Key Takeaways
- Core idea: The laws of thermodynamics explain how heat, work, energy, temperature, and entropy behave in physical systems.
- Engineering use: Engineers use these laws to analyze engines, turbines, compressors, heat exchangers, refrigeration cycles, HVAC systems, and power plants.
- What controls it: System boundaries, heat transfer, work transfer, temperature, pressure, mass flow, entropy generation, and assumptions about reversibility control the analysis.
- Practical check: Energy can be conserved while useful work potential is still lost through friction, heat loss, mixing, pressure drops, and other irreversible effects.
Table of Contents
Introduction
The laws of thermodynamics describe how energy moves, changes form, and loses useful work potential in physical systems. The zeroth law defines temperature and thermal equilibrium, the first law conserves energy, the second law explains entropy and irreversibility, and the third law describes behavior near absolute zero.
Visual Guide to the Laws of Thermodynamics
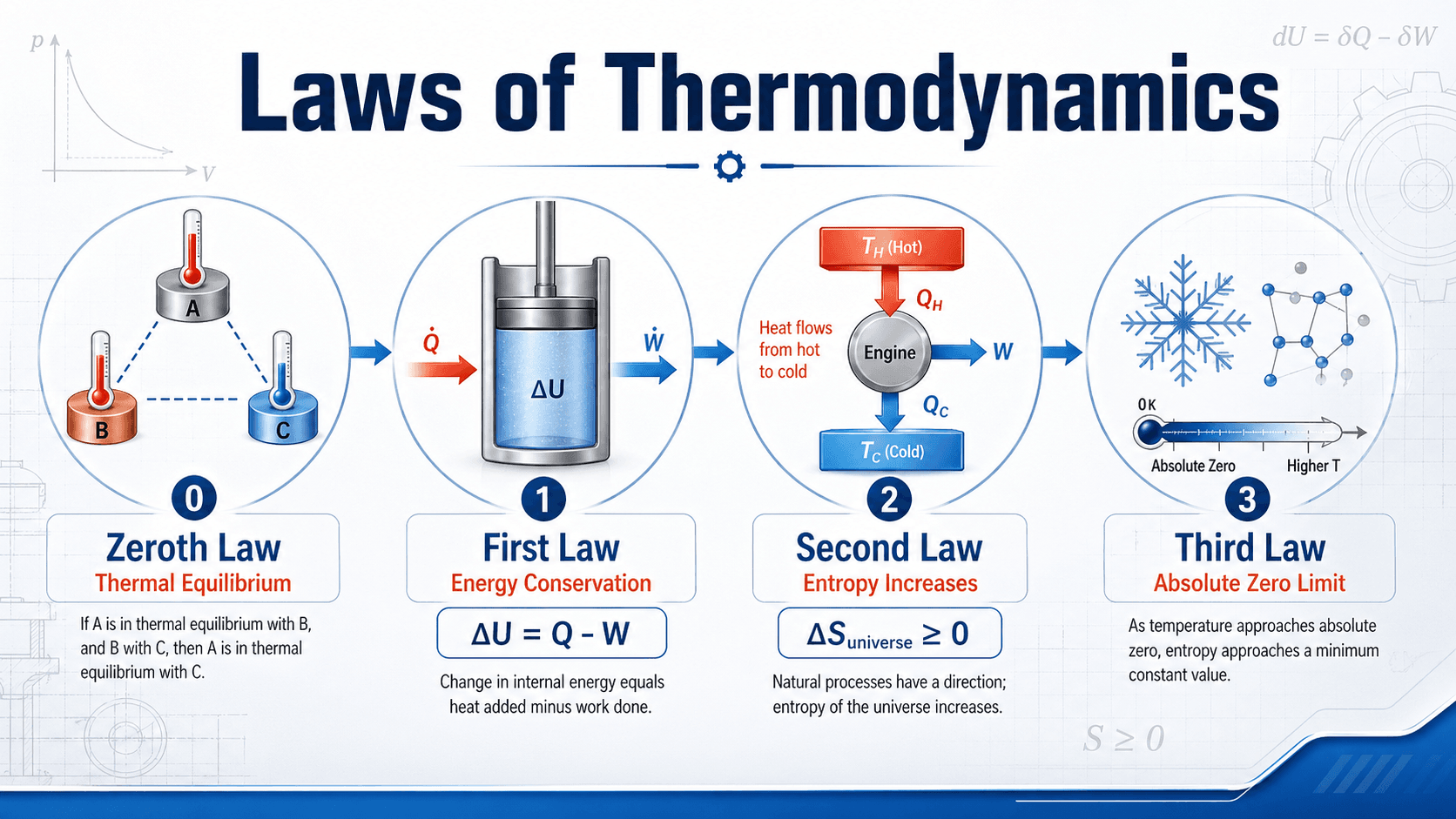
Notice that the laws are not separate trivia facts. They build a framework: define temperature, track energy, determine whether a process is physically realistic, and describe what happens as temperature approaches its theoretical lower limit.
What Are the Laws of Thermodynamics?
The laws of thermodynamics are the governing principles used to describe heat, work, energy, temperature, entropy, and equilibrium. In mechanical engineering, they form the foundation for analyzing power generation, refrigeration, air conditioning, combustion, heat exchangers, turbines, compressors, and many thermal systems.
A useful way to think about the laws is that they answer four different engineering questions: how temperature can be compared, how energy is accounted for, why real processes have a direction, and what happens as matter approaches absolute zero. A page that only says “energy is conserved” misses the practical point: real designs are limited not only by energy quantity, but also by energy quality.
| Law | Simple meaning | Engineering concept |
|---|---|---|
| Zeroth Law | If two systems are each in thermal equilibrium with a third system, they are in thermal equilibrium with each other. | Temperature measurement and thermal equilibrium |
| First Law | Energy cannot be created or destroyed; it can only cross boundaries or change form. | Energy balance, heat, work, and internal energy |
| Second Law | Real processes generate entropy, and heat does not spontaneously flow from cold to hot without work input. | Irreversibility, efficiency limits, and process direction |
| Third Law | As temperature approaches absolute zero, entropy approaches a limiting minimum for a perfect crystal. | Low-temperature behavior and material limits |
How the Four Laws Work Together
Thermodynamics starts by defining the system being studied. A system may be a fixed mass of gas in a piston, a refrigerant flowing through an evaporator, steam expanding through a turbine, or an entire power plant control volume. Once the boundary is clear, the laws describe what can be measured, what must balance, and what can never be achieved in a real process.
Zeroth Law: Temperature and Thermal Equilibrium
The zeroth law gives temperature a physical meaning. If a thermometer reaches thermal equilibrium with a pipe surface, and the pipe surface is in thermal equilibrium with the fluid condition being measured, the thermometer reading can represent the thermal state. Without this idea, temperature would be a vague sensation instead of an engineering property.
First Law: Energy Accounting
The first law is the energy bookkeeping law. Heat added to a system, work done by or on the system, mass crossing a control volume, and changes in internal energy must be accounted for. This is why a turbine, compressor, boiler, condenser, or heat exchanger can be analyzed using energy balances.
Second Law: Direction and Quality of Energy
The second law explains why energy conservation is not enough. A poorly insulated steam line may still conserve energy overall, but the lost heat is no longer as useful for producing work. Friction, throttling, uncontrolled expansion, mixing, heat transfer across large temperature differences, and pressure losses all generate entropy and reduce useful work potential.
Third Law: The Low-Temperature Limit
The third law matters most in cryogenics, materials science, statistical thermodynamics, and low-temperature applications. It explains why absolute zero is a limiting condition rather than a normal temperature target that can be reached with ordinary cooling equipment.
How Engineers Use the Laws of Thermodynamics
Engineers use the laws of thermodynamics to turn physical observations into calculations and design decisions. The same framework applies whether the system is a small compressor, a building HVAC unit, a combustion engine, a vapor-compression refrigeration cycle, or a utility-scale power plant.
- Power systems: turbines, boilers, condensers, and generators are evaluated by tracking heat input, work output, rejected heat, and losses.
- Refrigeration and heat pumps: work input is used to move heat from a lower-temperature region to a higher-temperature region without violating the second law.
- Thermal management: electronics cooling, batteries, heat exchangers, and engine radiators depend on controlled heat transfer and realistic temperature differences.
- Combustion and engines: fuel energy is converted into work, exhaust heat, cooling losses, friction losses, and other forms of rejected energy.
- Process equipment: compressors, nozzles, valves, pumps, and heat exchangers are checked with control-volume energy and entropy balances.
Before applying a thermodynamic equation, define the system boundary first. Many mistakes come from mixing closed-system thinking with control-volume equipment such as turbines, compressors, pumps, and heat exchangers.
Key Concepts That Control Thermodynamic Analysis
The laws of thermodynamics are simple to state, but the analysis depends heavily on boundary selection, property data, heat transfer assumptions, work interactions, mass flow, and whether the process is treated as reversible or irreversible.
| Concept | Why it matters | Engineering implication |
|---|---|---|
| System boundary | Determines what energy, mass, and work cross into or out of the analysis region. | A bad boundary can make a correct equation produce the wrong conclusion. |
| Heat transfer | Heat moves because of temperature difference, not simply because a system contains thermal energy. | Insulation, surface area, temperature gradients, and heat exchanger layout change real performance. |
| Work transfer | Work appears through shaft power, boundary movement, electrical input, or flow work. | Compressors, turbines, pumps, and pistons must be analyzed with the correct work convention. |
| Entropy generation | Entropy generation indicates irreversibility and lost ability to produce useful work. | Lower friction, smaller pressure drops, and better heat-transfer matching improve system efficiency. |
| Property data | Temperature, pressure, enthalpy, internal energy, entropy, and phase state determine the energy balance. | Steam tables, refrigerant tables, gas models, and software must match the working fluid and conditions. |
Important Thermodynamics Equations
The equations below are not the only forms used in thermodynamics, but they represent the core relationships readers usually need when learning the laws. Sign conventions can vary by textbook, so the physical interpretation matters more than memorizing one notation style.
For a simple closed system, \(\Delta U\) is the change in internal energy, \(Q\) is heat added to the system, and \(W\) is work done by the system. If work is done on the system instead, some references write the equation with a different sign convention.
This second-law statement means the total entropy of a system plus its surroundings cannot decrease for a real process. A local entropy decrease may occur, but only when the surroundings compensate with a larger entropy increase.
Thermal efficiency compares useful net work output to heat input. In real heat engines, the second law prevents all heat input from becoming work output because some heat must be rejected and irreversibilities always exist.
- \(Q\) Heat transfer, usually measured in joules, kilojoules, Btu, watts, or Btu/hr depending on whether energy or rate is being evaluated.
- \(W\) Work transfer, often shaft work, boundary work, electrical work, or net cycle work.
- \(U\) Internal energy, a property associated with microscopic molecular energy within a system.
- \(S\) Entropy, a thermodynamic property used to evaluate irreversibility, process direction, and energy quality.
Thermodynamics Design Review Checklist
Use this checklist when reviewing a basic thermodynamic calculation or concept explanation. It helps prevent the most common issue in thermal analysis: getting a mathematically neat answer that does not represent the actual system.
Define the system boundary → identify heat, work, and mass transfers → choose the correct property model → apply the first law for energy balance → apply the second law to test direction, efficiency, and losses → compare the result against physical limits and field conditions.
| Check or decision | What to look for | Why it matters |
|---|---|---|
| Boundary definition | Closed system, open system, isolated system, or control volume. | The selected boundary determines which energy and mass terms belong in the balance. |
| Heat direction | Temperature difference and sign convention for heat entering or leaving. | Heat transfer direction must match the second law unless work input is supplied. |
| Work convention | Work done by the system versus work done on the system. | A sign error can reverse the meaning of compression, expansion, turbine output, or pump input. |
| Property state | Pressure, temperature, phase, quality, enthalpy, internal energy, and entropy. | Incorrect property data can make a cycle analysis look plausible but physically wrong. |
| Irreversibility check | Friction, throttling, mixing, pressure drop, heat loss, and finite temperature differences. | These effects explain why real systems underperform ideal thermodynamic models. |
Example: Why a Heat Engine Cannot Be 100% Efficient
A heat engine receives heat from a hot source, produces useful work, and rejects remaining heat to a cooler sink. The first law says the energy must balance, but the second law says the engine cannot convert every unit of heat input into useful work in a cycle.
Energy Balance View
Suppose an engine receives \(1000 \, \text{kJ}\) of heat from combustion or a boiler and produces \(350 \, \text{kJ}\) of net work. The first-law balance requires the remaining energy to leave as rejected heat, losses, or stored energy change depending on the system boundary. For a complete cycle, stored energy returns to its original value, so the rejected heat is part of the cycle behavior.
Second-Law Interpretation
The second law explains why rejected heat is not just poor design. Heat engines require interaction with a lower-temperature sink, and real devices also generate entropy through friction, combustion irreversibility, heat transfer across finite temperature differences, pressure losses, and non-ideal expansion. Better design can reduce losses, but it cannot remove the second-law limit.
When reviewing an engine, turbine, or power cycle, do not judge performance only by whether energy balances. Also ask where useful work potential is destroyed.
Engineering Judgment and Field Reality
Textbook thermodynamics often begins with ideal gases, reversible processes, perfect insulation, frictionless pistons, steady flow, and clean property states. Real equipment usually operates with fouling, pressure drop, heat leakage, transient loads, sensor error, phase change, imperfect combustion, control limits, and maintenance conditions that shift performance away from ideal analysis.
Experienced engineers use the laws of thermodynamics as a reality check, not just a formula source. If a claimed device produces work without a heat source, cools without work input or another driving mechanism, exceeds ideal cycle limits, or ignores rejected heat, the claim should be treated with skepticism.
Many real failures are not caused by violating the first law; they are caused by underestimating second-law losses such as pressure drop, heat leakage, fouling, poor insulation, throttling, and off-design operation.
When This Breaks Down
The laws of thermodynamics are fundamental, but simplified explanations and beginner equations can break down when the system is poorly defined, the process is highly transient, the material model is wrong, or microscopic behavior becomes important.
- Unclear boundaries: a calculation can fail when heat, work, and mass transfers are assigned to the wrong side of the system boundary.
- Non-ideal working fluids: ideal gas assumptions can be poor near saturation, at high pressure, or during phase change.
- Transient operation: startup, shutdown, rapid cycling, and control changes may require time-dependent energy balances.
- Multiphase behavior: boilers, condensers, evaporators, and two-phase refrigerant lines need property data beyond simple gas equations.
- Microscale and quantum effects: low-temperature and microscopic systems may require statistical thermodynamics or quantum treatment.
Common Mistakes and Practical Checks
Many thermodynamics mistakes come from using the right law in the wrong context. A clean equation does not guarantee a correct analysis unless the boundary, sign convention, property state, and physical process are all consistent.
- Confusing heat with temperature: temperature is a property; heat is energy transfer caused by temperature difference.
- Saying energy is “lost” without context: energy is conserved, but useful work potential can be destroyed by irreversibility.
- Ignoring entropy generation: a device may satisfy the first law and still be impossible or inefficient under the second law.
- Using the wrong sign convention: first-law equations vary depending on whether work is defined as work done by or on the system.
- Treating ideal cycles as real performance: Carnot, Otto, Diesel, Rankine, and vapor-compression models are useful references, not perfect field predictions.
Do not use the first law alone to judge whether a proposed system is realistic. The first law checks energy accounting; the second law checks direction, irreversibility, and efficiency limits.
Useful References and Engineering Context
Thermodynamics is usually learned from engineering textbooks, property tables, manufacturer data, and equipment-specific codes or standards. The references below are context sources rather than a substitute for project-specific design requirements.
- Engineering thermodynamics textbooks: Used for the first law, second law, entropy, property tables, control-volume analysis, and thermodynamic cycles.
- ASHRAE Handbook resources: Commonly used in HVAC and refrigeration work where thermodynamics, psychrometrics, cooling loads, heat pumps, and refrigerants are central.
- Steam and refrigerant property data: Used to obtain enthalpy, entropy, saturation conditions, quality, and phase information for real fluids.
- Manufacturer performance data: Used to compare ideal thermodynamic expectations with actual compressors, turbines, heat exchangers, chillers, pumps, and heat pumps.
Frequently Asked Questions
The laws of thermodynamics explain how temperature, heat, work, energy, and entropy behave in physical systems. The zeroth law defines thermal equilibrium, the first law conserves energy, the second law explains entropy and process direction, and the third law describes entropy behavior near absolute zero.
There are four commonly named laws: the zeroth, first, second, and third laws. Many beginner resources focus on the first three because the zeroth law was named later and the third law is most important in low-temperature physics, materials, and statistical thermodynamics.
The second law is important because it explains irreversibility, entropy generation, and why real systems cannot convert all heat into useful work. It sets practical efficiency limits for engines, turbines, refrigeration cycles, heat pumps, and many energy-conversion systems.
Entropy can decrease locally within a system, but only if the surroundings gain enough entropy that the total entropy of the combined system and surroundings does not decrease. This distinction is important when analyzing refrigerators, heat pumps, biological systems, and controlled thermal processes.
Summary and Next Steps
The laws of thermodynamics explain the behavior of temperature, energy, heat, work, entropy, and low-temperature limits. For mechanical engineering, they are the foundation behind engines, refrigeration systems, turbines, compressors, HVAC equipment, power cycles, and thermal management.
The practical workflow is to define the system boundary, account for heat and work, use the first law to balance energy, use the second law to test direction and losses, and remember that ideal models must be checked against field conditions, property data, and equipment limitations.
Where to go next
Continue your learning path with related Turn2Engineering resources.
-
First Law of Thermodynamics
Learn how engineers track heat, work, and internal energy using energy balances.
-
Second Law of Thermodynamics
Study entropy, irreversibility, process direction, and why real energy systems have efficiency limits.
-
Thermodynamic Cycles
See how the laws are applied to repeating cycles used in engines, power plants, and refrigeration systems.
