Key Takeaways
- Definition: Mass-energy equivalence states that mass and energy are interchangeable, with rest energy given by \(E = mc^2\).
- Main use: Engineers most often use \(E = \Delta m c^2\) to convert a measured mass defect into released or absorbed energy.
- Watch for: The simple form is a rest-energy relation, so do not confuse it with total relativistic energy or ordinary kinetic-energy calculations.
- Outcome: After this page, you should be able to interpret the equation physically, keep units consistent, rearrange it, and sanity-check realistic results.
Table of Contents
How a small mass defect becomes released energy in nuclear fission
The mass-energy equivalence equation relates rest mass to energy and lets engineers convert a tiny mass change into a very large energy value.
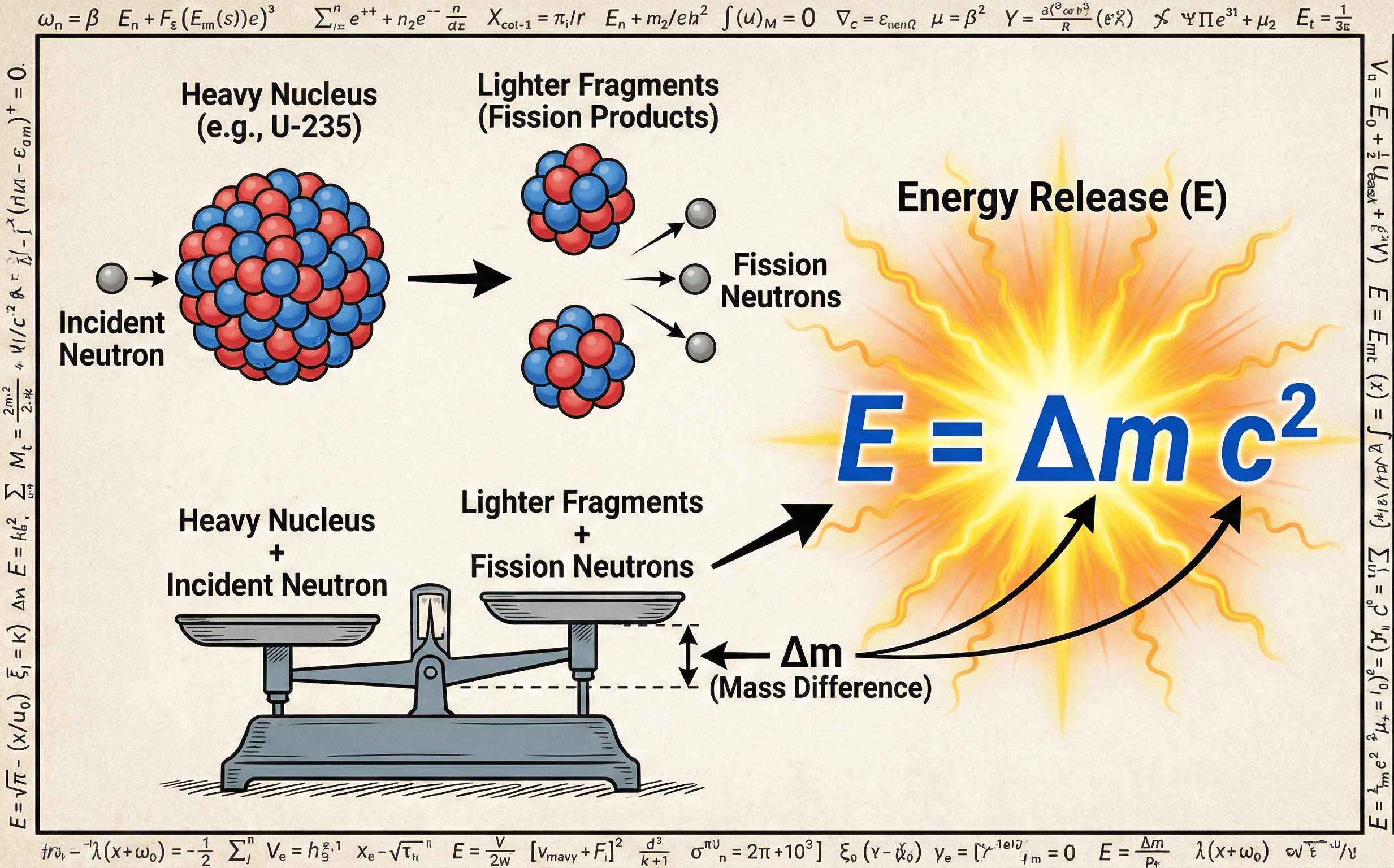
Notice that the image emphasizes \(\Delta m\), not just \(m\). That is the real workflow in reactor physics, particle interactions, and binding-energy problems: compare the initial and final mass, then convert the difference to joules or electron-volts.
What is the mass-energy equivalence equation?
The mass-energy equivalence equation expresses one of the most important results of special relativity: a body with mass possesses an intrinsic amount of energy even when it is not moving. That stored energy is called rest energy, and in its simplest form it is written as \(E = mc^2\).
In engineering work, the equation is most useful when a process changes the mass of a system slightly. Nuclear fission, fusion, radioactive decay, and particle interactions can all produce a small difference between the starting mass and the ending mass. Because the speed of light squared is enormous, even a tiny \(\Delta m\) corresponds to a very large energy release.
This is why the equation matters beyond physics history. It provides the bridge between mass bookkeeping and energy bookkeeping. When you know how much mass is missing, you can estimate the associated thermal, radiant, or kinetic energy.
The mass-energy equivalence formula
The most common form is the rest-energy relation. It tells you how much intrinsic energy is associated with a mass \(m\).
For reaction and binding-energy problems, engineers often use a mass-difference form instead. This version is usually the more practical one for nuclear calculations because it focuses on the amount of mass converted during the process.
Read the first equation as a rest-energy statement and the second as an energy-release statement. That distinction helps prevent one of the most common mistakes on this topic: using the symbol \(m\) when the problem really gives or implies a mass defect.
Variables and units
The equation is simple in appearance, but the symbols carry very specific meaning. Keeping those meanings clear is what makes the calculation trustworthy.
- \(E\) Energy, usually expressed in joules (J) in SI. In nuclear work it is also common to use electron-volts (eV), keV, MeV, or GeV.
- \(m\) Mass or rest mass of the body or system, commonly in kilograms (kg). In atomic and nuclear work, it may also be given in atomic mass units (u).
- \(\Delta m\) Mass defect or mass difference between initial and final states. This is the mass actually converted into energy in a reaction.
- \(c\) Speed of light in vacuum, approximately \(2.998 \times 10^8~\text{m/s}\). For most engineering calculations, \(3.00 \times 10^8~\text{m/s}\) is sufficient.
In SI, use kilograms for mass and meters per second for \(c\). Then the result comes out naturally in joules because \(1~\text{kg}\cdot\text{m}^2/\text{s}^2 = 1~\text{J}\).
If your mass change looks small but your energy result looks enormous, that may be exactly right. The large multiplier is not a bug in the math; it is the point of the equation.
| Variable | Meaning | SI units | Common alternate units | Typical use | Notes |
|---|---|---|---|---|---|
| \(E\) | Energy | J | eV, MeV, GeV | Report released or stored energy | Convert to MeV often for nuclear-scale problems |
| \(m\) | Mass / rest mass | kg | u | Rest-energy calculations | Do not confuse with weight |
| \(\Delta m\) | Mass difference | kg | u | Reaction-energy calculations | Usually the more relevant engineering quantity |
| \(c\) | Speed of light | m/s | ft/s if fully consistent | Constant | Square it before multiplying |
How to rearrange the mass-energy equivalence equation
Most readers look this up because they know the energy and need the equivalent mass, or because they know the mass defect and need the released energy. The algebra is straightforward, but the interpretation matters.
Use \(m = E/c^2\) when you are converting a known rest energy into an equivalent mass. Use \(\Delta m = E/c^2\) when a problem gives the energy released and asks for the missing mass responsible for it.
After rearranging, confirm that your answer makes physical sense. A large energy release should correspond to only a very small mass change, not a large one.
Where engineers use this equation
Although \(E = mc^2\) is famous, it is not an everyday sizing equation like a beam or pipe relation. It shows up where very high energy density matters and where mass changes are measurable or conceptually important.
- Nuclear engineering: estimating fission energy, fusion energy, and binding-energy changes from mass defects.
- Particle and accelerator physics: relating particle rest mass to energy scales and interpreting reaction products.
- Astrophysics and radiation analysis: connecting nuclear processes inside stars and high-energy events to mass loss.
- Energy-conservation education and thermodynamics context: understanding that energy bookkeeping is deeper than just heat and work balances.
One practical insight is that most real engineering problems do not convert all mass into usable energy. Instead, only a fraction of the system mass changes, and only part of the corresponding energy may appear in the form you care about.
Worked example
Example problem
A nuclear process produces a mass defect of \(1.00 \times 10^{-6}~\text{kg}\). Estimate the released energy using the mass-energy equivalence equation.
We will use the reaction form because the problem gives a mass defect, not the total rest mass of the original material.
First square the speed of light:
Then multiply by the mass defect:
That is \(90\) billion joules from only one milligram of mass change. The result feels huge because the energy density implied by \(c^2\) is huge. This is exactly why nuclear energy systems are so different from chemical-energy systems.
If you get a result that seems shockingly large, do not immediately assume an error. Check units first, then remember that the equation is supposed to magnify small mass changes into large energy values.
Assumptions and limits behind the equation
The equation itself is exact within special relativity, but how you apply it in engineering depends on what the symbols represent and what the problem is asking.
- 1 You are interpreting \(m\) as rest mass, not weight and not a moving-body total-energy quantity.
- 2 For reaction problems, the value used is the mass difference \(\Delta m\) between initial and final states.
- 3 You are using a consistent unit system so that the energy result is meaningful.
- 4 You recognize that the equation gives an energy equivalent, not automatically the useful output of a real engineered device.
Neglected factors
The simple rest-energy form does not by itself tell you how the energy is partitioned. In a real process, the energy may leave as radiation, kinetic energy of fragments, heat, neutrino losses, or other forms.
- Momentum effects: moving particles require broader relativistic relations, not just the rest-energy form.
- System efficiency: a reactor or device does not convert all released energy into useful shaft work or electricity.
- Measurement precision: mass defects can be extremely small, so rounding and conversion errors matter.
- Chemical-scale processes: the mass change exists in principle, but it is usually too small to be the practical analysis route.
Mass-energy equivalence vs. related equations
Engineers often confuse the rest-energy equation with broader energy or motion relations. The table below separates the roles clearly.
| Equation / method | Best used for | Key assumption | Main limitation |
|---|---|---|---|
| \(E = mc^2\) | Rest energy of a body | Mass is interpreted as rest mass | Does not directly show motion or momentum effects |
| \(E = \Delta m c^2\) | Energy release from mass defect | Initial and final masses are known consistently | Does not show where the energy goes physically |
| \(KE = \tfrac{1}{2}mv^2\) | Low-speed kinetic energy | Non-relativistic speeds | Not valid near light speed |
| \(E^2 = (pc)^2 + (mc^2)^2\) | Total relativistic energy | Relativistic mechanics framework | More complex than most quick-reference engineering needs |
A good decision rule is simple: if the problem is about missing mass or rest mass, start here. If it is about moving particles at high speed, extend the analysis to the broader relativistic energy-momentum relationship.
Common mistakes and engineering checks
- Using weight instead of mass.
- Using \(m\) when the problem actually gives a mass defect \(\Delta m\).
- Mixing kilograms, grams, and atomic mass units without careful conversion.
- Treating the result as useful device output instead of total energy equivalent.
- Applying the equation as if it were a chemical-energy estimation tool in ordinary thermodynamics problems.
Ask yourself whether the mass change is tiny and the energy result is huge. For this equation, that pattern is usually a sign that the math is behaving correctly.
| Check item | What to verify | Why it matters |
|---|---|---|
| Units | Mass is in kg or fully converted from u or g before substitution | Unit drift can make the answer wrong by orders of magnitude |
| Symbol choice | Use \(m\) for rest energy and \(\Delta m\) for released energy | This is the most common conceptual error |
| Magnitude | The energy should be extremely large relative to the mass change | Helps catch accidental use of \(c\) instead of \(c^2\) |
| Interpretation | Do not confuse energy equivalent with conversion efficiency | Real systems never deliver all theoretical energy as useful output |
Frequently asked questions
It means mass has an intrinsic rest energy equal to the mass times the square of the speed of light. The equation tells you that mass and energy are different expressions of the same underlying physical quantity.
Use \(E = \Delta m c^2\) when you are calculating released or absorbed energy from a difference between starting and ending mass, such as in fission, fusion, decay, or binding-energy problems.
In SI, use kilograms for mass, meters per second for the speed of light, and joules for energy. In nuclear work, it is also common to use atomic mass units for mass and MeV for energy after conversion.
No. The familiar equation is the rest-energy relation. Kinetic energy and full relativistic motion effects belong to broader energy expressions.
Summary and next steps
The mass-energy equivalence equation is simple to write but powerful to use. At the quick-reference level, it tells you that rest mass carries energy and that a small mass defect corresponds to a large energy change.
At the engineering level, the real skill is knowing when to use the rest-energy form, when to use the mass-defect form, and how to keep units, interpretation, and physical realism under control.
Where to go next
Continue your learning path with these curated next steps.
-
Prerequisite: First Law of Thermodynamics
Build the broader conservation-of-energy foundation behind all energy accounting.
-
Current topic: Mass-Energy Equivalence Equation
Return to this page whenever you need the direct relation between mass and energy or a quick check on \(E = \Delta m c^2\).
-
Advanced comparison: Kinetic Energy Equation
Contrast motion-based energy with rest energy so you choose the right model for the problem in front of you.