work even though the gas mass stays fixed.
- Using open-system equations: Closed systems do not include inlet and outlet enthalpy flow terms.
- Assuming all sealed systems are adiabatic: A sealed container may still gain or lose heat through its walls.
- Switching sign conventions mid-problem: Always state whether work done by the system or on the system is positive.
Do not add mass-flow terms to a closed-system first-law equation. If mass is crossing the boundary, the model is not closed for that process.
Useful References and Design Context
Closed systems are a foundational thermodynamics concept, so the most useful references are engineering thermodynamics texts that clearly define system, surroundings, boundary, open system, closed system, and isolated system.
- Engineering thermodynamics reference: BCcampus Introduction to Engineering Thermodynamics section on system and surroundings explains thermodynamic systems, boundaries, closed systems, open systems, isolated systems, and practical examples such as sealed bottles and piston-cylinder devices.
- Project-specific criteria: In engineering coursework, equipment modeling, or design review, the system boundary and sign convention should match the assigned textbook, project method, or analysis standard.
- Engineering use: Engineers use closed-system references to set up energy balances, identify allowable heat and work interactions, and avoid mixing control-mass and control-volume equations.
Frequently Asked Questions
A closed system is a thermodynamic system with fixed mass. Matter does not cross the system boundary, but energy may cross as heat or work. A sealed piston-cylinder, sealed rigid tank, or unopened bottle warming on a table can be modeled as a closed system when the same mass stays inside.
Yes. A closed system blocks mass transfer, not energy transfer. Heat can cross because of temperature difference, and work can cross through boundary motion, shaft work, electrical work, or other work interactions depending on how the system boundary is drawn.
A closed system allows energy transfer but not mass transfer. An isolated system allows neither mass nor energy transfer. In practice, isolated systems are idealizations, while closed-system models are commonly used for sealed tanks, piston-cylinder devices, and fixed-mass thermodynamic processes.
A piston-cylinder is usually modeled as a closed system when the gas or working fluid inside remains fixed and no mass enters or leaves. The piston may still move, so boundary work can occur even though the mass inside the system stays constant.
Summary and Next Steps
Closed systems are fixed-mass thermodynamic systems. Mass remains inside the boundary, while energy can still cross as heat or work. This makes the closed-system model essential for analyzing sealed tanks, piston-cylinder devices, and control-mass energy balances.
The key engineering skill is choosing the boundary correctly. Once the boundary is drawn, check for mass transfer, energy transfer, moving-boundary work, process assumptions, and sign convention before applying the first law.
Where to go next
Continue your learning path with related Turn2Engineering resources.
-
Open Systems
Learn how thermodynamic analysis changes when mass and energy can cross the system boundary.
-
Isolated Systems
Compare closed systems with the ideal case where neither mass nor energy crosses the boundary.
-
First Law of Thermodynamics
Review the broader energy conservation principle used for closed systems, open systems, and thermodynamic cycles.